Abstract
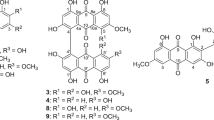
A new anthraquinone, 1,6-dihydroxy-2-hydroxymethyl-5-methoxy-9,10-anthracenedione (1), along with four known compounds, including 1,2-dimethoxyanthraquinone (2), morindone-5-methylether (3), scopoletin (4), and fraxidin (5) were isolated from the methylene chloride extract of the stem bark of Morinda lucida Benth. Structure elucidation was accomplished by analysis of the 1D and 2D NMR, IR and mass spectra. Compounds 1, 2, 4 and 5 were tested against five Salmonella strains. The minimal inhibitory concentrations (MICs) and minimal bactericidal concentrations (MBCs) were determined. The results indicated that the four compounds have different antibacterial activities, with MIC values ranging from 8 to 256 µg/mL. Among these four compounds, the best activity was observed for compound 4 against Salmonella enterica subsp. enterica serovar Typhimurim (STM), with a MIC value of 8 µg/mL. Compound 1 showed selective antibacterial activity toward the five Salmonella strains, with MICs of 16 µg/mL against STM, 32 µg/mL against S. enteritidis (STE), 64 µg/mL against S. enterica subsp. enterica serovar Typhi (ST) and S. enterica subsp. enterica serovar Typhi (ST566), and 128 µg/mL against S. enteritidis, S. enterica subsp. enterica serovar Typhi ATCC 6539 (STS). Compounds 1, 2, 4, and 5 have bactericidal effects (MBC/MIC ≤ 4) against the tested Salmonella strains, except for compound 4, which is bacteriostatic to STM with a MBC/MIC value of 16.


Similar content being viewed by others
References
Adeleye OO, Ayeni OJ, Ajamu MA (2018) Traditional and medicinal uses of Morinda lucida. J Med Plants Stud 6:249–254
Almeida ÉS, de Oliveira D, Hotza D (2019) Properties and applications of Morinda citrifolia (noni): a review. Compr Rev Food Sci Food Saf 18:883–909. https://doi.org/10.1111/1541-4337.12456
Babokhov P, Sanyaolu AO, Oyibo WA, Fagbenro-Beyioku AF, Iriemenam NC (2013) A current analysis of chemotherapy strategies for the treatment of human African trypanosomiasis. Pathog Glob Health 107:242–252. https://doi.org/10.1179/2047773213Y.0000000105
Bi K, Bedi G, Koffi A, Chalchat JC, Guessan T (2010) Volatiles constituents from leaves of Morinda morindoïdes (Rubiaceae): a medicinal plant from the Ivory Coast. Open Nat Prod J 3:6–9. https://doi.org/10.2174/1874848101003010006
Bussmann RW, Hennig L, Giannis A, Ortwein J, Kutchan TM, Feng X (2013) Anthraquinone content in noni (Morinda citrifolia L.). Evid Based Complement Altern Med 2013:208378. https://doi.org/10.1155/2013/208378
Carbonnelle B, Denis F, Marmonier A, Pinon G, Vague R (1987) Medical bacteriology: usual techniques. SIMEP, Paris
Cimanga RK, Tona GL, Mesia GK, Kambu OK, Bakana DP, Kalenda PDT, Penge AO, Muyembe JJT, Totté J, Pieters L, Vlietinck AJ (2006) Bioassay-guided isolation of antimalarial triterpenoid acids from the leaves of Morinda lucida. Pharm Biol 44:677–681. https://doi.org/10.1080/13880200601009123
Cuong NM, Long PQ, Son NT, Van DT, Khanh PN, Cuong TD, Ha VT, Tram NCT, Viet DQ, Huong TT (2019) Phenylethanoid glucoside and anthraquinone compounds from Morinda longissima Y. Z. ruan roots. Vietnam J Chem 54:133–138
