Abstract
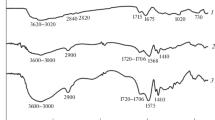
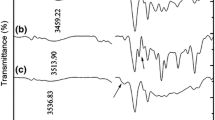
This research was an attempt to investigate the influence of polymerization parameters including monomer concentration, neutralization percent, initiator concentration, the presence of porogen agent and Perlite content on the swelling properties of acrylic-based hydrogels and their composites. With regard to water absorbency, the conclusion drawn was that the highest levels occurred in the following conditions: monomer concentration of 6.127 M, neutralization percentage of 75, initiator concentration of 0.197 M and sodium bicarbonate, as a porogen agent, of 0.375 M. The research was also focused on the absorbency under load (AUL) of the prepared hydrogel composites (acrylic acid/Perlite) at different amounts of Perlite. The composite with Perlite content of 8.33 wt% showed maximum water absorbency under load. The chemical structure of Perlite, hydrogel and their composite was studied using Fourier transform infrared spectroscopy (FTIR). Furthermore, field-emission scanning electron microscopy (FESEM) was used to evaluate the morphology of the prepared hydrogel; while, the energy-dispersive X-ray (EDS) was used to investigate the dispersion of the Perlite in the composite. The FTIR test results confirmed the presence of both components of hydrogel and Perlite in the composite. In addition, the FESEM images and EDS results confirmed induced porous structure after incorporating Perlite particles to the hydrogel, considering an acceptable dispersion.








Similar content being viewed by others
References
Abbadessa A, Landín M, Oude Blenke E, Hennink WE, Vermonden T (2017) Two-component thermosensitive hydrogels: phase separation affecting rheological behavior. Eur Polymer J 92:13–26. https://doi.org/10.1016/j.eurpolymj.2017.04.029
Alkan M, Karadaş M, Doğan M, Demirbaş Ö (2005) Zeta potentials of perlite samples in various electrolyte and surfactant media. Colloids Surf A 259:155–166. https://doi.org/10.1016/j.colsurfa.2005.02.024
Angar N-E, Aliouche D (2017) An enhanced immobilization of BSA biomolecule on anionic hydrogels: swelling and adsorption modeling. Chem Pap 71:1389–1397. https://doi.org/10.1007/s11696-017-0129-4
Cannazza G, Cataldo A, De Benedetto E, Demitri C, Madaghiele M, Sannino A (2014) Experimental assessment of the use of a novel superabsorbent polymer (SAP) for the optimization of water consumption in agricultural irrigation process. Water 6:2056–2069. https://doi.org/10.3390/w6072056
Chaudhri SG, Chaudhari JC, Singh PS (2018) Fabrication of efficient pervaporation desalination membrane by reinforcement of poly(vinyl alcohol)–silica film on porous polysulfone hollow fiber. J Appl Polym Sci 135:45718. https://doi.org/10.1002/app.45718
Chen H, Li J, Shao D, Ren X, Wang X (2012) Poly(acrylic acid) grafted multiwall carbon nanotubes by plasma techniques for Co(II) removal from aqueous solution. Chem Eng J 210:475–481. https://doi.org/10.1016/j.cej.2012.08.082
Choudhary MS (2009) Inverse suspension polymerization of partially neutralized and lightly cross-linked acrylic acid: effect of reaction parameters. Macromol Symp 277:171–176. https://doi.org/10.1002/masy.200950321
Durmaz S, Okay O (2000) Phase separation during the formation of poly(acrylamide) hydrogels. Polymer 41:5729–5735. https://doi.org/10.1016/s0032-3861(99)00809-5
Elliott J, Anseth JW, Bowman NC (2001) Kinetic modeling of the effect of solvent concentration on primary cyclization during polymerization of multifunctional monomers. Chem Eng Sci 56:3173–3184. https://doi.org/10.1016/s0009-2509(00)00547-9
Elliott JE, Macdonald M, Nie J, Bowman CN (2004) Structure and swelling of poly(acrylic acid) hydrogels: effect of pH, ionic strength, and dilution on the crosslinked polymer structure. Polymer 45:1503–1510. https://doi.org/10.1016/j.polymer.2003.12.040
Hussain YA, Liu T, Roberts GW (2012) Synthesis of cross-linked, partially neutralized poly(acrylic acid) by suspension polymerization in supercritical carbon dioxide. Ind Eng Chem Res 51:11401–11408. https://doi.org/10.1021/ie3014007
