Abstract
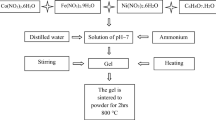
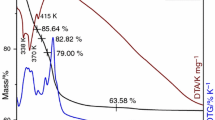
Chromium substituted cobalt ferrites CoFe2−xCrxO4 (x = 0; 1; 1.5) were synthesized through a soft chemistry method—the gluconate precursor route. The gluconate precursors were characterized by infrared spectroscopy (IR), ultraviolet–visible spectroscopy and thermal analysis. The spinel oxide powders were investigated by X-ray diffraction (XRD), scanning electron microscopy (SEM), Mössbauer spectroscopy and Brunauer–Emmett–Teller N2 adsorption–desorption analyses. XRD indicated the formation of the spinel-type phase with good crystallinity. The mean crystalline domains size decreased from 23.8 to 17.8 nm with the increase in the chromium content. SEM revealed faceted particles for which the particle sizes varied significantly with the chromium content. The chromium substituted cobalt ferrites were found to have high catalytic performance.









Similar content being viewed by others
References
Bellal YH, Zouaoui-Mahzoul N, Lounas I, Benadda A, Benrabaa R, Auroux A, Meddour-Boukhobza L, Djadoun A (2017) Cobalt and cobalt-iron spinel oxides as bulk and silica supported catalysts in the ethanol combustion reaction. J Mol Catal A Chem 426:97–106. https://doi.org/10.1016/j.molcata.2016.11.005
Bhasker SU, Ramana Reddy MV (2015) Effect of chromium substitution on structural, magnetic and electrical properties of magneto-ceramic cobalt ferrite nano-particles. J Sol Gel Sci Technol 73:396–402. https://doi.org/10.1007/s10971-014-3546-7
Carta D, Casula MF, Falqui A, Loche D, Mountjoy G, Sangregorio C, Corrias A (2009) A structural and magnetic investigation of the inversion degree in ferrite nanocrystals MFe2O4 (M = Mn Co, Ni). J Phys Chem C 113:8606–8615. https://doi.org/10.1021/jp901077c
Chagas CA, De Souza EF, De Carvalho MCNA, Martins RL, Schmal M (2016) Cobalt ferrite nanoparticles for the preferential oxidation of CO. Appl Catal A 519:139–145. https://doi.org/10.1016/j.apcata.2016.03.024
Chen J, Zhang X, Arandiyan H, Peng Y, Chang H, Li J (2013) Low temperature complete combustion of methane over cobalt chromium oxides catalysts. Catal Today 201:12–18. https://doi.org/10.1016/j.cattod.2012.03.026
Devaiah D, Smirniotis PG (2017) Effects of the Ce and Cr contents in Fe–Ce–Cr ferrite spinels on the high-temperature water–gas shift reaction. Ind Eng Chem Res 56:1772–1781. https://doi.org/10.1021/acs.iecr.6b04707
Dumitru R, Papa F, Balint I, Culita DC, Munteanu C, Stanica N, Ianculescu A, Diamandescu L, Carp O (2013) Mesoporous cobalt ferrite: a rival of platinum catalyst in methane combustion reaction. Appl Catal A 467:178–186. https://doi.org/10.1016/j.apcata.2013.07.013
Fino D, Solaro S, Russo N, Saracco G, Specchia V (2007) Catalytic removal of methane over thermal-proof nanostructured catalysts for CNG engines. Top Catal 42–43:449–454. https://doi.org/10.1007/s11244-007-0223-x
Gingasu D, Diamandescu L, Mindru I, Marinescu G, Culita DC, Calderon-Moreno JM, Preda S, Bartha C, Patron L (2015a) Chromium substituted cobalt ferrites by glycine–nitrates process. Croat Chem Acta 88:445–451. https://doi.org/10.5562/cca2743
Gingasu D, Mindru I, Culita DC, Patron L, Calderon-Moreno JM, Osiceanu P, Preda S, Oprea O, Parvulescu V, Teodorescu V, Walsh JPS (2015b) Structural, magnetic and catalytic properties of cobalt chromite through precursor method. Mater Res Bull 62:52–64. https://doi.org/10.1016/j.materresbull.2014.11.009
Goldman A (2006) Modern ferrite technology, 2nd edn. Springer, New York
Hankare PP, Sankpal UB, Patil RP, Mulla IS, Lokhande PD, Gajbhiye NS (2009) Synthesis and characterization of CoCxFe2−xO4 nanoparticles. J Alloys Compd 485:98–801. https://doi.org/10.1016/j.jallcom.2009.06.087