Demagos G, Baltus W, Höfle G (1981) New anthraquinones and anthraquinone glycosides from Morinda lucida. Z Naturforsch B 36:1180–1184. https://doi.org/10.1515/znb-1981-0925
Ee CL, Kiang LM, Taufiq-Yap YH, Go R (2005) Ferulic acid ester from Jatropha podagrica (Euphorbiaceae). Malaysian J Chem 7:45–48
Fraga B, Quintana N, Díaz C (2009) Anthraquinones from natural and transformed roots of Plocama pendula. Chem Biodivers 6:182–192. https://doi.org/10.1002/cbdv.200800047
Gatsing D, Tchakoute V, Ngamga D, Kuiate JR, Tamokou JDD, Nji-Nkah BF, Tchouanguep FM, Fodouop SPC (2009) In vitro antibacterial activity of Crinum Purpurascens Herb. leaf extract against the Salmonella species causing typhoid fever and its toxicological evaluation. Iran J Med Sci 34:126–136
Inada AC, Figueiredo PS, dos Santos-Eichler RA, de Cássia FK, Hiane PA, de Castro AP, de Cássia Avellaneda Guimarães R (2017) Morinda citrifolia Linn (noni) and its potential in obesity-related metabolic dysfunction. Nutrients 9:540. https://doi.org/10.3390/nu9060540
Khan NMMU, Hossain M (2015) Scopoletin and β-sitosterol glucoside from roots of Ipomoea digitata. J Pharmacogn Phytochem 4:5–7
Koffi B, Kouamé F, Claude K, Bedi G, Robins R, Adjou A, Félix T, N’Dri A, Adiko M, Akoubet A, Gondo G, Diomandé D, Akpa J, Tea I, Robins R, Adjou A, Tonzibo FZ (2019) A novel anthraquinone from Morinda lucida Benth (Rubiaceae). J Pharmacogn Phytochem 8:11–14
Koumaglo K, Gbeassor M, Nikabu O, de Souza C, Werner W (1992) Effects of three compounds extracted from Morinda lucida on Plasmodium falciparum. Planta Med 58:533–534. https://doi.org/10.1055/s-2006-961543
Lin CF, Ni CL, Huang YL, Sheu SJ, Chen CC (2007) Lignans and anthraquinones from the fruits of Morinda citrifolia. Nat Prod Res 21:1199–1204. https://doi.org/10.1080/14786410601132451
Longue Ekon JP, Zra T, Lobe Songue J, Wondja Ngoko ML, Ngassoum MB, Talla E, Kamdem Waffo AF, Sewald N, Wansi JD (2020) New anthraquinone derivatives from the stem barks of Morinda lucida Benth. Phytochem Lett 39:94–98. https://doi.org/10.1016/j.phytol.2020.06.010
Manojlović I, Bogdanović-Dusanović G, Gritsanapan W, Manojlović N (2006) Isolation and identification of anthraquinones of Caloplaca cerina and Cassia tora. Chem Pap 60:466–468. https://doi.org/10.2478/s11696-006-0084-y
Mfonku NA, Mbah JA, Kodjio N, Gatsing D, Zhan J (2020) Isolation and selective glycosylation of antisalmonellal anthraquinones from the stem bark of Morinda lucida Benth. (Rubiaceae). Phytochem Lett 37:80–84. https://doi.org/10.1016/j.phytol.2020.04.011
Murray RD, Méndez J, Brown S (1982) The natural coumarins: occurrence, chemistry, and biochemistry. John Wiley and Sons, New Jersey
Newman DJ, Cragg GM (2020) Natural products as sources of new drugs over the nearly four decades from 01/1981 to 09/2019. J Nat Prod 83:770–803. https://doi.org/10.1021/acs.jnatprod.9b01285
Newton SM, Lau C, Gurcha SS, Besra GS, Wrigh CW (2002) The evaluation of forty-three plant species for in vitro antimycobacterial activities; isolation of active constituents from Psoralea corylifolia and Sanguinaria canadensis. J Ethnopharmacol 79:57–67. https://doi.org/10.1016/s0378-8741(01)00350-6
