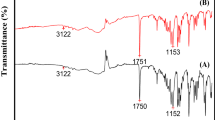
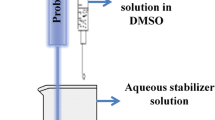
Abstract
This study investigates the effect of process parameters [like solvent to anti-solvent ratio (1:2–1:4), injection rate (1–10 mL/min), drug concentration (5–15 mg/mL), crystallization temperature (20–30 °C), sonication amplitude (20–60%), and sonication time (2–15 min)] on drug particle size and morphology via anti-solvent sonocrystallization using the design of experiment approach. A six-factor, three-level experimental design using Taguchi’s L18 orthogonal array has been employed to obtain nano-range particle size of recrystallized drug at lab scale batch study. The particle size obtained using optimum conditions obtained by Taguchi method was further improved by Box–Behnken design. The optimum conditions obtained by Box–Behnken design were 1:3 solvent to anti-solvent ratio, 9.87 mL/min injection rate, 11.09 mg/mL drug concentration, 20 °C crystallization temperature, 8.6 min of sonication time, and 40% of sonication amplitude, which provided the optimized particle size of 143.6 nm. Various analytical tools were employed to characterize the sample obtained at the optimum condition. This statistical approach can further be explored to obtain nano-range particle size of other biopharmaceutical classification system class II drugs having poor water solubility via anti-solvent sonocrystallization.






Similar content being viewed by others
Abbreviations
- A:
-
Solvent to anti-solvent ratio (–)
- B:
-
Injection rate (mL/min)
- C:
-
Drug concentration (mg/mL)
- D:
-
Crystallization temperature (°C)
- D:
-
Degree of freedom
- E:
-
Sonication amplitude (%)
- F:
-
Sonication time (min)
- R2 :
-
Coefficient of determination
- R 2adj :
-
Adjustment for the coefficient of determination
References
Abuzar SM, Hyun SM, Kim J-H, Park HJ, Kim M-S, Park J-S, Hwang S-J (2018) Enhancing the solubility and bioavailability of poorly water-soluble drugs using supercritical antisolvent (SAS) process. Int J Pharm 538:1–13. https://doi.org/10.1016/j.ijpharm.2017.12.041
Bajaj A, Rao MRP, Pardeshi A, Sali D (2012) Nanocrystallization by evaporation antisolvent technique for solubility and bioavailability enhancement of telmisartan. AAPS J 13:1331–1340. https://doi.org/10.1208/s12249-012-9860-x
Beck C, Dalvi SV, Dave RN (2010) Controlled liquid antisolvent precipitation using a rapid mixing device. Chem Eng Sci 65:5669–5675. https://doi.org/10.1016/j.ces.2010.04.001
Delgado A, Matijevic E (1991) Particle-size distribution of inorganic collidal dispersion: a comparision of different techniques. Part Part Syst Charact 8:128–135
Desai MA, Parikh J, De AK (2014) Modelling and optimization studies on extraction of lemingrass oil from cymbopogon flexuosus (Sted.) Wats. Chem Eng Res Des 92:793–803. https://doi.org/10.1016/j.cherd.2013.08.011
Duffy D, Barrett M, Glennon B (2013) Novel, calibration-free strategies for supersaturation in antisolvent crystallization process. Cryst Growth Des 13:3321–3332. https://doi.org/10.1021/cg301673g
Gonda K, Kadota K, Deki Y, Tozuka Y, Shimosaka A, Shirakawa Y, Hidaka J (2015) Fabrication of composite particles by liquid–liquid interfacial crystallization using an ultrasonic spray nozzle. Powder Technol 269:401–408. https://doi.org/10.1016/j.powtec.2014.09.042
Hu J, Johnston KP, Williams RO III (2004) Nanoparticle engineering process for enhancing the dissolution rates of poorly water soluble drugs. Drug Dev Ind Pharm 30:233–245. https://doi.org/10.1081/DDC-120030422
Isaac J, Ganguly S, Ghosh A (2016) Co-milling of telmisartan with poly(vinyl alcohol)—an alkalinizer free green approach to ensure its bioavailability. Eur J Pharm Biopharm 101:43–52. https://doi.org/10.1016/j.ejpb.2016.01.016
Kakran M, Sahoo NG, Judeh Z (2012) Fabrication of quercetin nanoparticles by anti-solvent precipitation method for enhacced dissolution. Powder Technol 223:59–64. https://doi.org/10.1016/j.powtec.2011.08.021
Kakran M, Sahoo NG, Li L, Judeh Z (2013) Particle size reduction of poorly water soluble artemisinin via antisolvent precipitation with a syringe pump. Powder Technol 237:468–476. https://doi.org/10.1016/j.powtec.2012.12.029
