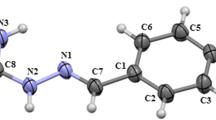
Abstract
Carbohydrazones are compounds that are increasingly studied due to their wide potential biological activity. Monocarbohydrazones (mCHs), as one of the carbohydrazone derivatives, so far have been poorly investigated. For a more detailed study, in this paper, eighteen compounds of monocarbohydrazones (eight known and ten newly synthesized derivatives) were synthesized and characterized using NMR and IR spectroscopy. As carbohydrazones show E/Z isomerization caused by the presence of the imino group, some of the synthesized mCHs are in the form of a mixture of these two isomers. The effects of specific and nonspecific solvent–solute interactions on the UV absorption maxima shifts were evaluated using linear free energy relationships principles, i.e., using Kamlet–Taft’s and Catalan’s models. For more information about interactions between dissolved substance and the surrounding medium, correlations have been made with Hansen’s solubility parameters. The influence of the structure on the spectral behavior of the compounds tested was interpreted using Hammett’s equation. Experimentally obtained physicochemical properties of mCHs were compared to and confirmed with computational methods that included TD-DFT calculations and MP2 geometry optimizations.
Graphic abstract











Similar content being viewed by others
References
Antić-Jovanović A (2006) Molekulska spektroskopija-spektrohemijski aspekti, fakultet za fizičku hemiju, Beograd
Babahan I, Anil H, Sarikavakli N (2011) Synthesis of novel tetraoxime derivative with hydrazone side groups and its metal complexes. Turk J Chem 35:613–624. https://doi.org/10.3906/kim-1003-105
Bacchi A, Carcelli M, Pelagatti P, Pelizzi C, Pelizzi G, Zani F (1999) Antimicrobial and mutagenicactivity of some carbono-and thiocarbonohydrazone ligands and their copper(II), iron(II) and zinc(II) complexes. J Inorg Biochem 75:123–133. https://doi.org/10.1016/S0162-0134(99)00045-8
Bauer M, Rollberg A, Barth A, Spange S (2008) Differentiating between dipolarity and polarizability effects of aolvents using the aolvatochromism of barbiturate dyes. Eur J Org Chem 2008:4475–4481. https://doi.org/10.1002/ejoc.200800355
Becke AD (1993) A new mixing of Hartree–Fock and local density-functional theories. J Chem Phys 98:1372–1377. https://doi.org/10.1063/1.464304
Božic AR, Filipovic NR, Novakovic IT, Bjelogrlic SK, Nikolic JB, Drmanic SŽ, Marinkovic AD (2017) Synthesis, antioxidant and antimicrobial activity of carbohydrazones. J Serb Chem Soc 82:495–508. https://doi.org/10.2298/JSC161220045B
Božić A, Marinković A, Bjelogrlić S, Todorović TR, Cvijetić IN, Novaković I, Muller CD, Filipović NR (2016) Quinoline based mono- and bis-(thio)carbohydrazones: synthesis, anticancer activity in 2D and 3D cancer and cancer stem cell models. RSC Adv 6:104763–104781. https://doi.org/10.1039/c6ra23940d
Božić AR, Filipović NR, Verbić TŽ, Milčić MK, Todorović TR, Cvijetić IN, Klisurić OR, Radišić M, Marinković AD (2017) A detailed experimental and computational study of monocarbohydrazones. Arab J Chem. https://doi.org/10.1016/j.arabjc.2017.08.010
Catalán J (2009) Toward a generalized treatment of the solvent effect based on four empirical scales: dipolarity (SdP, a new scale), polarizability (SP), acidity (SA), and basicity (SB) of the medium. J Phys Chem B 113:5951–5960. https://doi.org/10.1021/jp8095727
Catalán J, Hopf H (2004) Empirical treatment of the inductive and dispersive components of solute-solvent interactions: the solvent polarizability (SP) scale. Eur J Org Chem 2004:4694–4702. https://doi.org/10.1002/ejoc.200400311
