Abstract
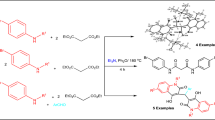
Reaction of equimolar amounts of 2,4(1H,3H)-quinolinediones and diethyl acetylenedicarboxylate in absolute ethanol, containing catalytic triethylamine, gave ethyl 5,6-dihydro-2,5-dioxo-6,9-disubstituted-2H-pyrano[3,2-c]quinoline-4-carboxylates in good yields. In a different manner, reaction of two equivalents of dialkyl acetylenedicarboxylates with one equivalent of 2,4(1H,3H)-quinolinediones afforded dialkyl 2(4-oxo-1,4-dihydroquinolin-3-yl)fumarates in good yields. The structures of the products were elucidated by 1H NMR, 13C NMR, two-dimensional NMR, IR, mass spectra and elemental analyses.
Graphical Abstract






Similar content being viewed by others
References
Aly AA, El-Shaieb KM (2007) Reaction of N-imidoylthioureas with dimethyl acetylenedicarboxylate: synthesis of new 1,3,5-thiadiazepines. J Chem Res 2007:563–565. doi:10.3184/030823407X255579
Aly AA, Hopf H, Ernst L, Dix I, Jones PG (2006) New cycloadditions of (E)-N,α-dimethyl-α-(4-[2.2]para-cylophanyl)-nitrone. Eur J Org Chem 2006:3001–3006. doi:10.1002/ejoc.200500745
Aly AA, Ahmed EK, El-Mokadam KM (2007) Reactions of aroylthioureas with acetylenic esters and dibenzoyl ethylene. Selectivity towards the formation of new 1,3-thiazines. J Heterocycl Chem 44:1431–1438
Aly AA, Brown AB, Hasaan AA, El-Shaieb KM, Bedair TMI (2009) Carbamothioates in the synthesis of diaroyl sulfides; selectivity of diaroyl sulfides and their addition to acetylenic and ethylenic deficient compounds. Arkivoc viii:66–77
Aly AA, Brown AB, Radwan MF, Gamal-Eldeen AM, Abdel-Aziz M, Abuo-Rahma GA, Ramadan M (2010) Thieno[2,3-d]pyrimidines in the synthesis of antitumor and antioxidant agents. Arch Pharm 343:301–309
Aly AA, Brown AB, Hassan AA (2014) Heterocycles from the reaction of thione groups with acetylenic bonds. Adv Heterocycl Chem 113:245–304
Anary-Abbasinejad M, Anaraki-Ardakani H, Ezadi A, Hassanabadi A (2007) Three-component reaction between triphenylphosphine, acetylenic esters and 4-hydroxycoumarin, 4-(phenylamino)coumarin, 4-hydroxyquinolin-2(1H)-one or 4-hydroxy-1-methylquinolin-2(1H)-one. J Chem Res 2007:605–608. doi:10.3184/030823407X256154
Asghari S, Ramezani S (2014) An efficient three-component synthesis of pyranoquinoline derivatives. J Heterocycl Chem 51:233–236
Ayafor JF, Sondengam BL, Ngadjui BT (1982) Veprisinium salt, a novel antibacterial quaternary alkaloid from Vepris louisii. Planta Med 44:139–142
Bandaru S, Majji RK, Bassa S, Chilla PN, Yellapragada R, Vasamsetty S, Jeldi RK, Korupolu RB, Sanasi PD (2016) Magnetic Nano cobalt ferrite catalyzed synthesis of 4H-pyrano[3,2-h]quinoline derivatives under microwave irradiation. Green Sust Chem 6:101–109
Barriga S, Fuertes P, Marcos CF, Torroba T (2004) Synthesis of highly branched sulfur–nitrogen heterocycles by cascade cycloadditions of [1,2]dithiolo[1,4]thiazines and [1,2]dithiolopyrroles. J Org Chem 69:3672
Buckle DR, Cantello BCC, Smith H, Spicer BA (1975) 4-Hydroxy-3-nitro-2-quinolones and related compounds as inhibitors of allergic reactions. J Med Chem 18:726–732
Cappelli A, Mohr GP, Gallelli A, Rizzo M, Anzini M, Vomero S, Mennuni L, Ferrari F, Makovec F, Menziani MC, De Benedetti PG, Giorgi G (2004) Design, synthesis, structural studies, biological evaluation, and computational simulations of novel potent AT1 angiotensin II receptor antagonists based on the 4-phenylquinoline structure. J Med Chem 47:2574–2586
Chen Y-L, Chung C-H, Chen I-L, Chen P-H, Jeng H-Y (2002) Synthesis and cytotoxic activity evaluation of indolo-, pyrrolo-, and benzofuro-quinolin-2(1H)-ones and 6-anilinoindoloquinoline derivatives. Bioorg Med Chem 10:2705–2712
Ellis D, Kuhen KL, Anaclerio B, Wu B, Wolff K, Yin H, Bursulaya B, Caldwell J, Karanewsky D, He Y (2006) Design, synthesis, and biological evaluations of novel quinolones as HIV-1 non-nucleoside reverse transcriptase inhibitors. Bioorg Med Chem Lett 16:4246–4251
Faber K, Stueckler H, Kappe T (1984) Non-steroidal antiinflammatory agents. 1. Synthesis of 4-hydroxy-2-oxo-1,2-dihydroquinolin-3-yl alkanoic acids by the wittig reaction of quinisatines. J Heterocycl Chem 21:1177–1181
Freeman GA, Andrews CW, Hopkins AL, Lowell GS, Schaller LT, Cowan JR, Gonzales SS, Koszalka GW, Hazen RJ, Boone LR, Ferris RG, Creech KL, Roberts GB, Short SA, Weaver K, Reynolds DJ, Milton J, Ren J, Stuart DI, Stammers DK, Chan JH (2004) Design of non-nucleoside inhibitors of HIV-1 reverse transcriptase with improved drug resistance properties. 2. J Med 47:5923–5936