Hankare PP, Sankpal UB, Patil RP, Lokhande PD, Sasikala R (2011) Synthesis, characterization and catalytic activity of chromium substituted cobalt ferrospinels. Mater Sci Eng B 176:103–109. https://doi.org/10.1016/j.mseb.2010.10.005
Iqbal MJ, Siddiquah MR (2008) Electrical and magnetic properties of chromium-substituted cobalt ferrite nanomaterials. J Alloys Compd 453:513–518. https://doi.org/10.1016/j.jallcom.2007.06.105
Jauhar S, Singhal S (2014) Substituted cobalt nano-ferrites, CoMxFe2−xO4 (M = Cr3+, Ni2+, Cu2+, Zn2+; 0.2 ≤ x ≤ 1.0) as heterogeneous catalysts for modified Fenton’s reaction. Ceram Int 40:11845–11855. https://doi.org/10.1016/j.ceramint.2014.04.019
Kantserova MR, Gavrilenko KS, Kosmambetova GR, Il’in VG, Orlik SN (2003) Deep oxidation of methane over nano-sized ferrites with spinel structures. Theor Exp Chem 39:322–329. https://doi.org/10.1023/B:THEC.0000003494.21579.14
Koseoglu Y, Oleiwi MIO, Yilgin R, Kocbay AN (2012) Effect of chromium addition on the structural, morphological and magnetic properties of nano-crystalline cobalt ferrite system. Ceram Int 38:6671–6676. https://doi.org/10.1016/j.ceramint.2012.05.055
Lever ABP (1984) Inorganic electronic spectroscopy, 2nd edn. Elsevier Publishing Company, Amsterdam
Liu M, Lu M, Wang L, Xu S, Zhao J, Li H (2016) Mössbauer study on the magnetic properties and cation distribution of CoFe2O4 nanoparticles synthesized by hydrothermal method. J Mater Sci 51:5487–5492. https://doi.org/10.1007/s10853-016-9853-3
Meshkani F, Rezaei M (2015) Preparation of nanocrystalline metal (Cr, Al, Mn, Ce, Ni, Co and Cu) modified ferrite catalysts for the high temperature water gas shift reaction. Renew Energy 74:588–598. https://doi.org/10.1016/j.renene.2014.08.037
Mindru I, Gingasu D, Culita DC, Marinescu G, Patron L (2014) Magnetic ferrites: design and synthesis. In: Lyshevski SE (ed) Dekker encyclopedia of nanoscience and nanotechnology, 3rd edn. CRC Press, New York, pp 2176–2189
Mindru I, Gingasu D, Patron L, Marinescu G, Calderon-Moreno JM, Diamandescu L, Preda S, Oprea O (2015) Chromium substituted copper ferrites via gluconate precursor route. Ceram Int 41:5318–5330. https://doi.org/10.1016/j.ceramint.2014.12.081
Nakamoto K (1986) Infrared and Raman spectra of inorganic and coordination compounds, 4th edn. Wiley, New York
Nogueira IM, Sabadia GQ, Moreira AA, Filho JM, Oliveira AC (2011) Investigation of the deactivation of iron nanocomposites by coking in the dehydrogenation of ethylbenzene. J Mol Catal A Chem 351:81–92. https://doi.org/10.1016/j.molcata.2011.09.020
Olar R, Badea M, Diamandescu L, Cristurean E, Brezeanu M (2004) Soft chemical synthesis and characterisation of some substituted ferrites. J Alloys Compd 363:262–267. https://doi.org/10.1016/S0925-8388(03)00452-3
Pervaiz E, Gul IH (2012) Enhancement of electrical properties due to Cr3+ substitution in Co-ferrite nanoparticles synthesized by two chemical techniques. J Magn Magn Mater 324:3695–3703. https://doi.org/10.1016/j.jmmm.2012.05.050
Petit GA, Forester DW (1971) Mössbauer study of cobalt zinc ferrites. Phys Rev B 4:3912–3926. https://doi.org/10.1103/PhysRevB.4.3912
Petit S, Righi D, Madejova J, Decarreau A (1999) Interpretation of the infrared NH4 + spectrum of the NH4 +-clays: application to the evaluation of the layer charge. Clay Miner 34:543–549. https://doi.org/10.1180/000985599546433
Qwabe LQ, Friedrich HB, Singh S (2015) Preferential oxidation of CO in a hydrogen rich feed stream using Co–Fe mixed metal oxide catalysts prepared from hydrotalcite precursors. J Mol Catal A Chem 404-405:167–177. https://doi.org/10.1016/j.molcata.2015.04.020
Raghasudha M, Ravinder D, Veerasomaiah P (2016) Investigation of superparamagnetism in pure and chromium substituted cobalt nanoferrite. J Magn Magn Mater 420:45–50. https://doi.org/10.1016/j.jmmm.2016.06.090
