Abstract
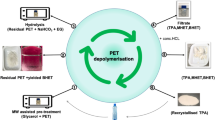
The crude glycerol is the main by-product of biodiesel production and contains large amounts of potassium (or sodium) soap. These alkaline compounds may be reacted with ester groups of polyethylene terephthalate (PET) to form the corresponding terephthalic acid salts. In this work, the process of PET depolymerization with crude glycerol with a high content of potassium salts was studied. Depolymerization is the multistage process and proceeds through the formation of oligomers of various molecular weights. Subsequently, the resulting intermediate compounds decompose to low molecular weight oligomers (dimers and trimers), up to dipotassium terephthalate (DPT). A general reaction scheme and a mathematical model of the process are represented, and rate constants of all reactions are estimated.












Similar content being viewed by others
References
Amaro LP, Coiai S, Ciardelli F, Passaglia E (2015) Preparation and testing of a solid secondary plasticizer for PVC produced by chemical degradation of post-consumer PET. Waste Manag 46:68–75. https://doi.org/10.1016/j.wasman.2015.09.005
Bhogle CS, Pandit AB (2017) Ultrasound-assisted alkaline hydrolysis of waste poly(ethylene terephthalate) in aqueous and non-aqueous media at low temperature. Indian Chem Eng 60(2):1–19. https://doi.org/10.1080/00194506.2017.1310634
Colomines G, Rivas F, Lacoste M-L, Robin J-J (2005) Study of polyurethane formulations containing diols obtained via glycolysis of poly(ethylene terephthalate) (PET) by oligoesters diols through a reactive extrusion process. Macromol Mater Eng 290(7):710–720. https://doi.org/10.1002/mame.200400391
Dzhabarov G, Sapunov V, Kozlovskiy R, Makarova E, Kha PD, Voronov M, Shadrina V, Nhi TD, Kurneshova T (2020) A method of polyethylene terephthalate depolymerization by biodiesel wastes. Pet Coal 62(1):19–26
Gupta P, Bhandari S (2019) Chemical depolymerization of PET Bottles via ammonolysis and aminolysis. In: Sabu T, Ajay VR, Krishnan K, Abitha VK, Martin GT (Eds) Recycling of polyethylene terephthalate bottles 1st Edition, pp 109–134. https://doi.org/10.1016/B978-0-12-811361-5.00006-7
Hoang CN, Le TTN, Hoang QD (2018) Glycolysis of poly(ethylene terephthalate) waste with diethyleneglycol under microwave irradiation and ZnSO4·7H2O catalyst. Polym Bull. https://doi.org/10.1007/s00289-018-2369-z
Lee WD, Im SS, Lim H-M, Kim K-J (2006) Preparation and properties of layered double hydroxide/poly(ethylene terephthalate) nanocomposites by direct melt compounding. Polymer 47:1364–1371. https://doi.org/10.1016/j.polymer.2005.12.056
Li X-K, Lu H, Guo W-Z, Cao G-P, Liu H-L, Shi Y-H (2014) Reaction kinetics and mechanism of catalyzed hydrolysis of waste PET using solid acid catalyst in supercritical CO2. AIChE J 61(1):200–214. https://doi.org/10.1002/aic.14632
Liu F, Chen J, Li Z, Ni P, Ji Y, Meng Q (2013) Alcoholysis of poly(ethylene terephthalate) to produce dioctyl terephthalate with sub- and super-critical isooctyl alcohol. J Anal Appl Pyrol 99:16–22. https://doi.org/10.1016/j.jaap.2012.11.008
Liu Q, Li R, Fang T (2015) Investigating and modeling PET methanolysis under supercritical conditions by response surface methodology approach. Chem Eng J 270:535–541. https://doi.org/10.1016/j.cej.2015.02.039
López-Fonseca R, González-Velasco JR, Gutiérrez-Ortiz JI (2009) A shrinking core model for the alkaline hydrolysis of PET assisted by tributylhexadecylphosphonium bromide. Chem Eng J 146(2):287–294. https://doi.org/10.1016/j.cej.2008.09.039
Ovalle-Sánchez AA, Elizondo-Martínez P, Pérez-Rodríguez NA, Hernández-Fernández E, Sánchez-Anguiano MG (2017) Degradation of poly(ethyleneterephtalate) waste to obtain oligomers using a zinc complex as catalyst. J Chil Chem Soc 62(4):3741–3745. https://doi.org/10.4067/s0717-97072017000403741
Panasyuk G, Azarova LA, Khaddaj M, Budova G, Voroshilov I, Grusha T, Izotov AD (2003) Preparation and properties of sodium, potassium, magnesium, calcium, and aluminum terephthalates. Inorg Mater 39:1292–1297. https://doi.org/10.1023/B:INMA.0000008916.84994.e1
Ragaert K, Delva L, Van Geem K (2017) Mechanical and chemical recycling of solid plastic waste. Waste Manag 69:24–58. https://doi.org/10.1016/j.wasman.2017.07.044
Sako T, Sugeta T, Otake K, Takebayashi Y, Kamizawa C, Tsugumi M, Hongo M (1998) Kinetic study on depolymerization of poly(ethylene terephthalate) with methanol at high temperature and pressure. Kobunshi Ronbunshu 55(11):685–690
Saron C, Bimestre B (2014) Mechanical recycling of PET waste from non-woven fabrics by reactive extrusion with chain extenders. J Res Updat Polym Sci 3:170–177. https://doi.org/10.6000/1929-5995.2014.03.03.4
Todorov N, Radenkov M, TodorovaD D (2015) Utilization of crude glycerol and waste polyethylene terephthalate for production of alkyd resins. J Chem Technol Metall 50:240–248
Wu H, Lv S, He Y, Qu J-P (2019) The study of the thermomechanical degradation and mechanical properties of PET recycled by industrial-scale elongational processing. Polym Test 77:105882. https://doi.org/10.1016/j.polymertesting.2019.04.029
Zope VS, Mishra S (2008) Kinetics of neutral hydrolytic depolymerization of PET (polyethylene terephthalate) waste at higher temperature and autogenious pressures. J Appl Polym Sci 110(4):2179–2183. https://doi.org/10.1002/app.28190
Acknowledgements
This work was financially supported by the Russian Foundation for Basic Research (Scientific Project No. 18-29-24009).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Dzhabarov, G.V., Sapunov, V.N., Shadrina, V.V. et al. A kinetic study on the depolymerization of polyethylene terephthalate waste with crude glycerol. Chem. Pap. 75, 6035–6046 (2021). https://doi.org/10.1007/s11696-021-01780-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-021-01780-y