Abstract
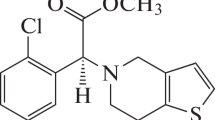
A poly (4-aminobenzene sulfonic acid)-modified glassy carbon electrode (p-ABSA/GCE) was fabricated by electropolymerization. It was found that phentolamine mesylate, an effective and important cardiovascular dilatation drug with low redox activity at the glassy carbon electrode (GCE) can produce a sensitive well-defined anodic peak current at p-ABSA/GCE. Investigation indicated that the oxidation of phentolamine at p-ABSA/GCE was one electron/one proton transfer process which was controlled by adsorption. In optimal conditions, the anodic peak current was linear to the concentrations of phentolamine over the range of 5.0 × 10–7–1.0 × 10–5 M with a detection limit of 2.0 × 10–7 M. The modified electrode showed good stability and reproducibility. The electroanalytical method proposed was successfully applied to the determination of phentolamine mesylate in pharmaceutical formulations.







Similar content being viewed by others
References
Bros FD, Wolshin EM (1978) Determination of phentolamine in blood and urine by high-performance liquid-chromatography. Anal Chem 50:521–525. https://doi.org/10.1021/ac50025a038
Godbillon J, Carnis G (1981) Determination of the major metabolite of phentolamine in human plasma and urine by high-performance liquid chromatography. J Chromatogr B 222:461–466. https://doi.org/10.1016/S0378-4347(00)84147-X
Jin GY, Zhang YZ, Cheng WX (2005) Poly(p-aminobenzene sulfonic acid)-modified glassy carbon electrode for simultaneous detection of dopamine and ascorbic acid. Sens Actuat B 107:528–534. https://doi.org/10.1016/j.snb.2004.11.018
Laviron E (1979) General expression of the linear potential sweep voltammogram in the case of diffusionless electrochemical systems. J Electroanal Chem 101:19–28. https://doi.org/10.1016/S0022-0728(79)80075-3
Li YF, Ye Z, Luo PL, Li Y, Ye BX (2014) A sensitive voltammetric sensor for salbutamol based on MWNTs composite nano-Au film modified electrode. Anal Methods 6:1928–1935. https://doi.org/10.1039/C3AY41758A
Liu LJ, Gou YQ, Gao X, Zhang P, Chen WX, Feng SL, Hua FD, Li YD (2014) Electrochemically reduced graphene oxide-based electrochemical sensor for the sensitive determination of ferulic acid in A. sinensis and biological samples. Mater Sci Eng C 42:227–233. https://doi.org/10.1016/j.msec.2014.05.045
Mikami E, Ohno T, Matsumoto H (2002) Simultaneous identification/determination system for phentolamine and sildenafil as adulterants in soft drinks advertising roborant nutrition. Forensic Sci Int 130:140–146. https://doi.org/10.1016/S0379-0738(02)00368-7
Ohnuki Y, Ohsaka T, Matsuda H, Oyama N (1983) Permselectivity of films prepared by electrochemical oxidation of phenol and aminoaromatic compounds. J Electroanal Chem 158:55–67. https://doi.org/10.1016/S0022-0728(83)80338-6
Padma-Nathan H, Goldstein I, Klimberg I et al (2002) Long-term safety and efficacy of oral phentolamine mesylate (Vasomax) in men with mild to moderate erectile dysfunction. Int J Impot Res 14:266–270. https://doi.org/10.1038/sj.ijir.3900885
Shrivastav R, Satsangee SP, Jain R (2013) Effect of surfactants on the voltammetric response and determination of an antihypertensive drug phentolamine at boron doped diamond electrode. ECS Trans 50(54):23–36. https://doi.org/10.1149/05054.0023ecst
Sioufi A, Pommier F, Mangoni P, Gauron S, Metayer JP (1981) The gas-chromatographic determination of phentolamine (Regitine) in human plasma and urine. J Chromatogr B 222:429–435. https://doi.org/10.1016/S0378-4347(00)84143-2
Ugarte F, Hurtado-Coll A (2002) Comparison of the efficacy and safety of sildenafil citrate (Viagra) and oral phentolamine for the treatment of erectile dysfunction. Int J Impot Res 14(Suppl 2):S48–S53. https://doi.org/10.1038/sj.ijir.3900898
Volkov A, Tourillon G, Lacaze PC, Dubois JE (1980) Electrochemical polymerization of aromatic amines: IR, XPS and PMT study of thin film formation on a Pt electrode. J Electroanal Chem 115:279–291. https://doi.org/10.1016/S0022-0728(80)80332-9
Wang J, Jiang M, Lu F (1998) Electrochemical quartz crystal microbalance investigation of surface fouling due to phenol oxidation. J Electronanl Chem 444:127–132. https://doi.org/10.1016/S0022-0728(97)00583-4
Webster GK, Lemmer RR, Greenwald S (2003) Rapid analysis of phentolamine by high-performance liquid chromatography. J Chromatogr Sci 41:57–62. https://doi.org/10.1093/chromsci/41.2.57
Acknowledgements
The authors thank senior professor Wei Ding (Polymer Institute of Northeast Petroleum University) for his help and useful discussions.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Fan, S., Ji, L., Mao, G. et al. Voltammetric determination of phentolamine mesylate in pharmaceutical formulations at poly (4-aminobenzene sulfonic acid)-modified glassy carbon electrode. Chem. Pap. 74, 4411–4417 (2020). https://doi.org/10.1007/s11696-020-01250-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-020-01250-x