Abstract
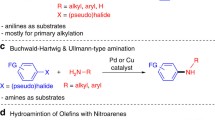
A novel protocol for chemoselective reduction of aromatic nitro compounds to aromatic amines has been established. The metal-free reduction goes through a hydrogen transfer process. Various easily reducible functional groups can be well tolerated under the optimized reaction conditions.




Similar content being viewed by others
References
Ankner T, Hilmersson G (2007) Instantaneous SmI2/H2O/amine mediated reduction of nitroalkanes and α, β-unsaturated nitroalkenes. Tetrahedron Lett 48:5707–5710. https://doi.org/10.1016/j.tetlet.2007.05.105
Arnold H, Dobert F, Gaube J (2008) In handbook of heterogeneous catalysis. Wiley-Interscience, New York, pp 3266–3284
Bae JW, Cho YJ, Lee SH, Yoon COM, Yoon CM (2000) Aone-pot synthesis of N-alkylaminobenzenes from nitroaromatics: reduction followed by reductive amination using B10H14. Chem Commun 19:1857–1858. https://doi.org/10.1039/b005194m
Basu MK, Becker FF, Banik BK (2000) Ultrasound promoted highly efficient reduction of aromatic nitro compounds to the aromatic amines by samarium/ammonium chloride. Tetrahedron Lett 41:5603–5606. https://doi.org/10.1016/S0040-4039(00)00917-5
Berthold H, Schotten T, Honig H (2002) Transfer hydrogenation in ionic liquids under microwave irradiation. Synthesis 11:1607–1610. https://doi.org/10.1055/s-2002-33349
Blaser HU (2006) A golden boost to an old reaction. Science 313:312–313. https://doi.org/10.1126/science.1131574
Blaser HU, Siegrist U, Steiner H, Studer M, Sheldon RA (2001) Fine chemicals through heterogeneous catalysi-sed. Wiley, Weinheim, p 389
Brinkman HR, Miles WH, Hilborn MD, Smith MC (1996) The reduction of nitrobenzenes by triethylsilane using Wilkinson’s catalyst. Synth Commun 26:973–980. https://doi.org/10.1080/00397919608003701
Cantillo D, Baghbanzadeh M, Kappe CO (2012) In situ generated iron oxide nanocrystals as efficient and selective catalysts for the reduction of nitroarenes using a continuous flow method. Angew Chem Int Ed 51:210190–210193. https://doi.org/10.1002/anie.201205792
Cantillo D, Moghaddam MM, Kappe CO (2013) Hydrazine mediated reduction of nitro and azide functionalities catalyzed by highly active and reusable magnetic iron oxide nanocrystals. J Org Chem 78:4530–4542. https://doi.org/10.1021/jo400556g
Chandrappa S, Vinaya K, Ramakrishnappa T, Rangappa KS (2010) An efficient method for aryl nitro reduction and cleavage of azo compounds using iron powder/calcium chloride. Synlett 20:3019–3022. https://doi.org/10.1055/s-0030-1259067
Chen Y, Qiu J, Wang X, Xiu J (2006) Preparation and application of highly dispersed gold nanoparticles supported on silica for catalytic hydrogenation of aromatic nitro compounds. J Catal 242:227–230. https://doi.org/10.1016/j.jcat.2006.05.028
Chinnappan A, Kim H (2013) Transition metal based ionic liquid (bulk and nanofiber composites) used as catalyst for reduction of aromatic nitro compounds under mild conditions. RSC Adv 3:3399–3406. https://doi.org/10.1039/c2ra22639a
Corma A, Serna P (2006) Chemoselective hydrogenation of nitro compounds with supported gold catalysts. Science 313:332–334. https://doi.org/10.1126/science.1128383
Corma A, Serna P, Concepcion P, Calvino JJ (2008) Transforming nonselective into chemoselective metal catalysts for the hydrogenation of substituted nitroaromatics. J Am Chem Soc 130:8748–8753. https://doi.org/10.1021/ja800959g
Cui X, Zhang Y, Shi F, Deng Y (2011) Ruthenium catalyze nitro and nitrile compounds coupling with alcohols: alternative route for N-substituted amine Synthesis. Chem. Eur. J 17:2587–2591. https://doi.org/10.1002/chem.201003095
Desai DG, Swami SS, Dabhade SK, Ghagare MG (2001) FeS–NH4Cl–CH3OH–H2O: an efficient and inexpensive system for reduction of nitroarenes to anilines. Synth Commun 31:1249–1251. https://doi.org/10.1081/SCC-100104012
