Abstract
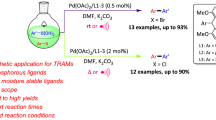
In this study, new six Pd-based complexes containing mixture N-heterocyclic carbene (NHC) and triphenylphosphine (PPh3) ligands were synthesized from the reaction of the (NHC)PdI2(pyridine) with PPh3. The new (NHC)PdI2(PPh3) complexes were characterized using FTIR, 1H-NMR, 13C-NMR, and 31P-NMR spectroscopy and elemental analyses techniques. These spectra are consistent with the proposed formula. Molecular and crystal structure of two complexes was obtained using single-crystal X-ray diffraction method. Based on the crystal results, a cis geometry was assigned to all the complexes. These new complexes have been examined as the catalyst in the Sonogashira cross-coupling reaction. The catalytic conversions of the complexes were obtained between 71 and 99% from the reaction of phenylacetylene and aryl bromides. Also, these new complexes have been examined as the catalyst in the Mizoroki–Heck cross-coupling reaction. The catalytic conversions of the complexes were obtained between 80 and 100% from the reaction of styrene and aryl bromides.





Similar content being viewed by others
References
Aktaş A, Gök Y (2014) 4-Vinylbenzyl-substituted silver(I) N-heterocyclic carbene complexes and ruthenium(II) N-heterocyclic carbene complexes: synthesis and transfer hydrogenation of ketones. Transit Met Chem 39:925–931. https://doi.org/10.1007/s11243-014-9877-y
Aktaş A, Gök Y (2015) N-Propylphthalimide-substituted silver(I) N-heterocyclic carbene complexes and ruthenium(II) N-heterocyclic carbene complexes: synthesis and transfer hydrogenation of ketones. Catal Lett 145:631–639. https://doi.org/10.1007/s10562-015-1626-0
Aktaş A, Gök Y, Akkoç S (2013) Palladium catalyzed Mizoroki–Heck and Suzuki–Miyaura reactions using naphthalenomethyl-substituted imidazolidin-2-ylidene ligands in aqueous media. J Coord Chem 66:2901–2909. https://doi.org/10.1007/s11164-017-2995-3
Aktaş A, Barut Celepci D, Gök Y, Aygün M (2018a) 2-Hydroxyethyl-substituted (NHC)Pd(II)PPh3 complexes: synthesis, characterization, crystal structure and its application on Sonogashira Cross-coupling reactions in aqueous media. ChemistrySelect 3:10932–10937. https://doi.org/10.1002/slct.201802519
Aktaş A, Barut Celepci D, Gök Y, Aygün M (2018b) 2-Hydroxyethyl-substituted Pd-PEPPSI complexes: synthesis, characterization and the catalytic activity in the Suzuki–Miyaura reaction for aryl chlorides in aqueous media. ChemistrySelect 3:9974–9980. https://doi.org/10.1002/slct.201802046
Aktaş A, Erdemir F, Barut Celepci D, Gök Y, Aygün M (2019) Mixed phosphine/N-heterocyclic carbene–palladium complexes: synthesis, characterization, crystal structure and application in the Sonogashira reaction in aqueous media. Transit Met Chem 44:229–236. https://doi.org/10.1007/s11243-018-0286-5
Alonso DA, Najera C, Pacheco MC (2003) C(sp2)–C(sp) and C(sp)–C(sp) coupling reactions catalyzed by oxime-derived palladacycles. Adv Synth Catal 345:1146–1158. https://doi.org/10.1002/adsc.200303067
Bagley MC, Dale JW, Merritt EA, Xiong X (2005) Thiopeptide antibiotics. Chem Rev 105:685–714. https://doi.org/10.1021/cr0300441
Beletskaya IP, Cheprakov AV (2000) The Heck reaction as a sharpening stone of palladium catalysis. Chem Rev 100:3009–3066. https://doi.org/10.1021/cr9903048
Beller M, Bolm C (1998) Transition metals for organic synthesis. 2nd ed. vol 1. Wiley-VCH, Weinheim
Boubakri L, Yasar S, Dorcet V, Roisnel T, Bruneau C, Hamdi N, Ozdemir İ (2017) Synthesis and catalytic applications of palladium N-heterocyclic carbene complexes as efficient pre-catalysts for Suzuki–Miyaura and Sonogashira coupling reactions. New J Chem 41:5105–5113. https://doi.org/10.1039/C7NJ00488E
