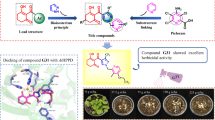
Abstract
In this work, a series of novel indole 3-substituted-[1,2,4]triazole derivatives was designed and synthesized, and their structures were characterized by 1H, 13C NMR, high-resolution mass spectrometry, and single-crystal diffraction. Moreover, the antibacterial activities of the title compounds were evaluated against Xanthomonas oryzae pv. oryzae (Xoo), Pseudomonas syringae pv. actinidae (Psa), and Xanthomonas axonopodis pv. citri (Xac) by a turbidimetric method. Results showed that the title compounds had excellent bioactivity against Xoo, and their 50% effective concentration (EC50) ranged from 2.32 to 35.4 µg/mL, which were better than those of the positive control drugs Bismerthiazol (38.6 µg/mL) and Thiodiazole-copper (58.4 µg/mL). In particular, the EC50 values of compounds 53 and 9 against Xoo were 3.1 and 2.3 µg/mL, respectively.
Graphical abstract















Similar content being viewed by others
References
Borthakur SK, Konwar LJ, Nath G, Kalita PK, Borthakur S (2020) Synthesis of some novel 7-substituted [1,3,4]thiadiazolo[3,2-c][1,3,5]thiadiazine-6,6-dioxides with antifungal evaluation against rice pathogens. J Heterocycl Chem 57(12):4210–4214. https://doi.org/10.1002/jhet.4128
Flieger J, Trębacz H, Pizoń M, Plazińska A, Plaziński W, Kowalska A, Szczęsna A, Plech T (2019) Thermodynamic study of new antiepileptic compounds by combining chromatography on the phosphatidylcholine biomimetic stationary phase and differential scanning calorimetry. J Sep Sci 42(16):2628–2639. https://doi.org/10.1002/jssc.201900248
Geng YH, Wei ZQ, Xu Z, Naasda LX, Zhang S, Guo HY, Liu ML, Feng LS, You XF (2019) Design, synthesis and antibacterial evaluation of 1-[(1R,2S)-2-fluorocyclopropyl]ciprofloxacin-(4-methyl-3-aryl)-1,2,4-triazole-5(4H)-thione hybrids. Rev Roum Chim 64(1):101–107. https://doi.org/10.33224/rrch.2019.64.1.10
Hovsepyan TR, Dilanyan SV, Minasyan NS, Arsenyan FG, Nersesyan LE, Agaronyan AS, Danielyan IS (2018) New DNA methylation inhibitors based on 1,2,4-triazole thioether derivatives. Russ J Gen Chem 88(7):1390–1396. https://doi.org/10.1134/S1070363218070071
Li P, Shi L, Yang X, Yang L, Chen XW, Wu F, Shi QC, Xu WM, He M, Hu DY, Song BA (2014) Design, synthesis, and antibacterial activity against rice bacterial leaf blight and leaf streak of 2,5-substituted-1,3,4-oxadiazole/thiadiazole sulfone derivative. Bioorg Med Chem 24(7):1677–1680. https://doi.org/10.1016/j.bmcl.2014.02.060
Li P, Hu DY, Xie DD, Chen JX, Jin LH, Song BA (2018) Design, synthesis, and evaluation of new sulfone derivatives containing a 1,3,4-oxadiazole moiety as active antibacterial agents. J Agric Food Chem 66(12):3093–3100. https://doi.org/10.1021/acs.jafc.7b06061
Liu DY, Zhang J, Zhao L, He WJ, Liu ZJ, Gan XH, Song BA (2019) First discovery of novel pyrido[1,2-a]pyrimidinone mesoionic compounds as antibacterial agents. J Agric Food Chem 67(43):11860–11866. https://doi.org/10.1021/acs.jafc.9b03606
Liu HW, Ji QT, Ren GG, Wang F, Su F, Wang PY, Zhou X, Wu ZB, Li Z, Yang S (2020) Antibacterial functions and proposed modes of action of novel 1,2,3,4-Tetrahydro-β-carboline derivatives that possess an attractive 1,3-diaminopropan-2-ol pattern against rice bacterial blight, kiwifruit bacterial canker, and citrus bacterial canker. J Agric Food Chem 68(45):12558–12568. https://doi.org/10.1021/acs.jafc.0c02528
Mane YD, Patil SS, Biradar DO, Khade BC (2018) 5-Bromo-1-(4-chlorobenzyl)-1H-indole-2-carboxamides as new potent antibacterial agents. Heterocycl Commun 24(6):327–332. https://doi.org/10.1515/hc-2018-0107
Rezki N, Al-Yahyawi AM, Bardaweel SK, Al-Blewi FF, Aouad MR (2015) Synthesis of novel 2,5-disubstituted-1,3,4-thiadiazoles clubbed 1,2,4-triazole, 1,3,4-thiadiazole, 1,3,4-oxadiazole and/or schiff base as potential antimicrobial and antiproliferative agents. Molecules 20(9):16048–16067. https://doi.org/10.3390/molecules200916048
Shi J, Ding MH, Luo N, Wan SR, Li PJ, Li JH, Bao XP (2020) Design, synthesis, crystal structure, and antimicrobial evaluation of 6-fluoroquinazolinylpiperidinyl-containing 1,2,4-triazole mannich base derivatives against pbacteria and fungi. J Agric Food Chem 68(36):9613–9623. https://doi.org/10.1021/acs.jafc.0c01365