Kabiri K, Zohuriaan-Mehr MJ (2004) Porous superabsorbent hydrogel composites: synthesis, morphology and swelling rate. Macromol Mater Eng 289:653–661. https://doi.org/10.1002/mame.200400010
Kabiri K, Omidian H, Zohuriaan-Mehr MJ (2003) Novel approach to highly porous superabsorbent hydrogels: synergistic effect of porogens on porosity and swelling rate. Polym Int 52:1158–1164. https://doi.org/10.1002/pi.1218
Karmakar M, Mondal H, Mahapatra M, Chattopadhyay PK, Chatterjee S, Singha NR (2019) Pectin-grafted terpolymer superadsorbent via N-H activated strategic protrusion of monomer for removals of Cd(II), Hg(II), and Pb(II). Carbohydr Polym 206:778–791. https://doi.org/10.1016/j.carbpol.2018.11.032
Kosemund K, Schlatter H, Ochsenhirt JL, Krause EL, Marsman DS, Erasala GN (2009) Safety evaluation of superabsorbent baby diapers. RTP 53:81–89. https://doi.org/10.1016/j.yrtph.2008.10.005
Laftah WA, Hashim S (2014) Synthesis, optimization, characterization, and potential agricultural application of polymer hydrogel composites based on cotton microfiber. Chem Papers 68:798–808. https://doi.org/10.2478/s11696-013-0507-5
Mitra M et al (2018) Carbohydrate and collagen-based doubly-grafted interpenetrating terpolymer hydrogel via N–H activated in situ allocation of monomer for superadsorption of Pb(II), Hg(II), dyes, vitamin-C, and p-nitrophenol. J Hazard Mater 1:369. https://doi.org/10.1016/j.jhazmat.2018.12.019
Mondal H et al (2018) Tetrapolymer network hydrogels via gum ghatti-grafted and N-H/C–H-activated allocation of monomers for composition-dependent superadsorption of metal ions. ACS Omega 3:10692–10708. https://doi.org/10.1021/acsomega.8b01218
Nanjunda Reddy BH, Rauta PR, Venkatalakshimi V, Sreenivasa S (2018) Synthesis and characterization of cloisite-30B clay dispersed poly (acryl amide/sodium alginate)/AgNp hydrogel composites for the study of BSA protein drug delivery and antibacterial activity. Mater Res Exp 5:025403. https://doi.org/10.1088/2053-1591/aaac4c
Omidian H, Zohuriaan J, Bouhendi H (2003) Polymerization of sodium acrylate in inverse-suspension stabilized by sorbitan fatty esters. Eur Polymer J 39:1013–1018. https://doi.org/10.1016/S0014-3057(02)00352-X
Oxley HR, Corkhill PH, Fitton JH, Tighe BJ (1993) Macroporous hydrogels for biomedical applications: methodology and morphology. Biomaterials 14:1064–1072
Patel VR, Amiji MM (1996) Preparation and characterization of freeze-dried chitosan-poly(ethylene oxide) hydrogels for site-specific antibiotic delivery in the stomach. Pharm Res 13:588–593
Sabale V, Vora S (2012) Formulation and evaluation of microemulsion-based hydrogel for topical delivery. Int J Pharmaceut Investig 2:140–149. https://doi.org/10.4103/2230-973X.104397
Sáez-Martínez V, Atorrasagasti G, Olalde B, Madarieta I, Morin F, Garagorri N (2013) Fabrication and characterization of macroporous poly(ethylene glycol) hydrogels generated by several types of porogens. Int J Polym Mater 62:502–508. https://doi.org/10.1080/00914037.2012.734353
Saifuddin NM, Kumaran P (2005) Removal of heavy metal from industrial wastewater using chitosan coated oil palm shell charcoal. Electron J Biotechnol 8:43–53. https://doi.org/10.4067/S0717-34582005000100008
Samar M, Saxena S (2016) Study of chemical and physical properties of perlite and its application in India. Int J Sci Technol Manag 5:70–80
Sevgili LM, Toprak S, Çavuş S (2015) Swelling of N-vinylcaprolactam-dodecyl methacrylate gel in heptane + toluene mixtures. Chem Papers 69:668–678. https://doi.org/10.1515/chempap-2015-0073
Shen J, Yan B, Li T, Long Y, Li N, Ye M (2012) Mechanical, thermal and swelling properties of poly(acrylic acid)–graphene oxide composite hydrogels. Soft Matter 8:1831–1836. https://doi.org/10.1039/c1sm06970e