Dragancea D, Talmaci N, Shova S, Novitchi G, Darvasiová D, Rapta P, Breza M, Galanski M, Kožı́šek J, Martins NMR, Martins LMDRS, Pombeiro AJL, Arion VB (2016) Vanadium(V) complexes with substituted 1,5-bis(2-hydroxybenzaldehyde) carbohydrazones and their use as catalyst precursors in oxidation of cyclohexane. Inorg Chem 55:9187–9203. https://doi.org/10.1021/acs.inorgchem.6b01011
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Petersson GA, Nakatsuji H, Li X, Caricato M, Marenich AV, Bloino J, Janesko BG, Gomperts R, Mennucci B, Hratchian HP, Ortiz JV, Izmaylov AF, Sonnenberg JL, Williams-Young D, Ding F, Lipparini F, Egidi F, Goings J, Peng B, Petrone A, Henderson T, Ranasinghe D, Zakrzewski VG, Gao J, Rega N, Zheng G, Liang W, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Throssell K, Montgomery JA, Peralta JE, Ogliaro F, Bearpark MJ, Heyd JJ, Brothers EN, Kudin KN, Staroverov VN, Keith TA, Kobayashi R, Normand J, Raghavachari K, Rendell AP, Burant JC, Iyengar SS, Tomasi J, Cossi M, Millam JM, Klene M, Adamo C, Cammi R, Ochterski JW, Martin RL, Morokuma K, Farkas O, Foresman JB, Fox DJ (2016) Gaussian 16, Revision B.01, Gaussian, Inc., Wallingford CT
Gangarapu K, Manda S, Jallapally A, Thota S, Karki SS, Balzarini J, De Clercq E, Tokuda H (2014) Synthesis of thiocarbohydrazide and carbohydrazide derivatives as possible biologically active agents. Med Chem Res 23:1046–1056. https://doi.org/10.1007/s00044-013-0684-3
Gholivand K, Farshadfar K, Roe SM, Gholami A, Esrafili MD (2016) Structural and photophysical characterization of mono- and binuclear Cu(I) complexes based on carbohydrazones: a combined experimental and computational study. CrystEngComm 18:2873–2884. https://doi.org/10.1039/c5ce02208h
Glendening ED, Reed AE, Carpenter JE, Weinhold F (2003) NBO Version 3.1. Gaussian Inc., Pittsburgh
Hammett P (1937) The effect of structure upon the reactions of organic compounds. Benzene derivatives. J Am Chem Soc 59:96–103. https://doi.org/10.1021/ja01280a022
Hansen ChM (2007) Solubility parameters: a user’s handbook. CRC Press-Taylor & Francis group, Boca Raton
Iqbal S, Saleem M, Azim MK, Taha M, Salar U, Khan KM, Perveen S, Choudhary MI (2017) Carbohydrazones as new class of carbonic anhydrase inhibitors: synthesis, kinetics and ligand docking studies. Bioorg Chem 72:89–101. https://doi.org/10.1016/j.bioorg.2017.03.014
Jacquemin D, Le Bahers T, Adamo C, Ciofini I (2012) What is the ‘‘best’’ atomic charge model to describe through-space charge-transfer excitations? Phys Chem Chem Phys 14:5383–5388. https://doi.org/10.1039/C2CP40261K
Kajal A, Bala S, Sharma N, Kamboj S, Saini V (2014) Therapeutic potential of hydrazones as anti-inflammatory agents. Int J Med Chem. https://doi.org/10.1155/2014/761030
Kamlet MJ, Taft RW (1976) The solvatochromic comparison method. I. The β-scale of solvent hydrogen-bond acceptor (HBA) basicities. J Am Chem Soc 98:377–383. https://doi.org/10.1021/ja00418a009
Kamlet MJ, Abboud JLM, Taft RW (1977) The solvatochromic comparison method. 6. The π scale of solvent polarities. J Am Chem Soc 99:6027–6038. https://doi.org/10.1021/ja00460a031
Kamyabi MA, Soleymani-Bonoti F, Bikas R, Hosseini-Monfared H (2017) Oxygen reduction catalyzed by a carbohydrazone based compound at liquid/liquid interfaces. J Electroanal Chem 794:235–243. https://doi.org/10.1016/j.jelechem.2017.04.021
Kurzer F, Wilkinson M (1970) Chemistry of carbohydrazide and thiocarbohidrazide. Chem Rev 70:111–149. https://doi.org/10.1021/cr60263a004
Le Bahers T, Adamo C, Ciofini I (2011) A qualitative index of spatial extent in charge-transfer excitations. J Chem Theory Comput 7:2498–2506. https://doi.org/10.1021/ct200308m
