Abstract
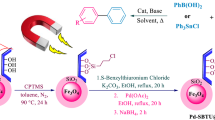
We revealed a new protocol to support Pt nanoparticles on magnetic ionic liquid functionalized organosilica nanoparticle matrix. The proposed catalyst system was synthesized by sol–gel condensation method, where two silica precursors [tetraethyl orthosilicate (TEOS) and bis-silylated ionic liquid monomer] were used to carry hydrophobic magnetic nanoparticles. The Pt metal was supported on the skeleton of magnetic organosilica hybrid nanoparticles by adsorbing platinum salt (Na2PtCl4) (replaced by ion exchange method). Furthermore, sodium borohydride was used to reduce platinum salt to Pt NPs. The synthesized catalytic system was used for CO2 hydrogenation reaction under high-pressure reaction condition. An external magnetic field was used to informally recover the catalyst and further recycled for three runs (without ionic liquid medium). Task-specific ionic liquids were also manipulated as a reaction medium not only to capture the CO2 gas but also to improve the reaction in a more precise manner. Desired results were attained while using 1,3-di (N, N-dimethylaminoethyl)-2-methylimidazolium nonafluorobutanesulfonate ([DAMI][CF3CF2CF2CF2SO3]) task-specific ionic liquid and we isolated the formic acid in good TON/TOF value. We easily reused the catalytic system up to 8 runs without decreasing the performance and stability of the catalytic system.












Similar content being viewed by others
References
Álvarez A, Bansode A, Urakawa A, Bavykina AV, Wezendonk TA, Makkee M, Gascon J, Kapteijn F (2017) Challenges in the Greener production of formates/formic acid, methanol, and DME by heterogeneously catalyzed CO2 hydrogenation processes. Chem Rev 117:9804–9838. https://doi.org/10.1021/acs.chemrev.6b00816
Bates ED, Mayton RD, Ntai I, Davis J (2002) CO2 capture by a TASK-specific ionic liquid. J Am Chem Soc 124:926–927. https://doi.org/10.1021/ja017593d
Beherns M (2016) Promoting the synthesis of methanol: understanding the requirements for an industrial catalyst for the conversion of CO2. Angew Chem Int Ed. 55:14906–14908
Bhanja P, Liu X, Modak A (2017) Pt and Pd nanoparticles immobilized on amine-functionalized hypercrosslinked porous polymer nanotubes as selective hydrogenation catalyst for α, β-unsaturated aldehydes. ChemistrySelect 2:7483–7757
Bhanja P, Modak A, Bhaumik A (2018) Supported porous nanomaterials as efficient heterogeneous catalysts for CO2 fixation reactions. Chem Eur J 24:7278–7297
Bhanja P, Modak A, Bhaumik A (2019) Porous organic polymers for CO2 storage and conversion reactions. ChemCatChem 11:244–257
Chaturvedi D (2011) Recent developments on task specific ionic liquids. Curr Org Chem 15:1236–1248. https://doi.org/10.2174/138527211795202997
Che M, Bennett CO (1989) The influence of particle size on the catalytic properties of supported metals. Adv Catal 36:55–172. https://doi.org/10.1016/S0360-0564(08)60017-6
Chen J, Zhang L, Zheng X, Zheng Y (2017) Revealing ruthenium and basicity synergetic effects in Ru–MgAl catalysts for isomerization of linoleic acid to conjugated linoleic acid. RSC Adv 7:54747–54755. https://doi.org/10.1039/C7RA10457J
Cuenya BR, Behafarid F (2015) Nanocatalysis: size-and shape-dependent chemisorption and catalytic reactivity. Surf Sci Rep 70:135–187. https://doi.org/10.1016/j.surfrep.2015.01.001
Diaz U, Boronat U, Corma A (2012) Hybrid organicinorganic structured materials as single-site heterogeneous catalysts. Proc R Soc London A 468:1927–1954. https://doi.org/10.1098/rspa.2012.0066