Feng C, Liu Y, Peng S, Shuai Q, Deng G, Li C-J (2010) Ruthenium catalyzed tertiary amine formation from nitroarenes and alcohols. Org Lett 12:4888–4891. https://doi.org/10.1021/ol1020527
Gallagher WP, Marlatt M, Livingston R, Kiau S, Muslehiddinoglu J (2012) The development of a scalable, chemoselective nitro reduction. Org Process Res Dev 16:1665–1668. https://doi.org/10.1021/op3002239
Gamble AB, Garner J, Gordon CP, O’Conner SMJ, Keller PA (2007) Aryl nitro reduction with iron powder or stannous chloride under ultrasonic irradiation. Synth Commun 37:2777–2786. https://doi.org/10.1080/00397910701481195
Gawande MB, Guo H, Rathi AK, Branco PS, Chen Y, Rajender S, Varmad RS, Peng DL (2013) First application of core shell Ag@Ni magnetic nanocatalyst for transfer hydrogenation reactions of aromatic nitro and carbonyl compounds. RSC Adv 3:1050–1054. https://doi.org/10.1039/c2ra22143h
Guillena G, Ramon DJ, Yus M (2010) Hydrogen autotransfer in the N-Alkylation of amines and related compounds using alcohols and amines as electrophiles. Chem Rev 110:1611–1641. https://doi.org/10.1021/cr9002159
Haber F (1898) About gradual reduction of the nitrobenzene with limited cathode potential. Zeitschrift fuer Ele-ktrochemie und Angewandte Physikalische Chemie 22:506–513. https://doi.org/10.1002/bbpc.18980042204
He L, Wang LC, Sun H, Ni J, Cao Y, He HY, Fan KN (2009) Efficient and selective room temperature gold catalyzed reduction of nitro compounds with co and H2O as the hydrogen source. Angew Chem Int Ed 48:9538–9541. https://doi.org/10.1002/anie.200904647
Jagadeesh RV, Wienhoefer G, Westerhaus FA, Surkus AE, Pohl MM, Junge H, Junge K, Beller M (2011) Efficient and highly selective iron catalyzed reduction of nitroarenes. Chem Commun 47:10972–10974. https://doi.org/10.1039/c1cc13728j
Jagadeesh RV, Surkus AE, Junge H, Pohl MM, Radnik J, Rabeah J, Huan H, Schuenemann V, Brueckner A, Beller M (2013) Nanoscale Fe2O3 based catalysts for selective hydrogenation of nitroarenes to anilines. Science 342:1073–1076. https://doi.org/10.1126/science.1242005
Jagadeesh RV, Banerjee D, Arockiam PB, Junge H, Junge K, Pohl MM, Radnik J, Brueckner A, Beller M (2015) Highly selective transfer hydrogenation of functionalised nitroarenes using cobalt based nanocatalysts. Green Chem 17:898–902. https://doi.org/10.1039/C4GC00731J
Junge K, Wendt B, Shaikh N, Beller M (2010) Iron catalyzed selective reduction of nitroarenes to anilines using organosilanes. Chem Commun 46:1769–1777. https://doi.org/10.1039/b924228g
Kabalka GW, Varma RS (1991) Reduction of nitro and nitroso compounds. In: Trost BM, Fleming I (eds) Comprehensive organic synthesis, vol 8, 1st edn. Pergamon Press, Oxford, p 363
Kelly SM, Lipshutz BH (2014) Chemoselective reductions of nitroaromatics in water at room temperature. Org Lett 16:98–101. https://doi.org/10.1021/ol403079x
Khan FA, Dash J, Sudheer C, Gupta RK (2003) Chemoselective reduction of aromatic nitro and azo compounds in ionic liquids using zinc and ammonium salts. Tetrahedron Lett 44:7783–7787. https://doi.org/10.1016/j.tetlet.2003.08.080
Kim S, Kim E, Kim BM (2011) Fe3O4 nanoparticles a conveniently reusable catalyst for the reduction of nitroarenes using hydrazine hydrate. Chem Asian J 6:1921–1925. https://doi.org/10.1002/asia.201100311
Kim JH, Park JH, Chung YK, Park KH (2012) Ruthenium nanoparticle-catalyzed, controlled and chemoselective hydrogenation of nitroarenes using ethanol as a hydrogen source. Adv Synth Catal 354:2412–2418. https://doi.org/10.1002/adsc.201200356
Kumarraja M, Pitchumani K (2004) Simple and efficient reduction of nitroarenes by hydrazine in Faujasite zeolites. Appl Catal A 265:135–139. https://doi.org/10.1016/j.apcata.2004.01.009
