Abstract
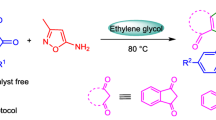
Calcium iodide (CaI2) catalyzes cycloaddition reaction in Nenitzescu synthesis to afford 5-hydroxyindoles from corresponding quinones and enamines. This method introduces CaI2 as a catalyst in performing this type of condensation reaction for the first time and presents advantages in using nontoxic metal, work-up simplicity, functional group tolerance, and can compete with other methods to overcome the current problems such as low yield and polymerization side reactions.
Graphical abstract




Similar content being viewed by others
References
Allen GR (2011) The Synthesis of 5-Hydroxyindoles by the Nenitzescu Reaction. Organic Reactions. Wiley, Hoboken, pp 337–454
Begouin JM, Niggemann M (2013) Calcium-based lewis acid catalysts. Chem A Eur J 19:8030–8041. https://doi.org/10.1002/chem.201203496
Berger M, Gray JA, Roth BL (2009) The expanded biology of serotonin. Annu Rev Med 60:355–366. https://doi.org/10.1146/annurev.med.60.042307.110802
Drago RS, Nusz JA, Courtright RC (1974) Solvation contributions to enthalpies measured in methylene chloride. J Am Chem Soc 96:2082–2086. https://doi.org/10.1021/ja00814a016
Drake SR, Otway DJ (1991) The synthesis of metal organic compounds of calcium, strontium and barium by ammonia gas-saturated ethereal solvents. J Chem Soc Chem Commun, 517–519. https://doi.org/10.1039/C39910000517
Dumitrascu F, Ilies MA (2021) Recent advances in the Nenitzescu indole synthesis (1990–2019), 1st edn. Elsevier Inc., Amsterdam
Ivan BC, Caira MR, Dumitrascu F (2020) Nenitzescu indole synthesis: 1929–2019 unexpected formation of a pyrrole-azepine hybrid in the Nenitzescu indole synthesis: a reinvestigation. Rev Chim 71:51–57. https://doi.org/10.37358/RC.20.5.8112
Katkevica D, Trapencieris P, Boman A et al (2004) The Nenitzescu reaction: an initial screening of experimental conditions for improvement of the yield of a model reaction. J Chemom 18:183–187. https://doi.org/10.1002/cem.863
Ketcha DM, Wilson LJ, Portlock DE (2000) The solid-phase Nenitzescu indole synthesis. Tetrahedron Lett 41:6253–6257. https://doi.org/10.1016/S0040-4039(00)00697-3
Kuckländer U (1973) Beobachtungen zum mechanismus der nenitzescu-reaktion-II. Tetrahedron 29:921–927. https://doi.org/10.1016/0040-4020(73)80040-7
Kumari A, Singh RK (2019) Medicinal chemistry of indole derivatives: Current to future therapeutic prospectives. Bioorg Chem 89:103021. https://doi.org/10.1016/j.bioorg.2019.103021
Monti SA, Johnson WO (1970) Position selective mannich reactions of some 5- and 6-hydroxyindoles. Tetrahedron 26:3685–3694. https://doi.org/10.1016/S0040-4020(01)92947-3
Nguyen DT, Lenstra DC, Mecinović J (2015) Chemoselective calcium-catalysed direct amidation of carboxylic esters. RSC Adv 5:77658–77661. https://doi.org/10.1039/c5ra18288c
Nozari MS, Jensen CD, Drago RS (1973) Eliminating solvation contributions to the enthalpy of adduct formation in weakly polar, acidic solvents. J Am Chem Soc, pp 3162–3165
Pauff SM, Miller SC (2012) 基因的改变NIH Public Access. Bone 78:711–716. https://doi.org/10.1016/j.tet.2011.06.040.Indole
Poletto JF, Allen GR, Sloboda AE, Weiss MJ (1973) Synthesis and antiinflammatory evaluation of Certain 5-Alkoxy-2,7-dialkyltryptamines. J Med Chem 16:757–765. https://doi.org/10.1021/jm00265a004
Proskurnina EV, Izmailov DY, Sozarukova MM et al (2020) Antioxidant potential of antiviral drug umifenovir. Molecules 25:1–11. https://doi.org/10.3390/molecules25071577
Satta G, Usala E, Solinas A et al (2021) Nenitzescu synthesis of 5-Hydroxyindoles with zinc, iron and magnesium salts in Cyclopentyl Methyl Ether. Euro J Org Chem 2021:5835–5842. https://doi.org/10.1002/ejoc.202101045
Shannon RD (1976) Database of Ionic Radii. In: Acta Cryst. http://abulafia.mt.ic.ac.uk/shannon/
Stempel E, Gaich T (2016) Cyclohepta[b]indoles: a privileged structure motif in natural products and drug design. Acc Chem Res 49. https://doi.org/10.1021/acs.accounts.6b00265
Suryavanshi PA, Sridharan V, Menéndez JC (2010) Expedient, one-pot preparation of fused indoles via CAN-catalyzed three-component domino sequences and their transformation into polyheterocyclic compounds containing pyrrolo[1,2-a]azepine fragments. Org Biomol Chem 8:3426–3436. https://doi.org/10.1039/c004703a
Tsubogo T, Shimizu S, Kobayashi S (2013) Chiral calcium iodide for asymmetric mannich-type reactions of malonates with imines providing β-aminocarbonyl compounds. Chem Asian J 8:872–876. https://doi.org/10.1002/asia.201300102
Van Order RB, Lindwall HG (1942) Indole. Chem Rev 30:69–96
Velezheva VS, Kornienko AG, Topilin SV et al (2006) Lewis Acid Catalyzed Nenitzescu Indole Synthesis. ChemInform 37. https://doi.org/10.1002/chin.200652120
Velezheva VS, Sokolov AI, Kornienko AG et al (2008) The role of a Lewis acid in the Nenitzescu indole synthesis. Tetrahedron Lett 49:7106–7109. https://doi.org/10.1016/j.tetlet.2008.09.087
Weissman SA, Zewge D (2005) Recent advances in ether dealkylation. Tetrahedron 61:7833–7863. https://doi.org/10.1016/j.tet.2005.05.041
Acknowledgements
This work was supported by the Research Council of Shahid Beheshti University of Medical Sciences.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
There are no conflicts to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Teymori, A., Sedaghat, A. & Kobarfard, F. Ca-mediated Nenitzescu synthesis of 5-hydroxyindoles. Chem. Pap. 77, 1791–1795 (2023). https://doi.org/10.1007/s11696-022-02463-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-022-02463-y