Farmer JA, Campbell CTC (2010) Ceria maintains smaller metal catalyst particles by strong metal-support bonding. Science 329:933–936. https://doi.org/10.1126/science.1191778
García-Diéguez M, Pieta IS, Herrera MC, Larrubia MA, Alemany LJ (2010) Nanostructured Pt- and Ni-based catalysts for CO2-reforming of methane. J Catal 270:136–145. https://doi.org/10.1016/j.jcat.2009.12.010
Giernoth R (2010) Task-specific ionic liquids. Angew Chem Int Ed 6:2834–2839. https://doi.org/10.1002/anie.200905981
Jayakumar S, Modak A, Guo M, Li H, Hu X, Yang Q (2017) Ultrasmall platinum stabilized on triphenylphosphine-modified silica for chemoselective hydrogenation. Chem Eur J 23:7791–7797
Koytsoumpa EI, Bergins C, Kakaras E (2018) The CO2 economy: review of CO2 capture and reuse technologies. J Supercrit Fluids 132:3–16. https://doi.org/10.1016/j.supflu.2017.07.029
Kudr J, Haddad Y, Richtera L, Heger Z, Cernak M, Adam V, Zitka O (2017) Magnetic nanoparticles: from design and synthesis to real world applications. Nanomaterials 7:243–272. https://doi.org/10.3390/nano7090243
Lai H, Xu F, Wang L (2018) A review of the preparation and application of magnetic nanoparticles for surface-enhanced Raman scattering. J Mater Sci 53:8677–8698. https://doi.org/10.1007/s10853-018-2095-9
Lee L-C, Xing X, Zhao Y (2017) Environmental engineering of Pd nanoparticle catalysts for catalytic hydrogenation of CO2 and bicarbonate. ACS Appl Mater Interfaces 9:38436–38444. https://doi.org/10.1021/acsami.7b10591
Li W, Wang H, Jiang X, Zhu J, Liu Z, Guo X, Song C (2018) A short review of recent advances in CO2 hydrogenation to hydrocarbons over heterogeneous catalysts. RSC Adv 8:7651–7669. https://doi.org/10.1039/C7RA13546G
Liu L, Corma A (2018) Metal catalysts for heterogeneous catalysis: from single atoms to nanoclusters and nanoparticles. Chem Rev 118:4981–5079. https://doi.org/10.1021/acs.chemrev.7b00776
Lockwood T (2017) A compararitive review of next-generation carbon capture technologies for coal-fired power plant. Energy Procedia 114:2658–2670. https://doi.org/10.1016/j.egypro.2017.03.1850
Lv X, Lu G, Wang Z-Q, Xu Z-N, Guo G-C (2017) Computational evidence for lewis base-promoted CO2 hydrogenation to formic acid on gold surfaces. ACS Catal 7:4519–4526. https://doi.org/10.1021/acscatal.7b00277
Ma J, Sun N, Zhang X, Zhao N, Xia F, Wei W, Sun Y (2009) A short review of catalysis for CO2 conversion. Catal Today 148:221–231. https://doi.org/10.1016/j.cattod.2009.08.015
Massart R (1981) Preparation of aqueous magnetic liquids in alkaline and acidic media. IEEE Trans Magn 17:1247–1248. https://doi.org/10.1109/TMAG.1981.1061188
Maucieria C, Barberab AC, Vymazalc J, Borina M (2017) A review on the main affecting factors of greenhouse gases emission in constructed wetlands. Agric For Meteorol 236:175–193. https://doi.org/10.1016/j.agrformet.2017.01.006
Modak A, Nandi M, Bhaumik A (2012) Titanium containing periodic mesoporous organosilica as an efficient catalyst for the epoxidation of alkenes. Catal Today 198:45–51
Modak A, Bhanja P, Bhaumik A (2018) Microporous nanotubes and nanospheres with iron-catechol sites: efficient lewis acid catalyst and support for Ag nanoparticles in CO2 fixation reaction. Chemistry 24:14189–14197. https://doi.org/10.1002/chem.201802319
Moret S, Dyson PJ, Laurenczy G (2012) Direct synthesis of formic acid from carbon dioxide by hydrogenation in acidic media. Nat Commun 5:1–7. https://doi.org/10.1038/ncomms5017
Onishi N, Iguchi M, Yang X, Kanega R, Kawanami H, Xu Q, Himeda Y (2018) Development of Effective catalysts for hydrogen storage technology using formic acid. Adv Energy Mater 8:6136–6142. https://doi.org/10.1002/aenm.201801275