Keck CM, Muller RH (2006) Drug nanocrystals of poorly soluble drugs produced by high pressure homogenization. Eur J Pharm Biopharm 62:3–16. https://doi.org/10.1016/j.ejpb.2005.05.009
Krishnaiah K, Shahabudeen P (2013) Applied design of experiment and Taguchi methods, 2nd edn. PHI Learning, Delhi
Liu W, Guo Y, Chen J, Yu X (2016) Measurement and correlation of the solubility of Telmisartan (Form A) in nine different solvents from 277.85 to 338.35 K. J Solut Chem 45:932–946. https://doi.org/10.1007/s10953-016-0484-8
Lonare AA, Patel SR (2014) Antisolvent crystallization of poorly water soluble drugs. Int J Chem Eng Appl 4:337–341. https://doi.org/10.7763/IJCEA.2013.V4.321
Luque de Castro MD, Priego-Capote F (2007) Ultrasound-assisted crystallization (sonocrystallization). Ultra Sonichem 14:717–724. https://doi.org/10.1016/j.ultsonch.2006.12.004
Masoumi HRF, Basri M, Samium WS, Izadiyan Z, Lim CJ (2015) Enhancement of encapsulation efficiency of nanoemulsion-containing aripiprazole for the treatment of schizophrenia using mixture experimental design. Int J Nanomed 10:6469–6476. https://doi.org/10.2147/IJN.S89364
Matteucci ME, Hotze MA, Johnston KP, Williams RO III (2006) Drug nanoparticles by antisolvent precipitation: mixing energy versus surfactant stabilization. Langmuir 22:8951–8959
Montgomery DC (2013) Design and analysis of experiments, 8th edn. Wiley, Hoboken
Mullin JW (2001) Crystallization, 4th edn. Butterworth-Heinemann, Oxford
Park MW, Yeo SD (2010) Antisolvent crystallization of Roxithromycin and the effect of ultrasound. Sep Sci Technol 45:1402–1410. https://doi.org/10.1080/01496391003689538
Park J, Cho W, Cha K-H, Ahm J, Han K, Hwang S-J (2013) Solubilization of the poorly water soluble drug, telmisartan, using supercritical anti-solvent (SAS) process. Int J Pharm 441:50–55. https://doi.org/10.1016/j.ijpharm.2012.12.020
Rahman N, Sameen S, Kashif M (2019) Application of Box–Behnken design and desirability function in the optmization of spectrophotometric method for the quantification of WADA banned drug: acetazolamide. J Mol Liq 274:270–277. https://doi.org/10.1016/j.molliq.2018.10.120
Rajalahti T, Kvalheim OM (2011) Multivariate data analysis in pharmaceutics: a tutorial review. Int J Pharm 417:280–290. https://doi.org/10.1016/j.ijpharm.2011.02.019
Ross PJ (1989) Taguchi techniques for quality engineering. McGraw-Hill, New York
Sharma C, Desai MA, Patel SR (2018) Ultrasound-assisted anti-solvent crystallization of telmisartan using dimethyl sulfoxide as organic solvent. Cryst Res Technol 53:1–9. https://doi.org/10.1002/crat.201800001
Sharma C, Desai MA, Patel SR (2019) Effect of surfactants and polymers on morphology and particle size of telmisartan in ultrasound-assisted anti-solvent crystallization. Chem Pap 73:1685–1694. https://doi.org/10.1007/s11696-019-00720-1
Solaiman A, Tatari AK, Elkordy AA (2016) Naproxen microparticulate systems prepared using in situ crystallisation and freeze-drying techniques. AAPS PharmSciTech. https://doi.org/10.1208/s12249-016-0682-0
Suryanarayanan R, Mitchell AG (1985) Evaluation of two concenpts of crystallinity using calcium gluceptate as a model compound. Int J Pharm 24:1–17
Thakker MR, Parikh JK, Desai MA (2018) Synergism between ionic liquid and ultrasound for greener extraction of geraniol: optimization using different statistical tools, comparison and prediction. Chem Eng Res Des 134:162–171. https://doi.org/10.1016/j.cherd.2018.04.003
Zhang Y, Zhi Z, Jiang T, Zhang J, Wang Z, Wang S (2010) Spherical mesoporous silica nanoparticles for loading and release of poorly water-soluble drug telmisartan. J Control Release 145:257–263. https://doi.org/10.1016/j.jconrel.2010.04.029
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sharma, C., Desai, M.A. & Patel, S.R. Anti-solvent sonocrystallization for nano-range particle size of telmisartan through Taguchi and Box–Behnken design. Chem. Pap. 74, 323–331 (2020). https://doi.org/10.1007/s11696-019-00886-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-019-00886-8