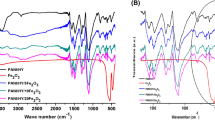
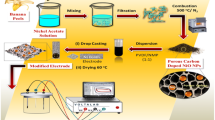
Abstract
The technological advancement relevant to energy storage devices are paramount considering the energy demand that will substantially increase in the future. Although electrodes are playing an essential role in these devices, the novel electrode technology carries a significant weight that can produce colossal impact on energy storage applications. The present study describe the advancement of graphite-clay composite electrodes, improving properties and performance by incorporating additional materials, which results in a quaternary electrode that is not only succeeded in energy storage but also in electroanalytical applications. The mechanical compression and thermal activation are the only technological processing steps that employed in fabricating quaternary composite electrode, which consists of graphite, colloidal graphite, kaolinite and cement (GCGKCeCE). The ideal voltammogram signal with higher sensitivity to both Fe2+ and Fe3+, which are 1.01 A m mol−1 and 1.07 A m mol−1, respectively, than the commercial electrodes is well-noticeable with GCGKCeCE, indicating better analyte detection ability. Aniline electropolymerization on GCGKCeCE led to PANI-GCGKCeCE that has low interface (0.30 Ω) and charge transfer (0.46 Ω) resistance due to a unique interwoven nanofiber network consisting of villi-like surface projections, which resulted from synergistic interactions between GCGKCeCE and PANI. The supercapacitor cell fabricated with an identical PANI-GCGKCeCE account for very high specific capacitance, which is 1029 F g−1 at 5 mV s−1, confirming the feasibility in energy storage applications. The 99% coulombic efficiency that observed with supercapacitor cell even after 1800 cycles indicates an improved cyclic stability, which ensures better reversibility and lifespan.
Graphical abstract









Similar content being viewed by others
References
Abaspour M, Pattipati KR, Shahrrava B, Balasingam B (2022) Robust approach to battery equivalent-circuit-model parameter extraction using electrochemical impedance spectroscopy. Energies 15(23):9251
Bellido-Milla D, Cubillana-Aguilera LM, El Kaoutit M, Hernández-Artiga MP, Hidalgo-Hidalgo de Cisneros JL, Naranjo-Rodríguez I, Palacios-Santander JM (2013) Recent advances in graphite powder-based electrodes. Anal Bioanal Chem 405(11):3525–3539
Chanda M (1980) Electrical, electronic and optical properties. In: Chanda M (ed) Science of engineering materials: volume 3 engineering properties. Macmillan Education, London, pp 61–126
Chen W-C, Wen T-C, Hu C-C, Gopalan A (2002) Identification of inductive behavior for polyaniline via electrochemical impedance spectroscopy. Electrochim Acta 47(8):1305–1315
Chen W, Rakhi RB, Alshareef HN (2013) Morphology-dependent enhancement of the pseudocapacitance of template-guided tunable polyaniline nanostructures. J Phys Chem C 117(29):15009–15019
Chen B-R, Police YR, Li M, Chinnam PR, Tanim TR, Dufek EJ (2023) A mathematical approach to survey electrochemical impedance spectroscopy for aging in lithium-ion batteries. Front Energy Res 11:167
Dhirde AM, Dale NV, Salehfar H, Mann MD, Han TH (2010) Equivalent electric circuit modeling and performance analysis of a pem fuel cell stack using impedance spectroscopy. IEEE Trans Energy Convers 25(3):778–786
Elgrishi N, Rountree KJ, McCarthy BD, Rountree ES, Eisenhart TT, Dempsey JL (2018) A practical beginner’s guide to cyclic voltammetry. J Chem Educ 95(2):197–206
Gogotsi Y, Penner RM (2018) Energy storage in nanomaterials–capacitive, pseudocapacitive, or battery-like? ACS Nano 12(3):2081–2083