Chan K-T, Tsai Y-H, Lin W-S, Wu J-R, Chen S-J, Liao F-X, Hu C-H, Lee HM (2010) Palladium complexes with carbene and phosphine ligands: synthesis, structural characterization, and direct arylation reactions between aryl halides and alkynes. Organometallics 29:463–472. https://doi.org/10.1021/om9009203
Clark RC, Reid JS (1995) The analytical calculation of absorption in multifaceted crystals. Acta Crystallogr A 51:887–897. https://doi.org/10.1107/S0108767395007367
Cornils B, Herrmann WA (1996) Applied homogeneous catalysis with organometallic compounds. Wiley-VCH, Weinheim
Cosford ND, Tehrani L, Roppe J, Schweiger E, Smith ND, Anderson J, Bristow L, Brodkin J, Jiang X, McDonald I, Rao S, Washburn M, Varney MA (2003) 3-[(2-Methyl-1, 3-thiazol-4-yl) ethynyl]-pyridine: a potent and highly selective metabotropic glutamate subtype 5 receptor antagonist with anxiolytic activity. J Med Chem 46:204–206. https://doi.org/10.1021/jm025570j
CrysAlisPro Software System (2015) Version 1.171.38.43 Rigaku Corporation. Oxford UK
Dehimat ZI, Yaşar S, Tebbani D, Özdemir İ (2018) Sonogashira cross-coupling reaction catalyzed by N-heterocyclic carbene–Pd(II)–PPh3 complexes under copper free and aerobic conditions. Inorg Chim Acta 469:325–334. https://doi.org/10.1016/j.ica.2017.09.048
Diebolt O, Jurčík V, Correa da Costa R, Braunstein P, Cavallo L, Nolan SP, Slawin AMZ, Cazin CSJ (2010) Mixed phosphite/N-heterocyclic carbene complexes: synthesis, characterization and catalytic studies. Organometallics 29:1443–1450. https://doi.org/10.1021/om9011196
Dieck HA, Heck RF (1974) Organophosphinepalladium complexes as catalysts for vinylic hydrogen substitution reactions. J Am Chem Soc 96:1133–1136. https://pubs.acs.org/doi.org/10.1021/ja00811a029
Diez-Gonzalez S, Marion N, Nolan SP (2009) N-Heterocyclic carbenes in late transition metal catalysis. Chem Rev 109:3612–3676. https://doi.org/10.1021/cr900074m
Dolomanov OV, Bourhis LJ, Gildea RJ, Howard JAK, Puschmann H (2009) OLEX2: a complete structure solution, refinement and analysis program. J Appl Cryst 42:339–341. https://doi.org/10.1107/S0021889808042726
Dounay AB, Overman LE (2003) The asymmetric intramolecular Heck reaction in natural product total synthesis. Chem Rev 103:2945–2964. https://doi.org/10.1021/cr020039h
Erdemir F, Barut Celepci D, Aktaş A, Gök Y (2019) 2-hydroxyethyl-substituted (NHC)PdI2(pyridine) (Pd-PEPPSI) complexes: synthesis, characterization and the catalytic activity in the Sonogashira cross-coupling reaction. ChemistrySelect 4:5585–5590. https://doi.org/10.1002/slct.201901033
Erdoğan H, Aktaş A, Gök Y, Sarı Y (2018) N-Propylphthalimide-substituted bis-(NHC) PdX2 complexes: synthesis, characterization and catalytic activity in direct arylation reactions. Transit Met Chem 43:31–37. https://doi.org/10.1007/s11243-017-0190-4
Etter MC, MacDonald JC (1990) Graph-set analysis of hydrogen-bond patterns in organic crystals. J. Bernstein Acta Crystallogr B 46:256–262. https://doi.org/10.1107/S0108768189012929
Glorius F (2006) N-Heterocyclic carbenes in catalysis—an introduction. Top Organomet Chem 21:1–20. https://doi.org/10.1007/3418_2006_059
Gök Y, Aktaş A, Erdoğan H, Sarı Y (2018) New 4-vinylbenzyl-substituted bis (NHC)-Pd (II) complexes: synthesis, characterization and the catalytic activity in the direct arylation reaction. Inorg Chim Acta 471:735–740. https://doi.org/10.1016/j.ica.2017.12.019