Kumbha PS, Sanehez-Valente J, Figueras F (1998) Reduction of aromatic nitro compounds with hydrazine hydrate in the presence of the lron(III) oxide-MgO catalyst prepared from a Mg–Fe hydrotalcite precursor. Tetrahedron Lett 39:2573–2574. https://doi.org/10.1016/S0040-4039(98)00264-0
Kunfi A, Szabó V, Mastalir A, Bucsi I, Mohai M, NémethP Bertóti I, London G (2017) Palladium on polydopamine: its true potential in catalytic transfer hydrogenations and heck coupling reactions. ChemCatChem 9:3236–3244. https://doi.org/10.1002/cctc.201700609
Lawrence SA (2004) Amines: synthesis properties and applications. Cambridge University Press, Cambridge
Leleu S, Papamicae C, Marsais F, Dupas G, Levacher V (2004) Preparation of axially chiral quinolinium salts related to NAD+ models: new investigations of these biomimetic models as chiral amide-transferring agents. Tetrahedron Asymmetry 15:3919–3928. https://doi.org/10.1016/j.tetasy.2004.11.004
Li B, Xu Z (2009) A nonmetal catalyst for molecular hydrogen activation with comparable catalytic hydrogenation capability to noble metal catalyst. J Am Chem Soc 131:16380–16382. https://doi.org/10.1021/ja9061097
Li J, Shi XY, Bi YY, Wei JF, Chen ZG (2011) Pd nanoparticles in ionic liquid brush a highly active and reusable heterogeneous catalytic assembly for solvent free or on water hydrogenation of nitroarene under mild conditions. ACS Catal 1:657–664. https://doi.org/10.1021/cs200105u
Liu Y, Chen W, Feng C, Deng G (2011) Ruthenium catalyzed one-pot aromatic secondary amine formation from nitroarenes and alcohols. Chem Asian J 6:1142–1146. https://doi.org/10.1002/asia.201000945
Mahdavi H, Tamami B (2005) Reduction of nitro aryl compounds with zinc in the presence of Poly[N-(2-aminoethyl)acrylamido]-trimethylammonium chloride as a phase-transfer catalyst. Synth Commun 35:1121–1127. https://doi.org/10.1081/SCC-200054221
Maity SK, Pradhan NC, Patwardhan AV (2006) Kinetics of the reduction of nitrotoluenes by aqueous ammonium sulfide under liquid–liquid phase transfer catalysis. Appl Catal A 301:251–258. https://doi.org/10.1016/j.apcata.2005.12.015
Mazaheri O, Kalbasi RJ (2015) Preparation and characterization of Ni/mZSM-5 zeolite with a hierarchical pore structure by using KIT-6 as silica template: an efficient bi-functional catalyst for the reduction of nitro aromatic compounds. RSC Adv 53:4398–34414. https://doi.org/10.1039/c5ra02349a
McLaughlin MA, Barnes DM (2006) A practical and selective reduction of nitroarenes using elemental sulfur and mild base. Tetrahedron Lett 47:9095–9097. https://doi.org/10.1016/j.tetlet.2006.10.079
Mondal U, Puthankot A, Sen S, Singh G (2016) Novelties of triphasic phase transfer catalysed zinin reduction of nitrochlorobenzene by H2S-laden monoethanolamine. RSC Adv 6:23666–23676. https://doi.org/10.1039/c5ra26856g
Motoyama Y, Kamo K, Nagashima H (2009) Catalysis in polysiloxane gels platinum-catalyzed hydrosilylation of polymethylhydrosiloxaneleading to reusable catalysts for reduction of nitroarenes. Org Lett 11:1345–1348. https://doi.org/10.1021/ol9001366
Nakamula I, Yamanoi Y, Imaoka T, Yamamoto K, Nishihara H (2011) A uniform bimetallic rhodium/iron nanoparticle catalyst for the hydrogenation of olefins and nitroarenes. Angew Chem Int Ed 50:5830–5833. https://doi.org/10.1002/anie.201102836
Niknam K, Kiasat AR, Kazemi F, Hossieni A (2003) Efficient reduction of nitroarenes to the corresponding anilines with sulfur in basic media under solvent-free conditions. Phosphorus Sulfur Silicon 178:1385–1389. https://doi.org/10.1080/10426500390200701
Nishimura S (2001) Handbook of heterogeneous catalytic hydrogenation for organic synthesis. Wiley Interscience, New York, p 784
Nixon TD, Whittlesey MK, Williams JMJ (2009) Transitio-n metal catalysed reactions of alcohols using borrowing hydrogen methodology. Dalton Trans 5:753–762. https://doi.org/10.1039/B813383B
