Abstract
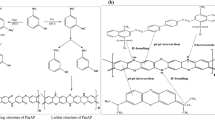
The polymer of m-phenylenediamine (poly(m-PDA)) which was synthesized by mild laccase catalysis in an aqueous buffer (10 mM citrate phosphate, pH 3) was investigated for the adsorption of several heavy metal ions including Pb2+, Cu2+, Co2+, and Cr3+. In the preliminary study for the adsorption of Pb2+ by poly(m-PDA), the adsorbed amount of Pb2+ was strongly influenced by solution pH being the highest in distilled water and decreased as solution pH was lowered from 5 to 2. The adsorption isotherms of the four heavy metal ions on poly(m-PDA) were well described by Langmuir equation. Poly(m-PDA) shows high maximum adsorption capacities (qmax) for Cu2+, Pb2+, and Cr3+ with the values of 556, 526, and 476 μmol g−1, respectively. This study proposes that the synthesis of polymeric absorbents to remove heavy metal ions from water can be accomplished through simple and environmentally friendly enzymatic methods.




Similar content being viewed by others
References
Alyuz B, Veli S (2009) Kinetics and equilibrium studies for the removal of nickel and zinc from aqueous solutions by ion exchange resins. J Hazard Mater 167:482–488. https://doi.org/10.1016/j.jhazmat.2009.01.006
Bohli T, Villaescusa I, Quederni A (2013) Comparative study of bivalent cationic metals adsorption Pb(II), Cd(II), Ni(II) and Cu(II) on olive stones chemically activated carbon. J Chem Eng Process Technol 4:158. https://doi.org/10.4172/2157-7048.1000158
Chong KH, Volesky B (1995) Description of two-metal biosorption equilibria by Langmuir-type models. Biotechnol Bioeng 47:451–460. https://doi.org/10.1002/bit.260470406
Djebbar M, Djafri F (2016) Adsorption of Cu(II) on natural and treated clays. Water Qual Res J Can 51:26–32. https://doi.org/10.2166/wqrjc.2015.051
Fu F, Wang Q (2011) Removal of heavy metal ions from wastewaters: a review. J Environ Manag 92:407–418. https://doi.org/10.1016/j.jenvman.2010.11.011
Gross RA, Kumar A, Kalra B (2001) Polymer synthesis by in vitro enzyme catalysis. Chem Rev 101:2097–2124
Ho YS (2006) Review of second-order models for adsorption systems. J Hazard Mater 136:681–689. https://doi.org/10.1016/j.jhazmat.2005.12.043
Huang MR, Peng QY, Li XG (2006) Rapid and effective adsorption of lead ions on fine poly(phenylenediamine) microparticles. Chem Eur J 12:4341–4350. https://doi.org/10.1002/chem.200501070
Ichinohe D, Muranaka T, Kise H (1998) Oxidative polymerization of phenylenediamines by enzyme and magnetic properties of the products. J Appl Polym Sci 70:717–721. https://doi.org/10.1002/(SICI)1097-4628(19981024)70:4%3C717:AID-APP11%3E3.0.CO;2-U
Kabbashi NA, Atieh MA, Al-Mamun A, Mirghami MES, Alam MDZ, Yahya N (2009) Kinetic adsorption of application of carbon nanotubes for Pb(II) removal from aqueous solution. J Environ Sci 21:539–544. https://doi.org/10.1016/S1001-0742(08)62305-0
Kongsuwan A, Patnukao P, Pavasant P (2009) Binary component sorption of Cu(II) and Pb(II) with activated carbon from Eucalyptus camaldulensis Dehn bark. J Ind Eng Chem 15:465–470. https://doi.org/10.1016/j.jiec.2009.02.002
Li XG, Huang MR, Duan W, Yang YL (2002) Novel multifunctional polymers from aromatic diamines by oxidative polymerizations. Chem Rev 102:2925–3030
Madhavi V, Lele SS (2009) Laccase: properties and applications. BioResources 4:1694–1717
Martínez-Huitle CA, Brillas E (2009) Decontamination of wastewaters containing synthetic organic dyes by electrochemical methods: a general review. Appl Catal B Environ 87:105–145. https://doi.org/10.1016/j.apcatb.2008.09.017
Pan B, Pan B, Zhang W, Lv L, Zhang Q, Zheng S (2009) Development of polymeric and polymer-based hybrid adsorbents for pollutants removal from waters. Chem Eng J 151:19–29. https://doi.org/10.1016/j.cej.2009.02.036
Persson I (2010) Hydrated metal ions in aqueous solution: how regular are their structures? Pure Appl Chem 82:1901–1917. https://doi.org/10.1351/PAC-CON-09-10-22
Raseda N, Hong S, Kwon OY, Ryu K (2014) Kinetic evidence for the interactive inhibition of laccase from Trametes versicolor by pH and chloride. J Microbiol Biotechnol 24:1673–1678
Raseda N, Park J, Ryu K (2016) Laccase-catalyzed polymerization of m-phenylenediamine in aqueous buffers. Korean J Chem Eng 33:3011–3015
Samiey B, Cheng CH, Wu J (2014) Organic-inorganic hybrid polymers as adsorbents for removal of heavy metal ions from solutions: a review. Materials 7:673–726
Su Z, Zhang L, Chai L, Wang H, Yu W, Wanga T, Yang J (2014) High-yield synthesis of poly(m-phenylenediamine) hollow nanostructures by a diethanolamine assisted method and their enhanced ability for Ag+ adsorption. New J Chem 38:3984–3991
Tolian G, Jafari SA, Zarei S (2015) Optimization of biosorption of nickel(II) and cadmium(II) by indigenous seaweed Enteromorpha using response surface methodology. Water Qual Res J Can 50:109–122. https://doi.org/10.2166/wqrjc.2015.007
Verma AK, Dash RR, Bhunia P (2012) A review on chemical coagulation/flocculation technologies for removal of colour from textile wastewaters. J Environ Manag 93:154–168. https://doi.org/10.1016/j.jenvman.2011.09.012
Xiao B, Thomas KM (2005) Adsorption of aqueous metal ions on oxygen and nitrogen functionalized nanoporous activated carbons. Langmuir 21:3892–3902
Yu W, Zhang L, Wang H, Chai L (2013) Adsorption of Cr(VI) using synthetic poly(m-phenylenediamine). J Hazard Mater 260:789–795. https://doi.org/10.1016/j.jhazmat.2013.06.045
Zhang Y, Wang L, Tian J, Li H, Luo Y, Sun X (2011) Ag@poly(m-phenylenediamine) core–shell nanoparticles for highly selective, multiplex nucleic acid detection. Langmuir 27:2170–2175
Zularisam AW, Ismail AF, Salim R (2006) Behaviours of natural organic matter in membrane filtration for surface water treatment—a review. Desalination 194:211–231. https://doi.org/10.1016/j.desal.2005.10.030
Acknowledgements
This work was financially supported by the faculty research grant from the University of Ulsan, Korea (2017-0170).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that no conflict of interest exists.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Xu, Y., Yoo, IK., Lee, H. et al. Adsorptive removal of heavy metal ions in water using poly(m-phenylenediamine) synthesized by laccase. Chem. Pap. 73, 1705–1711 (2019). https://doi.org/10.1007/s11696-019-00723-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-019-00723-y