Abstract
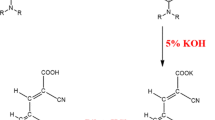
In this study, photovoltaic cell devices based on phthalocyanine structures with metal-free and zinc metal-containing D–π–A substitutes consisting of biphenyl and cyano-containing substituents have been synthesized. The non-symmetrical phthalocyanine photo-sensitizer has three biphenyls and one cyano group, respectively, which act as electron release and withdrawal. The new light-sensitive dye cells were tested in dye-sensitive solar cells and the performance was compared to N719 dye. All compounds synthesized were characterized using FTIR, 1 H NMR, C NMR, UV–Vis and MS spectroscopic data. First, the relationship between symmetric and non-symmetric ZnPc and H2Pc morphology and photovoltaic properties was discussed. The effects of symmetric and asymmetric cyano groups on solar cell efficiency were investigated. Furthermore, according to the results from the non-symmetrical ZnPc device prepared at room temperature, it was determined that it exhibited relatively higher photovoltaic cell efficiency due to significant improvements in short-circuit photo-current (Jsc) and open-circuit voltage (Voc).




Similar content being viewed by others
References
Ali HEA, Altındal A, Altun S, Odabaş Z (2016) Highly efficient dye-sensitized solar cells based on metal-free and copper (II) phthalocyanine bering 2-phenylphenoxy moiety. Dyes Pigments 124:180–187
Erdoğmuş A, Nyokong T (2009) New soluble methylendioxy-phenoxy-substituted zinc phthalocyanine derivatives: synthesis, photo physical and photochemical studies. Polyhedron 28(14):2855–2862
Grätzel M (2003) Dye-sensitized solar cells. J Photochem Photobiol C 4(2):145–153
Hu L, Dai S, Weng J, Xiao S, Sui Y, Huang Y, Wang K (2007) Microstructure design of nanoporous TiO2 photoelectrodes for dye-sensitized solar cell modules. J Phys Chem B 111(2):358–362
Jono R, Fujisawa JI, Segawa H, Yamashita K (2011) Theoretical study of the surface complex between TiO2 and TCNQ showing interfacial charge-transfer transitions. J Phys Chem Lett 2(10):1167–1170
Kalkan A, Bayır ZA (2006) Phthalocyanines with rigid carboxylic acid containing pendant arms. Polyhedron 25(1):39–42
Kamat PV (1993) Photochemistry on nonreactive and reactive (semiconductor) surfaces. Chem Rev 93(1):267–300
Kanaparthi RK, Kandhadi J, Giribabu L (2012) Metal-free organic dyes for dye-sensitized solar cells: recent advances. Tetrahedron 68(40):8383–8393
Kao PC, Chu SY, Liu SJ, You ZX, Chuang CA (2006) Improved performance of organic light-emitting diodes using a metal-phthalocyanine hole-injection layer. J Electrochem Soc 153(6):H122–H126
Kay A, Graetzel M (1993) Artificial photosynthesis. 1. Photosensitization of titania solar cells with chlorophyll derivatives and related natural porphyrins. J Phys Chem 97(23):6272–6277
Kim BH, Freeman HS (2013) New N-methyl pyrrole and thiophene based D–π–A systems for dye-sensitized solar cells. Dyes Pigments 96(2):313–318
Kim S, Kim D, Choi H, Kang MS, Song K, Kang SO, Ko J (2008) Enhanced photovoltaic performance and long-term stability of quasi-solid-state dye-sensitized solar cells via molecular engineering. Chem Commun 40:4951–4953
Mao J, He N, Ning Z, Zhang Q, Guo F, Chen L, Tian H (2012) Stable dyes containing double acceptors without COOH as Anchors for highly efficient dye-sensitized solar cells. Angew Chem Int Ed 51(39):9873–9876
Mao J, Wang D, Liu SH, Hang Y, Xu Y, Zhang Q, Hua J (2014) Dye-sensitized solar cells based on functionally separated D–π–A dyes with 2-cyanopyridine as an electron-accepting and anchoring group. Asian J Org Chem 3(2):153–160
Michinobu T, Satoh N, Cai J, Li Y, Han L (2014) Novel design of organic donor–acceptor dyes without carboxylic acid anchoring groups for dye-sensitized solar cells. J Mater Chem C 2(17):3367–3372
Nazeeruddin MK, Splivallo R, Liska P, Comte P, Grätzel M (2003) A swift dye uptake procedure for dye sensitized solar cells. Chem Commun 12:1456–1457
O’regan B, Grätzel M (1991) A low-cost, high-efficiency solar cell based on dye-sensitized colloidal TiO2 films. Nature 353(6346):737
Smestad GP (1998) Education and solar conversion: demonstrating electron transfer. Sol Energy Mater Sol Cells 55(1–2):157–178
Song JL, Amaladass P, Wen SH, Pasunooti KK, Li A, Yu YL, Liu XW (2011) Aryl/hetero-arylethyne bridged dyes: the effect of planar π-bridge on the performance of dye-sensitized solar cells. New J Chem 35(1):127–136
Wang L, Liang M, Zhang Y, Cheng F, Wang X, Sun Z, Xue S (2014) Influence of donor and bridge structure in D–A–π–A indoline dyes on the photovoltaic properties of dye-sensitized solar cells employing iodine/cobalt electrolyte. Dyes Pigments 101:270–279
Wu Y, Zhu W (2013) Organic sensitizers from D–π–A to D–A–π–A: effect of the internal electron-withdrawing units on molecular absorption, energy levels and photovoltaic performances. Chem Soc Rev 42(5):2039–2058
Wu G, Kong F, Li J, Chen W, Fang X, Zhang C, Dai S (2013) Influence of different acceptor groups in julolidine-based organic dye-sensitized solar cells. Dyes Pigments 99(3):653–660
Yanagisawa M, Korodi F, Bergquist J, Holmberg A, Hagfeldt A, Åkermark B, Sun L (2004) Synthesis of phthalocyanines with two carboxylic acid groups and their utilization in solar cells based on nano-structured TiO 2. J Porphyr Phthalocyanines 8(10):1228–1235
Zhang L, Cole JM (2015) Anchoring groups for dye-sensitized solar cells. ACS Appl Mater Interfaces 7(6):3427–3455
Zhang J, Kan YH, Li HB, Geng Y, Wu Y, Su ZM (2012) How to design proper π–spacer order of the D–π–A dyes for DSSCs? A density functional response. Dyes Pigments 95(2):313–321
Zhou R, Josse F, Göpel W, Öztürk ZZ, Bekaroğlu Ö (1996) Phthalocyanines as sensitive materials for chemical sensors. Appl Organomet Chem 10(8):557–577
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Karadoğan, B., Kılıçarslan, F.A. & Erden, I. Synthesis of phthalocyanines with donor–acceptor properties and their applications to dye-sensitized solar cell. Chem. Pap. 73, 2283–2288 (2019). https://doi.org/10.1007/s11696-019-00778-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-019-00778-x