Montazerozohori M, Yadegari S, Naghiha A (2014) Synthesis, characterization, electrochemical behavior and antibacterial/antifungal activities of [Cd(L)X2] complexes with a new Schiff base ligand. J Serb Chem Soc 79:793–804. https://doi.org/10.2298/JSC130520110M
Okawara T, Imai K, Ochiai T (2006) Free radical scavenger containing semicarbazide derivatives. WO 2006093124
Peach MJG, Tozer DJ (2009) Illustration of a TDDFT spatial overlap diagnostic by basis function exponent scaling. J Mol Struct Theochem 914:110–114. https://doi.org/10.1016/j.theochem.2009.03.009
Peach MJG, Benfield P, Helgaker T, Tozer DJ (2008) Excitation energies in density functional theory: an evaluation and a diagnostic test. J Chem Phys 128:044118. https://doi.org/10.1063/1.2831900
Peach MJG, Le Sueur CR, Ruud K, Guillaume M, Tozer DJ (2009) TDDFT diagnostic testing and functional assessment for triazene chromophores. Phys Chem Chem Phys 11:4465–4470. https://doi.org/10.1039/b822941d
Reed AE, Weinhold F (1985) Natural localized molecular orbitals. J Chem Phys 83:1736–1740. https://doi.org/10.1063/1.449360
Reichardt Ch (2003) Solvents and solvent effects in organic chemistry, 3rd edn. Wiley-VCH, Weinheim
Riedel F, Spange S (2012) Solvatochromism of catechol derivatives—solute/solvent interactions. J Phys Org Chem 25:1261–1268. https://doi.org/10.1002/poc.3003
Rollas S, Küçükgüzel SG (2007) Biological activities of hydrazone derivatives. Molecules 12:1910–1939. https://doi.org/10.3390/12081910
Sánchez Rojas F, Cristófol Alcaraz E, Cano Pavón JM (1994) Determination of aluminium in water by flow injection with fluorimetric detection by using salicylaldehydecarbohydrazone as reagent in a micellar medium. Analyst 119:1221–1223. https://doi.org/10.1039/AN9941901221
Shebl M, Khalil SME (2015) Synthesis, spectral, X-ray diffraction, antimicrobial studies and DNA binding properties of binary and ternary complexes of pentadentate N2O3 carbohydrazone ligands. Monatsh Chem 146:15–33. https://doi.org/10.1007/s00706-014-1302-x
Taft RW, Kamlet MJ (1976) The solvatochromic comparison method. 2. The α-scale of solvent hydrogen-bond donor (HBD) acidities. J Am Chem Soc 98:2886–2894. https://doi.org/10.1021/ja00426a036
Wang LH, Liang L, Li PF (2017) Synthesis, crystal structure, catalytic properties, and luminescent of a novel Eu(III) complex material with 4-Imidazolecarboxaldehyde-pyridine-2-carbohydrazone. Bull Chem React Eng Catal 12:185–190. https://doi.org/10.9767/bcrec.12.2.764.185-190
Warkentin J, Ramakrishnan K, Jain RC, Wandelmaier FW (1977) Oxadiazoline derivatives. US424272000
Wiggins P, Williams JAG, Tozer DJ (2009) Excited state surfaces in density functional theory: a new twist on an old problem. J Chem Phys. https://doi.org/10.1063/1.3222641
Yanai T, Tew DP, Handy NC (2004) A new hybrid exchange-correlation functional using the Coulomb-attenuating method (CAM-B3LYP) Chem. Phys Lett 393:51–57. https://doi.org/10.1016/j.cplett.2004.06.011
Zelenin KN, Alekseev VV, Kuznetsova OB, Saminskaya AG, Yakimovich SI, Zverova IV (1999) Carbohydrazones and their ring-chain tautomerism. Russ J Org Chem 35:357–363
Zhang J, Zhang T, Yu K (2006) The preparation, molecular structure and theoretical study of carbohydrazide (CHZ). Struct Chem 17:249–254. https://doi.org/10.1007/s11224-006-9015-3
Acknowledgements
This work was supported by the Ministry of Education, Science and Technological Development of the Republic of Serbia (Project 172013).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mrdjan, G.S., Matijević, B.M., Vastag, G.G. et al. Synthesis, solvent interactions and computational study of monocarbohydrazones. Chem. Pap. 74, 2653–2674 (2020). https://doi.org/10.1007/s11696-020-01106-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-020-01106-4