Singha N, Mahapatra M, Karmakar M, Dutta A, Mondal H, Chattopadhyay P (2017a) Synthesis of sustainable Guar gum-g-(acrylic acid-co-acrylamide-co-3-acrylamido propanoic acid) interpenetrating polymer network via in situ attachment of 3-acrylamido propanoic acid for analyzing superadsorption mechanism of Pb(II)/Cd(II)/Cu(II) and dyes: comparative studies of microstructures. Polym Chem. https://doi.org/10.1039/c7py01564j
Singha NR, Karmakar M, Mahapatra M, Mondal H, Dutta A, Roy C, Chattopadhyay PK (2017b) Systematic synthesis of pectin-g-(sodium acrylate-co-N-isopropylacrylamide) interpenetrating polymer network for superadsorption of dyes/M(ii): determination of physicochemical changes in loaded hydrogels. Polym Chem 8:3211–3237. https://doi.org/10.1039/c7py00316a
Singha NR, Dutta A, Mahapatra M, Karmakar M, Mondal H, Chattopadhyay PK, Maiti DK (2018a) Guar gum-grafted terpolymer hydrogels for ligand-selective individual and synergistic adsorption: effect of comonomer composition. ACS Omega 3:472–494. https://doi.org/10.1021/acsomega.7b01682
Singha NR et al (2018b) An in situ approach for the synthesis of a gum ghatti-g-interpenetrating terpolymer network hydrogel for the high-performance adsorption mechanism evaluation of Cd(ii), Pb(ii), Bi(iii) and Sb(iii). J Mater Chem A 6:8078–8100. https://doi.org/10.1039/c8ta01106k
Singha NR et al (2018c) Situ allocation of a monomer in pectin-g-terpolymer hydrogels and effect of comonomer compositions on superadsorption of metal ions/dyes. ACS Omega 3:4163–4180. https://doi.org/10.1021/acsomega.8b00446
Singhal R, Singh Tomar R, Nagpal KA (2009) Effect of cross-linker and initiator concentration on the swelling behaviour and network parameters of superabsorbent hydrogels based on acrylamide and acrylic acid. Int J Plast Technol 13:22–37. https://doi.org/10.1007/s12588-009-0004-4
Sunitha S, Jitendra W, Sujatha D, Santhosh Kumar M (2013) Design and evaluation of hydrogel-thickened microemulsion for topical delivery of Minoxidil Iranian. J Pharm Sci 9:1–14
Vorsina I, Grigorieva T, Barinova A, Lyakhov N (2011) Mechanochemical interaction of silicon dioxide with organic acids. Chem Sustain Dev 19:447–455
Wang G, Li M, Chen X (1997) Inverse suspension polymerization of sodium acrylate. J Appl Polym Sci 65:789–794. https://doi.org/10.1002/(SICI)1097-4628(19970725)65:4%3c789:AID-APP16%3e3.0.CO;2-O
Wittenberg NFG, Preusser C, Kattner H, Stach M, Lacík I, Hutchinson RA, Buback M (2016) Modeling acrylic acid radical polymerization in aqueous solution. Macromol React Eng 10:95–107. https://doi.org/10.1002/mren.201500017
Wu G, Li S, He W, Xiao X (2018) Effect of percentage of graphene oxide on phase transition of water induced shape memory behavior of PVA-go-PEG hydrogel. Mater Res Exp 5:115703. https://doi.org/10.1088/2053-1591/aadd71
Yarimkaya S, Basan H (2007) Synthesis and swelling behavior of acrylate-based hydrogels. J Macromol Sci Part A 44:699–706. https://doi.org/10.1080/10601320701351268
Ye L, Liu QL, Gen Zhang Q, Zhu A, Bo Zhou G (2007) Pervaporation characteristics and structure of poly(vinyl alcohol)/poly(ethylene glycol)/tetraethoxysilane hybrid membranes. J Appl Polym Sci 105:3640–3648. https://doi.org/10.1002/app.26446
Yu J et al (2017) Superabsorbent polymer properties and concentration effects on water retention under drying conditions. Soil Sci Soc Am J 81:889–901. https://doi.org/10.2136/sssaj2016.07.0231
Zavgorodnya O, Kozlovskaya V, Liang X, Kothalawala N, Catledge SA, Dass A, Kharlampieva E (2014) Temperature-responsive properties of poly(N-vinylcaprolactam) multilayer hydrogels in the presence of Hofmeister anions. Mater Res Exp 1:035039. https://doi.org/10.1088/2053-1591/1/3/035039
Zohourian Mehr MJAD, Kabiri K (2008) Superabsorbent polymer materials: a review. Iran Polym J 17:447–451
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Safaei, F., Khalili, S., Khorasani, S.N. et al. Preparation of an acrylic acid-based superabsorbent composite: investigation of synthesis parameters. Chem. Pap. 74, 939–949 (2020). https://doi.org/10.1007/s11696-019-00927-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-019-00927-2