Noiarsa P, Ruchirawat S, Otsuka H, Kanchanapoom T (2006) A new iridoid glucoside from the Thai medicinal plant, Morinda elliptica Ridl. J Nat Med 60:322–324. https://doi.org/10.1007/s11418-006-0003-0
O’Neil MJ, Heckelman PE, Koch CB, Roman KJ (2006) The Merck index: an encyclopedia of chemicals, drugs, and biologicals, 14th edn. Merck and Co. Inc, Whitehouse Station, NJ
Osman CP, Ismail NH, Ahmad R, Ahmat N, Awang K, Jaafar FM (2010) Anthraquinones with antiplasmodial activity from the roots of Rennellia elliptica Korth. (Rubiaceae). Molecules 15:7218–7226. https://doi.org/10.3390/molecules15107218
Pawlus AD, Su BN, Keller WJ, Kinghorn AD (2005) An anthraquinone with potent quinone reductase-inducing activity and other constituents of the fruits of Morinda citrifolia (noni). J Nat Prod 68:1720–1722. https://doi.org/10.1021/np050383k
Petrovska BB (2012) Historical review of medicinal plants’ usage. Pharmacogn Rev 6:1–5. https://doi.org/10.4103/0973-7847.95849
Rath G, Ndonzao M, Hostettmann K (1995) Antifungal anthraquinones from Morinda lucida. Int J Pharmacogn 33:107–114. https://doi.org/10.3109/13880209509055208
Ridley RG (2002) Medical need, scientific opportunity and the drive for antimalarial drugs. Nature 415:686–693. https://doi.org/10.1038/415686a
Rumzhum N, Sohrab M, Al-Mansur M, Mansur A, Rahman M, Hasan C, Rashid M (2012) Secondary metabolites from Jatropha podagrica Hook. J Phys Sci 23:29–37
Saifi M, Beg T, Harrath H, Altayalan F, Al-Quraishy S (2013) Antimalarial drugs: mode of action and status of resistance. Afr J Pharm Pharmacol 7:148–156. https://doi.org/10.5897/AJPPX12.015
Shao L, Wang G, Guo M, Yang L, Jiang D, Li R, Zhu J (2020) Determination of 9,10-anthraquinone in tea consumed in Shandong Province of China. Chem Pap. https://doi.org/10.1007/s11696-020-01254-7
Singh R (2015) Medicinal plants: a review. J Plant Sci 3:50–55. https://doi.org/10.11648/j.jps.s.2015030101.18
Su BN, Pawlus AD, Jung HA, Keller WJ, McLaughlin JL, Kinghorn AD (2005) Chemical constituents of the fruits of Morinda citrifolia (noni) and their antioxidant activity. J Nat Prod 68:592–595. https://doi.org/10.1021/np0495985
The Plant List: a working list of all plant species. Version 1.1. (2013) Royal Botanic Gardens, Kew. http://www.theplantlist.org/browse/A/Rubiaceae/Morinda/.
West BJ, Zhou BN (2008) Identification of major aroma compounds in the leaf of Morinda citrifolia Linn. J Nat Med 62:485–487. https://doi.org/10.1007/s11418-008-0269-5
Acknowledgements
JAM acknowledges the Fulbright Fellowship that supported his visit to Utah State University, USA. The Bruker Avance III HD Ascend-500 NMR instrument was funded by the National Science Foundation Award CHE–1429195.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mfonku, N.A., Tadjong, A.T., Kamsu, G.T. et al. Isolation and characterization of antisalmonellal anthraquinones and coumarins from Morinda lucida Benth. (Rubiaceae). Chem. Pap. 75, 2067–2073 (2021). https://doi.org/10.1007/s11696-020-01460-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-020-01460-3