Gök Y, Aktaş A, Sarı Y, Erdoğan H (2019) 2-Methyl-1,4-benzodioxan-substituted bis(NHC)PdX2 complexes: synthesis, characterization and the catalytic activity in the direct arylation reaction of some 2-alkyl-heterocyclic compounds. J Iran Chem Soc 16:423–433. https://doi.org/10.1007/s13738-018-1512-y
Hahn FE, Lügger T, Beinhoff M (2004) Palladium(II) complexes with benzoxazol-2-ylidene ligands: crystal structures of trans-chloro(benzoxazol-2-ylidene)-bis(triphenyl phosphine)palladium(II) chloride and cis-diiodo(benzoxazol-2-ylidene)(triphenylphosphine) palladium(II). Z Naturforsch 59b:196–201. https://doi.org/10.1515/znb-2004-0212
Heck RF (1978) New applications of palladium in organic syntheses. Pure Appl Chem 50:691–701. https://doi.org/10.1351/pac197850080691
Heck RF (1982) Palladium-catalyzed vinylation of organic halides. Org React 27:345–390. https://doi.org/10.1002/0471264180.or027.02
Heck RF (1979) Palladium-catalyzed reactions of organic halides with olefins. Acc Chem Res 12:146–151. https://pubs.acs.org/doi/10.1021/ar50136a006
Heck RF, Nolley JP (1972) Palladium-catalyzed vinylic hydrogen substitution reactions with aryl, benzyl, and styryl halides. J Org Chem 37:2320–2322. https://pubs.acs.org/doi/10.1021/jo00979a024
Herrmann WA, Böhm VPW, Gstöttmayr GWK, Grosche M, Reisinger C-P, Weskamp T (2001) Synthesis, structure and catalytic application of palladium (II) complexes bearing N-heterocyclic carbenes and phosphines. J Organomet Chem 617–618:616–628. https://doi.org/10.1016/S0022-328X(00)00722-1
Herrmann WA, Öfele K, von Preysing D, Schneider SK (2003) Phospha-palladacycles and N-heterocyclic carbene palladium complexes: efficient catalysts for C–C-coupling reactions. J Organomet Chem 687:229–248. https://doi.org/10.1016/j.jorganchem.2003.07.028
Kondolff I, Doucet H, Santelli M (2003) Tetraphosphine/palladium-catalyzed Heck reactions of aryl halides with disubstituted alkenes. Tetrahedron Lett 44:8487–8491. https://doi.org/10.1016/j.tetlet.2003.09.092
Li W, Ivanov S, Mozaffari S, Shanaiah N, Karim AM (2019) Palladium acetate trimer: understanding its ligand-induced dissociation thermochemistry using isothermal titration calorimetry, X-ray absorption fine structure, and 31P nuclear magnetic resonance. Organometallics 38:451–460. https://doi.org/10.1021/acs.organomet.8b00787
Liao C-Y, Chan K-T, Tu C-Y, Chang Y-W, Hu C-H, Lee HM (2009) Robust and electron-rich cis-palladium (II) complexes with phosphine and carbene ligands as catalytic precursors in Suzuki coupling reactions. Chem Eur J 15:405–417. https://doi.org/10.1002/chem.200801296
Menezes da Silva VH, Braga AAC, Cundari TR (2016) N-Heterocyclic carbene based nickel and palladium complexes: a DFT comparison of the Mizoroki–Heck catalytic cycles. Organometallics 35:3170–3181. https://doi.org/10.1021/acs.organomet.6b00532
Mizoroki T, Mori K, Ozaki A (1971) Arylation of olefin with aryl iodide catalyzed by palladium. Bull Chem Soc Jpn 44:581–581. https://doi.org/10.1246/bcsj.44.581
Modak S, Gangwar MK, Rao MN, Madasu M, Kalita AC, Dorcet V, Shejale MA, Butcher RJ, Ghosh P (2015) Fluoride-free Hiyama coupling by palladium abnormal N-heterocyclic carbene complexes. Dalton Trans 44:17617–17628. https://doi.org/10.1039/C5DT02317C
Mori K, Mizoroki T, Ozaki A (1973) Arylation of olefin with iodobenzene catalyzed by palladium. Bull Soc Chem Jpn 46:1505–1508. https://doi.org/10.1246/bcsj.46.1505
Nishihara Y, Ikegashira K, Hirabayashi K, Ando J-I, Mori A, Hiyama T (2000) Coupling reactions of alkynylsilanes mediated by a Cu(I) salt: novel syntheses of conjugate diynes and disubstituted ethynes. J Organomet Chem 65:1780–1787. https://doi.org/10.1021/jo991686k