Rao GSN, Rao BP, Hamdeh HH (2014) Mössbauer spectroscopic study of high magnetostrictive cobalt chromium ferrites for automobile torque sensors. Procedia Mater Sci 6:1511–1515. https://doi.org/10.1016/j.mspro.2014.07.131
Royer S, Alamdari H, Duprez D, Kaliaguine S (2005) Oxygen storage capacity of La1−xA′xBO3 perovskites (with A′ = Sr, Ce; B = Co, Mn)—relation with catalytic activity in the CH4 oxidation reaction. Appl Catal B 58:273–288. https://doi.org/10.1016/j.apcatb.2004.12.010
Sastri MVC, Viswanath RP, Visvanathan B (1982) Studies on the reduction of iron oxide with hydrogen. Int J Hydrog Energy 7:951–955. https://doi.org/10.1016/0360-3199(82)90163-X
Sawatzky GA, Van Der Woude F, Morrish AH (1969) Mössbauer study of several ferrimagnetic spinels. Phys Rev 187:747–757. https://doi.org/10.1103/PhysRev.187.747
Sharma S, Choudhary N, Verma MK, Sharma ND, Singh D (2017) Cation distribution and magnetic properties of nano and bulk CoCrFeO4 ferrite synthesized by glycine-nitrate combustion method. Ceram Int 43:11083–11089. https://doi.org/10.1016/j.ceramint.2017.05.154
Sijo AK (2017) Magnetic and structural properties of CoCrxFe2−xO4 spinels prepared by solution self combustion method. Ceram Int 43:2288–2290. https://doi.org/10.1016/j.ceramint.2016.11.010
Sing KSW, Everett DH, Haul RAW, Moscou L, Pierotti RA, Rouquerol J, Siemieniewska T (1985) Reporting physisorption data for gas/solid systems with special reference to the determination of surface area and porosity (IUPAC recommendations 1984). Pure Appl Chem 57:603–619. https://doi.org/10.1351/pac198557040603
Tasca JE, Quincoces CE, Lavat A, Alvarez AM, Gonzalez MG (2011) Preparation and characterization of CuFe2O4 bulk catalysts. Ceram Int 37:803–812. https://doi.org/10.1016/j.ceramint.2010.10.023
Toksha BG, Shirsath SE, Mane ML, Patange SM, Jadhav SS, Jadhav KM (2011) Autocombustion high-temperature synthesis, structural, and magnetic properties of CoCrxFe2−xO4 (0 ≤ x ≤ 1.0). J Phys Chem C 115:20905–20912. https://doi.org/10.1021/jp205572m
Vadivel M, Babu RR, Sethuraman K, Ramamurthi K, Arivanandhan M (2014) Synthesis, structural, dielectric, magnetic and optical properties of Cr substituted CoFe2O4 nanoparticles by co-precipitation method. J Magn Magn Mater 362:122–129. https://doi.org/10.1016/j.jmmm.2014.03.016
Zasada F, Janas J, Piskorz W, Gorczyńska M, Sojka Z (2017) Total oxidation of lean methane over cobalt spinel nanocubes controlled by the self-adjusted redox state of the catalyst: experimental and theoretical account for interplay between the Langmuir–Hinshelwood and Mars–Van Krevelen mechanisms. ACS Catal 7:853–2867. https://doi.org/10.1021/acscatal.6b03139
Zhang W, Zuo X, Zhang D, Wu C, Silva SRP (2016) Cr3+ substituted spinel ferrite nanoparticles with high coercivity. Nanotechnology 27:245707. https://doi.org/10.1088/0957-4484/27/24/245707
Acknowledgments
Support of the EU (ERDF) and Romanian Government, allowing for the acquisition of the research infrastructure under POS-CCE O 2.2.1 project INFRANANOCHEM-Nr. 19/01.03.2009, is gratefully acknowledged. The work also benefits from the support of the “Materials Science and Advanced Characterization Methods” Programme of the “Ilie Murgulescu” Institute of Physical Chemistry, financed by the Romanian Academy. One of the authors (LD) acknowledges the financial support under the Core Program PN 10N/2017.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mindru, I., Gingasu, D., Diamandescu, L. et al. CoFe2−xCrxO4 ferrites: synthesis, characterization and their catalytic activity. Chem. Pap. 72, 3203–3213 (2018). https://doi.org/10.1007/s11696-018-0553-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-018-0553-0