Noronha RG, Romao CC, Fernandes AC (2009) Highly chemo and regioselective reduction of aromatic nitro compounds using the system silane/oxo-rhenium complexes. J Org Chem 74:6960–6964. https://doi.org/10.1021/jo9008657
Nugent TC (2010) Chiral amine synthesis: methods, developments and applications. Wiley, Weinheim
Orlandi M, Brenna D, Harms R, Jost S, Benaglia M (2018) Recent developments in the reduction of aromatic and aliphatic nitro compounds to amines. Org Process Res Dev 22:430–445. https://doi.org/10.1021/acs.oprd.6b00205
Pandarus V, Ciriminna R, Beland F, Pagliaro M (2011) A new class of heterogeneous platinum catalysts for the chemoselective hydrogenation of nitroarenes. Adv Synth Catal 353:1306–1316. https://doi.org/10.1002/adsc.201000945
Patai S (1996) The chemistry of amines, nitroso, nitro and related groups. Wiley, Chichester
Pehlivan L, Métay E, Laval S, Dayoub W, Demonchaux P, Mignani G, Lemaire M (2010) Iron catalyzed selective reduction of nitro compounds to amines. Tetrahedron Lett 51:1939–1941. https://doi.org/10.1016/j.tetlet.2010.01.067
Pogorelic I, Filipan-Litvic M, Merkas S, Ljubic G, Cepanec I, Litvic M (2007) Rapid, efficient and selective reduction of aromatic nitro compounds with sodium borohydride and raney nickel. J Mol Catal A Chem 274:202–207. https://doi.org/10.1016/j.molcata.2007.05.020
Rahaim RJ, Maleczka RE (2005) Pd-catalyzed silicon hydride reductions of aromatic and aliphatic nitro groups. Org Lett 7:5087–5090. https://doi.org/10.1021/ol052120n
Rahaim RJ, Maleczka RE (2006) Palladium-catalyzed silane/siloxane reductions in the one-pot conversion of nitro compounds into their amines, hydroxylamines, amides, sulfonamides, and carbamates. Synthesis 19:3316–3340. https://doi.org/10.1055/s-2006-950231
Rai RK, Mahata A, Mukhopadhyay S, Gupta S, Li PZ, Nguyen KT, Zhao Y, Pathak B, Singh SK (2014) Room temperature chemoselective reduction of nitro groups using non-noble metal nanocatalysts in water. Inorg Chem 53:2904–2909. https://doi.org/10.1021/ic402674z
Ricci A (2008) Amino group chemistry: from synthesis to the life sciences. Wiley, Weinheim
Saha A, Ranu B (2008) Highly Chemoselective reduction of aromatic nitro compounds by copper nanoparticles ammonium formate. J Org Chem 73:6867–6870. https://doi.org/10.1021/jo800863m
Sharma U, Kumar P, Kumar N, Kumar V, Singh B (2010) Highly chemo and regioselective reduction of aromatic nitro compounds catalyzed by recyclable Copper(II) as well as Cobalt(II) phthalocyanines. Adv Synth Catal 352:1834–1840. https://doi.org/10.1002/adsc.201000191
Sharma U, Verma PK, Kumar N, Kumar V, Bala M, Singh B (2011) Phosphane-free green protocol for selective nitro reduction with an iron-based catalyst. Chem Eur J 17:5903–5907. https://doi.org/10.1002/chem.201003621
Sharma S, Kumar M, Kumar V, Kumar N (2014) Metal Free transfer hydrogenation of nitroarenes in water with vasicine: revelation of organocatalytic facet of an abundant alkaloid. J Org Chem 79:9433–9439. https://doi.org/10.1021/jo5019415
Shi Q, Lu R, Jin K, Zhang Z, Zhao D (2006) Simple and ecofriendly reduction of nitroarenes to the corresponding aromatic amines using polymer-supported hydrazine hydrate over iron oxide hydroxide catalyst. Green Chem. https://doi.org/10.1039/b606705k
Shi X, Wang X, Shang X, Zou X, Ding W, Lu X (2017) High performance and active sites of a ceria supported palladium catalyst for solvent free chemoselective hydrogenation of nitroarenes. ChemCatChem 9:3743–3751. https://doi.org/10.1002/cctc.201700631
Sorribes I, Wienhoefer G, Vicent C, Junge K, Llusar R, Beller M (2012) Chemoselective transfer hydrogenation to nitroarenes mediated by cubane type Mo3S4 cluster catalysts. Angew Chem Int Ed 51:7794–7798. https://doi.org/10.1002/anie.201202584