Oestreich M (2009) The Mizoroki–Heck reaction, vol 1. Wiley, Chichester
Sarı Y, Aktaş A, Barut Celepci D, Gök Y, Aygün M (2017) Synthesis, characterization and crystal structure of new 2-morpholinoethyl-substituted bis-(NHC)Pd(II) complexes and the catalytic activity in the direct arylation reaction. Catal Lett 147:2340–2351. https://doi.org/10.1007/s10562-017-2132-3
Schmid TE, Jones DC, Songis O, Diebolt O, Furst MRL, Slawin AMZ, Cazin CSJ (2013) Mixed phosphine/N-heterocyclic carbene palladium complexes: synthesis, characterization and catalytic use in aqueous Suzuki–Miyaura reactions. Dalton Trans 42:7345–7353
Sheldrick GM (2015a) SHELXT-integrated space-group and crystal-structure determination. Acta Crystallog Sect A 71:3–8. https://doi.org/10.1039/C2DT32858E
Sheldrick GM (2015b) Crystal structure refinement with SHELXL. Acta Crystallog Sect C 71:3–8. https://doi.org/10.1107/S2053229614024218
Siemeling C, Färber U, Bruhn C, Fürmeier S, Schulz T, Kurlemann M, Tripp S (2012) Group 10 metal complexes of a ferrocene-based N-heterocyclic carbene: syntheses, structures and catalytic applications. Eur J Inorg Chem 2012:1413–1422. https://doi.org/10.1002/ejic.201100856
Sonogashira K (2002) Development of Pd–Cu catalyzed cross-coupling of terminal acetylenes with sp2-carbon halides. J Organomet Chem 653:46–49. https://doi.org/10.1016/S0022-328X(02)01158-0
Touj N, Yaşar S, Özdemir N, Hamdi N, Özdemir İ (2018) Sonogashira cross-coupling reaction catalysed by mixed NHC-Pd-PPh3 complexes under copper free conditions. J Organomet Chem 860:59–71. https://doi.org/10.1016/j.jorganchem.2018.01.017
Watson DA (2016) Legacy of Richard Heck. Organometallics 35:1177–1178. https://pubs.acs.org/doi/10.1021/acs.organomet.6b00261
Whitcombe NJ, Hii KK, Gibson SE (2001) Advances in the Heck chemistry of aryl bromides and chlorides. Tetrahedron 57:7449–7476. https://doi.org/10.1016/S0040-4020(01)00665-2
Zamora MT, Ferguson MJ, McDonald R, Cowie M (2012) Unsymmetrical dicarbenes based on N-heterocyclic/mesoionic carbene frameworks: a stepwise metalation strategy for the generation of a dicarbene-bridged mixed-metal Pd/Rh complex. Organometallics 31:5463–5477. https://doi.org/10.1021/om3004543
Acknowledgements
The authors acknowledge Dokuz Eylul University for the use of the Rigaku Oxford Xcalibur Eos Diffractometer (purchased under University Research Grant no.: 2010.KB.FEN.13). The authors thank the Inonu University Faculty of Science Department of Chemistry for the spectroscopy and elemental analysis characterization of compounds.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interests
The authors declare that there are no conflicts of interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix A: Supplementary data
Appendix A: Supplementary data
CCDC 1897240 for 1b and 1897153 for 1f contain the supplementary crystallographic data. Copies of the data can be obtained free of charge at http://www.ccdc.cam.ac.uk/conts/retrieving.html or from the Cambridge Crystallographic Data Center, 12, Union Road, Cambridge CB2 1EZ, UK. fax: (+44) 1223-336-033, e-mail: deposit@ccdc.cam.ac.uk.
Rights and permissions
About this article
Cite this article
Erdemir, F., Aktaş, A., Barut Celepci, D. et al. New (NHC)Pd(II)(PPh3) complexes: synthesis, characterization, crystal structure and its application on Sonogashira and Mizoroki–Heck cross-coupling reactions. Chem. Pap. 74, 99–112 (2020). https://doi.org/10.1007/s11696-019-00859-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-019-00859-x