Hu C-C, Chu C-H (2001) Electrochemical impedance characterization of polyaniline-coated graphite electrodes for electrochemical capacitors—effects of film coverage/thickness and anions. J Electroanal Chem 503(1):105–116
Huang H, Zeng X, Li W, Wang H, Wang Q, Yang Y (2014) Reinforced conducting hydrogels prepared from the in situ polymerization of aniline in an aqueous solution of sodium alginate. J Mater Chem A 2(39):16516–16522
Karunadasa KSP, Manoratne CH, Pitawala HMTGA, Rajapakse RMG (2019) A potential working electrode based on graphite and montmorillonite for electrochemical applications in both aqueous and molten salt electrolytes. Electrochem Commun 108:106562
Karunadasa KSP, Rathnayake D, Manoratne C, Pitawala A, Rajapakse G (2021) A binder-free composite of graphite and kaolinite as a stable working electrode for general electrochemical applications. Electrochem Sci Adv 1(4):e2100003
Li G-R, Feng Z-P, Zhong J-H, Wang Z-L, Tong Y-X (2010a) Electrochemical synthesis of polyaniline nanobelts with predominant electrochemical performances. Macromolecules 43(5):2178–2183
Li X, Li X, Dai N, Wang G, Wang Z (2010b) Preparation and electrochemical capacitance performances of super-hydrophilic conducting polyaniline. J Power Sources 195(16):5417–5421
Liu P, Yan J, Guang Z, Huang Y, Li X, Huang W (2019) Recent advancements of polyaniline-based nanocomposites for supercapacitors. J Power Sources 424:108–130
Ma Y, Hou C, Zhang H, Qiao M, Chen Y, Zhang H, Zhang Q, Guo Z (2017) Morphology-dependent electrochemical supercapacitors in multi-dimensional polyaniline nanostructures. J Mater Chem A 5(27):14041–14052
Madhushanka PMH, Karunadasa KSP, Gamini Rajapakse RM, Manoratne CH, Bandara HMN (2024) Low-cost composite electrode consisting of graphite, colloidal graphite and montmorillonite with enhanced electrochemical performance for general electroanalytical techniques and device fabrication. Chem Pap 78(1):633–643
Mehdinia A, Dejaloud M, Jabbari A (2013) Nanostructured polyaniline-coated anode for improving microbial fuel cell power output. Chem Pap 67(8):1096–1102
Mirceski V, Gulaboski R, Lovric M, Bogeski I, Kappl R, Hoth M (2013) Square-wave voltammetry: a review on the recent progress. Electroanalysis 25(11):2411–2422
Mondal S, Sangaranarayanan MV (2016) Permselectivity and thickness-dependent ion transport properties of overoxidized polyaniline: a mechanistic investigation. Phys Chem Chem Phys 18(44):30705–30720
Ngamchuea K, Eloul S, Tschulik K, Compton RG (2014) Planar diffusion to macro disc electrodes—What electrode size is required for the Cottrell and Randles-Sevcik equations to apply quantitatively? J Solid State Electrochem 18(12):3251–3257
Qin Q, He F, Zhang W (2016) One-step electrochemical polymerization of polyaniline flexible counter electrode doped by graphene. J Nanomater 2016:1076158
Okpoli CC (2013) Sensitivity and resolution capacity of electrode configurations. Int J Geophys 2013:608037
Rajkumar M, Hsu C-T, Wu T-H, Chen M-G, Hu C-C (2015) Advanced materials for aqueous supercapacitors in the asymmetric design. Prog Nat Sci Mater Int 25(6):527–544
Rathnayake DT, Karunadasa KSP, Wijekoon ASK, Manoratne CH, Rajapakse RMG (2022) Low-cost ternary composite of graphite, kaolinite and cement as a potential working electrode for general electrochemical applications. Chem Pap 76(10):6653–6658
Rathnayake DT, Karunadasa KSP, Wijekoon ASK, Manoratne CH, Gamini Rajapakse RM, Pitawala HMTGA (2023) Polyaniline-conjugated graphite–montmorillonite composite electrode prepared by in situ electropolymerization for supercapacitor applications. Chem Pap 77(5):2923–2928
