Abstract
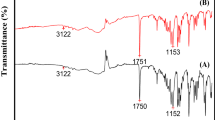
The major challenge faced by the pharmaceutical industry is the aggregation of drug nanoparticles. Controlling the surface forces between the particles with the help of surfactants and polymers may overcome this problem. An attempt has been made to develop nanocrystals of the telmisartan to improve its solubility by applying an anti-solvent crystallization in the presence of surfactants, polymers, and ultrasound. Various surfactants (sodium lauryl sulfate, Tween 80) and polymers (polyvinylpyrrolidone K30, polyvinylpyrrolidone K90, polyethylene glycol 6000, and hydroxypropyl methyl cellulose) at different concentrations (0.2, 0.4, 0.6 and 0.8% w/w) with and without ultrasound were studied to observe their effect on particle size and morphology. It was observed that crystal size was found to be 290 nm and 181.7 nm for Tween 80 and polyvinylpyrrolidone K30, respectively, which was further reduced by ~ 35% and 9% in the presence of ultrasound. The possible interactions among telmisartan, surfactants, and polymers in the solid state were analyzed by Fourier transform infrared spectroscopy, X-ray diffraction patterns, and morphology by scanning electron microscope, respectively. It was found that raw telmisartan showed the rod-like morphology with the size range of 6–10 µm and the recrystallized drug showed acicular-shaped morphology with size range of 100–400 nm.
Graphical abstract









Similar content being viewed by others
Abbreviations
- K :
-
Boltzmann constant
- N o :
-
Number of solute molecules per unit volume
- S :
-
Degree of supersaturation
- v :
-
Frequency of molecular transport into the solid–liquid interface
- σ :
-
Surface tension
- v s :
-
Volume of solute molecules
- T :
-
Temperature
References
Bajaj A, Rao MRP, Pardeshi A, Sali D (2012) Nanocrystallization by evaporative antisolvent technique for solubility and bioavailability enhancement of telmisartan. AAPS PharmaSciTech 12:1331–1340. https://doi.org/10.1208/s12249-012-9860-x
Beck C, Dalvi SV, Dave RN (2010) Controlled liquid antisolvent precipitation using a rapid mixing device. Chem Eng Sci 65:5669–5675. https://doi.org/10.1016/j.ces.2010.04.001
Chan H-K, Kwok PCL (2011) Production methods for nanodrug particles using the bottom-up approach. Adv Drug Deliv Rev 63:406–416. https://doi.org/10.1016/j.addr.2011.03.011
Dalvi SV, Dave RN (2009) Controlling Particle size of a poorly water-soluble drug using ultrasound and stabilizers in antisolvent precipitation. Ind Eng Chem Res 48:7581–7593. https://doi.org/10.1021/ie900248f
Farooq U, Patel R, Ali A (2018) Interaction of a surface active Ionic liquid with an antidepressant drug: micellization and spectroscopic studies. J Solut Chem 47:568–585. https://doi.org/10.1007/s10953-018-0739-7
Frizon F, Eloy JDO, Donaduzzi CM, Mitsui M-L, Marchetti J-M (2013) Dissolution rate enhancement of loratadine in polyvinyl pyrrolidone K-30 solid dispersion by solvent methods. Powder Technol 235:532–539. https://doi.org/10.1016/j.powtec.2012
Gradl J, Schwarzer H-C, Schwertfirm F, Manhart M, Peukert W (2006) Precipitation of nanoparticles in a T-mixer: coupling the particle population dynamic with hydrodynamics through direct numerical simulation. Chem Eng Process 45:908–916. https://doi.org/10.1016/j.cep.2005.11.012
Guo Z, Zhang M, Li H, Wang J, Kougoulos E (2005) Effect of ultrasound on antisolvent crystallization process. J Cryst Growth 273:555–563. https://doi.org/10.1016/j.jcrysgro.2004.09.049
Iiyas SU, Pendyala R, Marneni N (2014) Preparation, sedimentation, and agglomeration of nanofluids. Chem Eng Technol 37:1–12. https://doi.org/10.1002/ceat.201400268
Johnson BK, Prud’homme RK (2003) Flash nanoprecipitation of organic actives and block copolymers using a confined impinging jets mixer. Aust J Chem 56:1021–1024. https://doi.org/10.1071/CH03115
Jones AG (2002) Crystallization process system. Butterworth-Heinemann, London
Kane RJ, Kuchekar BS (2010) Preparation, Physicochemical characterization dissolution and formulation studies of telmisartan cyclodextrin inclusion complexes. Asia J Pharm 4:52–59. https://doi.org/10.4103/0973-8398.63983