Shujah S, Ali S, Khalid N, Alam MJ, Ahmad S, Meetsma A (2018) Synthesis, spectroscopic characterization, X-ray structure, DFT calculations, and antimicrobial studies of diorganotin (IV) complexes of monotopic oxygen nitrogen donor Schiff base. Chem Pap 72(4):903–919. https://doi.org/10.1007/s11696-017-0333-2
Tao QQ, Liu LW, Wang PY, Long QS, Zhao YL, Jin LH, Xu WM, Chen Y, Li Z, Yang S (2019) Synthesis and in vitro and in vivo biological activity evaluation and quantitative proteome profiling of oxadiazoles bearing flexible heterocyclic patterns. J Agric Food Chem 67(27):7626–7639. https://doi.org/10.1021/acs.jafc.9b02734
Toma A, Mogosan C, Vlase L, Leonte D, Zaharia V (2017) Synthesis, characterization and evaluation of the anti-inflammatory activity of thiazolo[3,2-b][1,2,4]triazole derivatives bearing pyridin-3/4-yl moiety. Med Chem Res 26(10):2602–2613. https://doi.org/10.1007/s00044-017-1959-x
Wang MW, Zhu HH, Wang PY, Zeng D, Wu YY, Liu LW, Wu ZB, Li Z, Yang S (2019a) Synthesis of thiazolium-labeled 1,3,4-oxadiazole thioethers as prospective antimicrobials: in vitro and in vivo bioactivity and mechanism of action. J Agric Food Chem 67(46):12696–12708. https://doi.org/10.1021/acs.jafc.9b03952
Wang PY, Zhou L, Zhou J, Wu ZB, Xue W, Song BA, Yang S (2016) Synthesis and antibacterial activity of pyridinium-tailored 2,5-substituted-1,3,4-oxadiazole thioether/sulfoxide/sulfone derivatives. Bioorg Med Chem 26(4):1214–1217. https://doi.org/10.1016/j.bmcl.2016.01.029
Wang PY, Wang MW, Zeng D, Xiang M, Rao JR, Liu QQ, Liu LW, Wu ZB, Li Z, Song BA, Yang S (2019b) Rational optimization and action mechanism of novel imidazole (or imidazolium)-labeled 1,3,4-oxadiazole thioethers as promising antibacterial agents against plant bacterial diseases. J Agric Food Chem 67(13):3535–3545. https://doi.org/10.1021/acs.jafc.8b06242
Yu XQ, Zhu XY, Zhou Y, Li QL, Hu Z, Li T, Tao J, Dou ML, Zhang M, Shao Y, Sun RF (2019) Discovery of N-aryl-pyridine-4-ones as novel potential agrochemical fungicides and bactericides. J Agric Food Chem 67(50):13904–13913. https://doi.org/10.1021/acs.jafc.9b06296
Zhou WJ, Zhang L, Xiao W, Chen HJ, Wu WN, Ouyang GP (2017) Synthesis and biological activity of anthranilic diamide derivatives incorporating 1,3,4-oxadiazole or nitrogen-containing saturated heterocyclic moieties. J Heterocycl Chem 54(2):1423–1429. https://doi.org/10.1002/jhet.2724
Zhou X, Feng YM, Qi PY, Shao WB, Wu ZB, Liu LW, Wang Y, Ma HD, Wang PY, Li Z, Yang S (2020) Synthesis and docking study of N‑(Cinnamoyl)‑N′‑(substituted)acryloyl hydrazide derivatives containing pyridinium moieties as a novel class of filamentous temperature-sensitive protein Z inhibitors against the intractable Xanthomonas oryzae pv. oryzae infections in rice. J Agric Food Chem 68(31):8132–8142. https://doi.org/10.1021/acs.jafc.0c01565
Zhu ZS, Wang SB, Deng XQ, Liu DC, Quan ZS (2014) Synthesis and anticonvulsant activity evaluation of 4-butyl-5-(4-alkoxyphenyl)-2H-1,2,4-triazole-3(4H)-ones. Lett Drug Des Discov 11(5):628–635. https://doi.org/10.2174/1570180810666131122003939
Acknowledgements
This research was financially supported by the Natural Science Foundation of China (Nos. 21867004, 22007022), Guizhou Provincial Natural Science Foundation (No. ZZK[2021]), Top Science and Technology Talent Program of Guizhou Education Department (2022), Guizhou Provincial Department of Education (Qjh KY Zi [2021] No. 041), and innovation and entrepreneurship training program for college students (GuiZhou University 2020033).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interests
The author declares that there are no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhu, L., Tang, SY., Chen, DP. et al. Synthesis and antibacterial activity of indole 3-substituted-[1,2,4]triazole derivatives. Chem. Pap. 77, 895–907 (2023). https://doi.org/10.1007/s11696-022-02393-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-022-02393-9