SuwiBski J, Wagner P (1996) Reduction of aromatic nitrocompounds by sodium borohydride in methanol in the presence of sodium methoxide. Tetrahedron 52:9541–9552. https://doi.org/10.1016/0040-4020(96)00491-7
Takasaki M, Motoyama Y, Higashi K, Yoon SH, Mochida I, Nagashima H (2008) Chemoselective hydrogenation of nitroarenes with carbon nanofiber-supported platinum and palladium nanoparticles. Org Lett 10:1601–1604. https://doi.org/10.1021/ol800277a
Tang CH, He L, Liu YM, Cao Y, He HY, Fan KN (2011) Direct one-pot reductive N-alkylation of nitroarenes by using alcohols with supported gold catalysts. Chem Eur J 17:7172–7177. https://doi.org/10.1002/chem.201100393
Ullmann F (2012) Ullmann’s encyclopedia of industrial chemistry, vol A2. Verlag Chemie, Weinheim, pp 647–718
Vass A, Dudas J, Toth J, Varma RS (2001) Solvent free reduction of aromatic nitro compounds with alumina supported hydrazine under microwave irradiation. Tetrahedron Lett. 42:5347–5349. https://doi.org/10.1016/S0040-4039(01)01002-4
Wang J, Yuan Z, Nie R, Hou Z, Zheng X (2010) Hydrogenation of nitrobenzene to aniline over silica gel supported nickel catalysts. Ind Eng Chem Res 49:4664–4669. https://doi.org/10.1021/ie1002069
Wang X, Cárdenas-Lizana F, Keane MA (2014) Toward sustainable chemoselective nitroarene hydrogenation using supported gold as catalyst. ACS Sustain Chem Eng 2:2781–2789. https://doi.org/10.1021/sc500544s
Watson AJA, Williams JMJ (2010) The Give and Take of Alcohol Activation. Science 329:635–636. https://doi.org/10.1126/science.1191843
Wienhoefer G, Sorribes I, Boddien A, Westerhaus F, JungeK Junge H, Llusar R, Beller M (2011) General and selective iron catalyzed transfer hydrogenation of nitroarenes without base. J Am Chem Soc 133:12875–12879. https://doi.org/10.1021/ja2061038
Wu H, Zhuo L, He Q, Liao X, Shi B (2009) Heterogeneous hydrogenation of nitrobenzenes over recyclable Pd(0) nanoparticle catalysts stabilized by polyphenol grafted collagen fibers. Appl Catal 366:44–56. https://doi.org/10.1016/j.apcata.2009.06.024
Xie Y, Liu S, Liu Y, Wen Y, Deng GJ (2012) Palladium catalyzed one pot diarylamine formation from nitroarenes and cyclohexanones. Org Lett 14:1692–1695. https://doi.org/10.1021/ol3002442
Yadav GD, Jadhav YB, Sengupta S (2003) Novelties of kinetics and mechanism of liquid–liquid phase transfer catalysed reduction of p-nitroanisole to p-anisidine. Chem Eng Sci 58:2681–2689. https://doi.org/10.1016/S0009-2509(03)00099-X
Yamane Y, Liu X, Hamasaki A, Ishida T, Haruta M, Yokoyama T, Tokunaga M (2009) One-pot synthesis of indoles and aniline derivatives from nitroarenes under hydrogenation condition with supported gold nano-particles. Org Lett 11:5162–5165. https://doi.org/10.1021/ol902061j
Yang XJ, Chen B, Zheng LQ, Wu LZ, Tung CH (2014) Highly efficient and selective photocatalytic hydrogenation of functionalized nitrobenzenes. Green Chem 16:1082–1086. https://doi.org/10.1039/c3gc42042f
Zhou YH, Yang Q, Chen YZ, Jiang HL (2017) Low-cost CuNiMIL-101 as an excellent catalyst toward cascade reaction integration of ammonia borane dehydrogenation with nitroarene hydrogenation. Chem Commun 53:12361–12364. https://doi.org/10.1039/c7cc06530b
Zimmermann V, Avemaria F, Brase S (2007) Chemoselective reduction of nitroarenes in the presence of acid-sensitive functional groups: solid phase syntheses of amino aryl azides and benzotriazoles. J Comb Chem 9:200–203. https://doi.org/10.1021/cc060116c
Acknowledgements
This work was supported by the National Natural Science Foundation of China (21172185, 21372187, 21606202).
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Shuai, Q., Li, J., Zhao, F. et al. Metal-free chemoselective reduction of nitroaromatics to anilines via hydrogen transfer strategy. Chem. Pap. 73, 965–975 (2019). https://doi.org/10.1007/s11696-018-0634-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-018-0634-0