Oschatz M, Antonietti M (2018) A search for selectivity to enable CO2 capture with porous adsorbents. Energy Environ Sci 11:57–70. https://doi.org/10.1039/C7EE02110K
Rafiee A, Khalilpour KR, Milani D, Panahi M (2018) Trends in CO2 conversion and utilization: a review from process systems perspective. J Environ Chem Eng 5:5771–5794. https://doi.org/10.1016/j.jece.2018.08.065
Rioux RM, Song H, Hoefelmeyer JD, Yang P, Somorjai GA (2005) High-Surface-area catalyst design: synthesis, characterization, and reaction studies of platinum nanoparticles in mesoporous SBA-15 silica. J Phys Chem B 109:2192–2202. https://doi.org/10.1021/jp048867x
Saeidi S, Amin NAS, Rahimpour MR (2014) Hydrogenation of CO2 to value-added products—a review and potential future developments. J CO2 Util 5:66–81. https://doi.org/10.1016/j.jcou.2013.12.005
Schauermann S, Hoffmann J, Johánek V, Hartmann J, Libuda J, Freund H-J (2002) Catalytic activity and poisoning of specific sites on supported metal nanoparticles. Angew Chem Int Ed 41:2532–2535. https://doi.org/10.1002/1521-3773
Schauermann S, Nilius N, Shaikhutdinov S, Freund H-J (2013) Nanoparticles for heterogeneous catalysis: new mechanistic insights. Acc Chem Res 46:1673–1681. https://doi.org/10.1021/ar300225s
Sethi VK, Savita V (2017) An innovative approach for carbon capture and sequestration on a thermal power plant through conversion to multi-purpose fuels—a feasibility study in Indian ContextJ. Energy Procedia 114(5):1288–1296. https://doi.org/10.1016/j.egypro.2017.03.1240
Srivastava V (2014) Selective hydrogenation of CO2 gas to formic acid over nanostructured Ru–TiO2 catalysts. Catal Lett 144:2221–2226. https://doi.org/10.1039/C6RA03660K
Tollefson J (2018) Sucking carbon dioxide from air is cheaper than scientists thought. Nature 558:173. https://doi.org/10.1038/d41586-018-05357-w
Upadhyay P, Srivastava V (2016a) Carbon sequestration: hydrogenation of CO2 to formic acid. PESD 10:13–34. https://doi.org/10.1515/pesd-2016-0022
Upadhyaya PR, Srivastava V (2016b) Selective hydrogenation of CO2 gas to formic acid over nanostructured Ru–TiO2 catalysts. RSC Adv 6:42297–42306. https://doi.org/10.1039/C6RA03660K
Upadhyaya PR, Srivastava V (2016c) Selective hydrogenation of CO2 using ruthenium nanoparticles intercalated montmorillonite clay. Lett Org Chem 13:380–387. https://doi.org/10.2174/1570178613666160815095733
Upadhyay P, Srivastava V (2017) Ionic liquid mediated in situ synthesis of Ru nanoparticles for CO2 hydrogenation reaction. Catal Lett 147:1051–1060. https://doi.org/10.1007/s10562-017-1995-7
Wang J, Gu H (2015) Novel metal nanomaterials and their catalytic applications. Molecules 20:17070–17092. https://doi.org/10.3390/molecules200917070
Yan X, Yang X (2018) Mechanistic insights into iridium catalyzed disproportionation of formic acid to CO2 and methanol: a DFT study. Organometallics 37:1519–1525. https://doi.org/10.1021/acs.organomet.7b00913
Yang H, Zhang C, Gao P, Wang H, Li X, Zhong L, Wei W, Sun Y (2017) A review of the catalytic hydrogenation of carbon dioxide into value-added hydrocarbons. Catal Sci Technol 7:4580–4598. https://doi.org/10.1039/C7CY01403A
Zhan G, Zeng HC (2017) Smart nanocatalysts with streamline shapes. ACS Cent Sci 3:794–799. https://doi.org/10.1021/acscentsci.7b00216
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Upadhyay, P.R., Gautam, P. & Srivastava, V. Magnetic organic-silica hybrid supported Pt nanoparticles for carbon sequestration reaction. Chem. Pap. 73, 2241–2253 (2019). https://doi.org/10.1007/s11696-019-00773-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-019-00773-2