Grundon MF (1998) Alkaloids, vol 32. Academic Press, London, pp 341–439
Hassanin HM, Ibrahim MA, Gabr YA, Alnamer YA (2012) Synthesis and chemical reactivity of pyrano[3,2-c]quinolones. J Heterocycl Chem 49:1269–1289
Hopkins AL, Ren J, Milton J, Hazen RJ, Chan JH, Stuart DI, Stammers DK (2004) Design of non-nucleoside inhibitors of HIV-1 reverse transcriptase with improved drug resistance properties. J Med Chem 47:5912–5922
Kalita PK, Baruah B, Bhuyan PJ (2006) Synthesis of novel pyrano[2,3-b]quinolines from simple acetanilides via intramolecular 1,3-dipolar cycloaddition. Tetrahedron Lett 47:7779–7782
McCormick JL, McKee TC, Cardinella JH, Boyd MR (1996) HIV inhibitory natural products. 26. Quinoline alkaloids from Euodia roxburghiana. J Nat Prod 59:469–471
Mederski WWKR, Osswald M, Dorsch D, Christadler M, Schmitges C-J, Wilm C (1997) 1,4-Diaryl 2-oxo-1,2-dihydro-quinoline-3-carboxylic acids as endothelin receptor antagonists. Bioorg Med Chem Lett 7:1883–1886
Michael JP (1998) Quinoline, quinazoline and acridone alkaloids. Nat Prod Rep 15:595–606
Mohtat B, Nahavandian S, Azar ZN, Djahaniani HZ (2011) Triphenylphosphine-promoted C-vinylation of 4-hydroxyquinolines. Naturforsch 66B:700–704
Moshapo PT, Sokamisa M, Mmutlane EM, Mampa RM, Kinfe HH (2016) A convenient domino Ferrier rearrangement-intramolecular cyclization for the synthesis of novel benzopyran-fused pyranoquinolines. Org Biomol Chem 14:5627–5638
Müller TJJ (2014) In Multicomponent reactions 2. Reaction involving an β-unsaturated carbonyl compound as electrophilic component, cycloadditions, and Born-, Silicon-, Free radical-, and Metal-Reactions. Georg-Thieme, Verlag
Nair V, Vinod AU, Ramesh R, Menon RS, Varma L, Mathew S, Chiaroni A (2002) An efficient multicomponent reaction involving the interception of the zwitterionic intermediate between DMAD and isocyanides with some active methylene compounds. Heterocycles 58:147–151
Nair V, Devipriya S, Suresh E (2008) A novel three-component reaction involving quinolone, dimethyl acetylenedicarboxylate, and C-H acids leading to the formation of pyrroloquinoline derivatives. Synthesis 2008:1065–1068. doi:10.1055/s-2008-1032128
Nicolaides DN, Litinas KE, Psaroulis I, Makri A, Adamopoulos S (2011) Reactions between 4-hydroxycoumarins or 4-hydroxy-1-methylquinoline and DMAD in the presence of phosphites. Synthesis of (chromen-3-yl)- and (quinolin-3-yl)-(dialkoxyphosphoryl)succinates. Phosp Sulfur Silicon Relat Elem 186:2104–2111
Rad-Moghadam K, Valadi AK, Alipour A (2014) LiBF4 integrated into [BMIm]BF4: an ionic-liquid metal composite and homogeneous catalyst for efficient synthesis of pyran-annulated heterocycles. Appl Organomet Chem 28:146–150
Ramesh M, Mohan PS, Shanmugam P (1984) A convenient synthesis of flindersine, atanine and their analogues. Tetrahedron 40:4041–4049
Rao VS, Darbarwar M (1988) One pot synthesis of 7-[1, 2-dihydro-4-hydroxy-1-methyl/phenyl-2-OXO-3-Quinolinyl]-5, 7-dihydro-5-methyl/phenyl-6H-[1]-benzo pyrano [3, 2-c] quinolin-6-ones. Synth Commun 18:2267–2272
Rechac VL, Cirujano FG, Corma A, Llabrés XFX (2016) Diastereoselective synthesis of pyranoquinolines on zirconium-containing UiO-66 metal–organic frameworks. Eur J Inorg Chem 2016:4512–4516. doi:10.1002/ejic.201600372
Sekar M, Rajendra PKJ (1988) Quinoline alkaloids: synthesis of pyrano[2,3-b]quinolines, haplofoline, lunacrine, and demethoxylunacrine. J Nat Prod 61:294–296
Steiner MK, Perston IR (2008) Optimizing endothelin receptor antagonist use in the management of pulmonary arterial hypertension. Vasc Health Risk Manag 4(5):943–952
Tzeng CC, Lee KH, Wang TC, Han CH, Chen YL (2000) Pharm Res 17:715–719
Yadav JS, Reddy BVS, Rao RS, Kumar SK, Kunwar AC (2002) InCl3-Catalyzed hetero-Diels–Alder reaction: an expeditious synthesis of pyranoquinolines. Tetrahedron 58:7891–7896
Acknowledgements
The NMR spectrometer at Florida Institute of Technology was purchased with assistance from the National Science Foundation (CHE 03-42251), USA.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
El-Sheref, E.M., Aly, A.A., Mourad, AF.E. et al. Synthesis of pyrano[3,2-c]quinoline-4-carboxylates and 2-(4-oxo-1,4-dihydroquinolin-3-yl)fumarates. Chem. Pap. 72, 181–190 (2018). https://doi.org/10.1007/s11696-017-0269-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11696-017-0269-6