Sarkar S, Akshaya R, Ghosh S (2020) Nitrogen doped graphene/CuCr2O4 nanocomposites for supercapacitors application: effect of nitrogen doping on coulombic efficiency. Electrochim Acta 332:135368
Shanthi PM, Hanumantha PJ, Ramalinga K, Gattu B, Datta MK, Kumta PN (2019) Sulfonic acid based complex framework materials (CFM): nanostructured polysulfide immobilization systems for rechargeable lithium-sulfur battery. J Electrochem Soc 166(10):A1827
Sk MM, Yue CY (2014) Synthesis of polyaniline nanotubes using the self-assembly behavior of vitamin C: a mechanistic study and application in electrochemical supercapacitors. J Mater Chem A 2(8):2830–2838
Smith MA (1982) A consideration of graphite electrodes. IEEE Trans Ind Appl 18(4):431–434
Song E, Choi J-W (2013) Conducting polyaniline nanowire and its applications in chemiresistive sensing. Nanomaterials 3(3):498–523
Sumboja A, Wang X, Yan J, Lee PS (2012) Nanoarchitectured current collector for high rate capability of polyaniline based supercapacitor electrode. Electrochim Acta 65:190–195
Tamilarasan P, Ramaprabhu S (2014) Ionic liquid-functionalized partially exfoliated multiwalled carbon nanotubes for high-performance supercapacitors. J Mater Chem A 2(34):14054–14063
Temenoff JS, Mikos AG (2008) Biomaterials: the intersection of biology and materials science. Pearson/Prentice Hall Upper Saddle River
Wang Q, Li J-L, Gao F, Li W-S, Wu K-Z, Wang X-D (2008) Activated carbon coated with polyaniline as an electrode material in supercapacitors. New Carbon Mater 23(3):275–280
Wang K, Huang J, Wei Z (2010) Conducting polyaniline nanowire arrays for high performance supercapacitors. J Phys Chem C 114(17):8062–8067
Wang X, Deng J, Duan X, Liu D, Guo J, Liu P (2014) Crosslinked polyaniline nanorods with improved electrochemical performance as electrode material for supercapacitors. J Mater Chem A 2(31):12323–12329
Wang H, Lin J, Shen ZX (2016) Polyaniline (PANi) based electrode materials for energy storage and conversion. J Sci Adv Mater Devices 1(3):225–255
Wring SA, Hart JP (1992) Chemically modified, carbon-based electrodes and their application as electrochemical sensors for the analysis of biologically important compounds: a review. Analyst 117(8):1215–1229
Xie Y, Du H (2015) Electrochemical capacitance of a carbon quantum dots–polypyrrole/titania nanotube hybrid. RSC Adv 5(109):89689–89697
Yang W, Gao Z, Song N, Zhang Y, Yang Y, Wang J (2014) Synthesis of hollow polyaniline nano-capsules and their supercapacitor application. J Power Sources 272:915–921
Zhang Ja, Wang P, Liu Y, Cheng Z (2021) Variable-order equivalent circuit modeling and state of charge estimation of lithium-ion battery based on electrochemical impedance spectroscopy. Energies 14(3):769
Zhao H-B, Yuan L, Fu Z-B, Wang C-Y, Yang X, Zhu J-Y, Qu J, Chen H-B, Schiraldi DA (2016) Biomass-based mechanically strong and electrically conductive polymer aerogels and their application for supercapacitors. ACS Appl Mater Interfaces 8(15):9917–9924
Acknowledgements
This work was financially supported by the Industrial Technology Institute of Sri Lanka (Grant No. TG 21/193 and TG 24/246).
Funding
Industrial Technology Institute Sri Lanka, TG 21-193, TG 24/246
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no competing interests to declare that are relevant to the content of this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Madhushanka, P.M.H., Karunadasa, K.S.P. & Rajapakse, R.M.G. Graphite-colloidal graphite–kaolinite-cement quaternary composite electrode with improved synergetic matrix effect towards efficient energy storage and electroanalytical applications. Chem. Pap. (2024). https://doi.org/10.1007/s11696-024-03502-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11696-024-03502-6