Khossravi D (1997) Drug-surfactant interactions: effect on transport properties. Int J Pharm 155:179–190
Liversidge GG, Cundy KC (1995) Particle size reduction for improvement of oral bioavailability of hydrophobic drugs: I. Absolute oral bioavailability of nanocrystalline danazol in beagle dogs. Int J Pharm 125:91–97
Luque de Castro MD, Priego-Capote F (2007) Ultrasound-assisted crystallization (sonocrystallization). Ultrason Sonochem 14:717–724. https://doi.org/10.1016/j.ultsonch.2006.12.004
Malvern Instrument Ltd. (2004) Zetasizer nano series user manual, MAN0317 Issue 2.2, Worcestershire, UK
Matteucci ME, Hotze MA, Johnston KP, Williams RO III (2006) Drug nanoparticles by antisolvent precipitation: mixing energy versus surfactant stabilization. Langmuir 22:8951–8959. https://doi.org/10.1021/la061122t
Muller RH, Peters K (1998) Nanosuspensions for the formulation of poorly soluble drugs I. preparation by size-reduction technique. Int J Pharm 160:229–237
Noyes AA, Whitney WR (1897) The rate of solution of solid substances in their own solutions. J Am Chem Soc 19:930–935
Park M-W, Yeo S-D (2010) Antisolvent crystallization of roxithromycin and the effect of ultrasound. Sep Sci Technol 45:1402–1410. https://doi.org/10.1080/01496391003689538
Park M-W, Yeo S-D (2012) Antisolvent crystallization of carbamazepine from organic solutions. Chem Eng Res Des 90:2202–2208. https://doi.org/10.1016/j.cherd.2012.05.001
Park J, Cho W, Cha K-H, Ahn J, Han K, Hwang S-J (2013) Solubilization of the poorly water-soluble drug, telmisartan using supercritical anti-solvent (SAS) Process. Int J Pharm 441:50–65. https://doi.org/10.1016/j.ijpharm.2012.12.020
Peukert W, Schwarzer H-C, Stenger F (2005) Control of aggregation in production and handling of nanoparticles. Chem Eng Process 44:245–252. https://doi.org/10.1016/j.cep.2004.02.018
Rastogi R, Kaushal R, Tripathi SK, Sharma AL, Kaur I, Bharadwaj LM (2008) Comparative study of carbon nanotube dispersion using surfactants. J Colloid Interface Sci 328:421–428. https://doi.org/10.1016/j.jcis.2008.09.015
Sander JRG, Zeiger BW, Suslick KS (2014) Sonocrystallization and sonofragmentation. Ultrason Sonochem 21:1908–1915. https://doi.org/10.1016/j.ultsonch.2014.02.005
Sangwai M, Vavia P (2013) Amorphous ternary cyclodextrin nanocomposites of telmisartan for oral drug delivery: improved solubility and reduced pharmacokinetic variability. Int J Pharm 453:423–432. https://doi.org/10.1016/j.ijpharm.2012.08.034
Sharma C, Desai MA, Patel SR (2018) Ultrasound-assisted antisolvent crystallization of telmisartan using Dimethyl sulfoxide as an organic solvent. Cryst Res Technol 80001:1–9. https://doi.org/10.1002/crat.201800001
Singh SK, Nilsson S (1999) Thermodynamics of interaction between some cellulose ethers and SDS by titration microcalorimetry: I. EHEC and HPMC. J Colloid Interface Sci 213:133–151
Tran PHL, Tran HTT, Lee B-J (2008) Modulation of microenvironmental pH and crystallinity of ionizable telmisartan using alkalizers in solid dispersion for controlled release. J Control Release 129:59–65. https://doi.org/10.1016/j.jconrel.2008.04.001
Tran PH-L, Tran TT-D, Park J-B, Min DH, Choi H-G, Han H-K, Rhee Y-S, Lee B-J (2011) Investigation of physicochemical factors affecting the stability of pH-modulated solid dispersion and a tablet during storage. Int J Pharm 414:48–55. https://doi.org/10.1016/j.ijpharm.2011.04.065
Varshosaz J, Minayian M, Ahmadi M, Ghassami E (2016) Enhancement of solubility and antidiabetic effects of Repaglinide using spray drying technique in STZ-induced diabetic rats. Pharm Dev Technol 22:1–10. https://doi.org/10.3109/10837450.2016.1143001
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sharma, C., Desai, M.A. & Patel, S.R. Effect of surfactants and polymers on morphology and particle size of telmisartan in ultrasound-assisted anti-solvent crystallization. Chem. Pap. 73, 1685–1694 (2019). https://doi.org/10.1007/s11696-019-00720-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-019-00720-1