Abstract
Background
Obesity is a major risk factor for transplant. Laparoscopic bariatric surgery (LBS) offers transplant patient benefits including improved comorbidities, graft function, and longevity. We completed a scoping review and analyzed the Metabolic and Bariatric Surgery Accreditation and Quality Improvement Program (MBSAQIP) to determine the risk-benefit profile of LBS after transplant. We also compared laparoscopic sleeve gastrectomy (LSG) to laparoscopic Roux-en-Y gastric bypass (LRYGB) following transplant.
Methods
Univariate analysis determined between group differences with subgroup analysis comparing LSG versus LRYGB in transplant patients. Multivariable analysis assessed whether prior transplant was independently associated with major complications or mortality.
Results
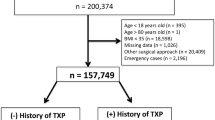
A total of 469 (0.1%) patients had previous transplant and had more comorbidities and more often underwent LSG. Operative time (93.9 min vs 83 min, p < 0.001) and length of stay were longer. Major complications were threefold higher in patients with a transplant history (9.6% vs 3.2%; p < 0.001. Previous transplant was the second greatest independent predictor for major complication (OR 2.14 [1.54–2.98], p = < 0.001) but was not predictive of death (OR 1.06 [0.14–8.13] p = 0.956).
Amongst transplant patients, LRYGB demonstrated higher rates of leak (n = 1), VTE, AKI, unplanned intubation, and readmission.
Conclusions
The 30-day complication rate from LBS is three times higher amongst patients with a transplant. LSG is likely the best surgical approach. Despite risks, post-transplant patients incur important benefits from LBS. Surgeons must be aware of this risk-benefit profile when determining LBS candidacy.
Similar content being viewed by others
References
Patel YA, Berg CL, Moylan CA. Nonalcoholic fatty liver disease: key considerations before and after liver transplantation. Dig Dis Sci. 2016;61(5):1406–16.
Lazzati A, Iannelli A, Schneck AS, et al. Bariatric surgery and liver transplantation: a systematic review a new frontier for bariatric surgery. Obes Surg. 2015 Jan;25(1):134–42.
Suraweera D, Saab EG, Choi G, et al. Bariatric surgery and liver transplantation. Gastroenterol Hepatol. 2017;13(3):170–5.
Tichansky DS, Madan AK. Laparoscopic Roux-en-Y gastric bypass is safe and feasible after orthotopic liver transplantation. Obes Surg. 2005;15(10):1481–6.
Hadjievangelou N, Kulendran M, McGlone ER, et al. Is bariatric surgery in patients following renal transplantation safe and effective? A best evidence topic. Int J Surg. 2016;28:191–5.
Modanlou KA, Muthyala U, Xiao H, et al. Bariatric surgery among kidney transplant candidates and recipients: analysis of the United States renal data system and literature review. Transplantation. 2009;87(8):1167–73.
Lin MY, Tavakol MM, Sarin A, et al. Safety and feasibility of sleeve gastrectomy in morbidly obese patients following liver transplantation. Surg Endosc. 2013;27(1):81–5.
Tsamalaidze L, Elli EF. Bariatric surgery is gaining ground as treatment of obesity after heart transplantation: report of two cases. Obes Surg. 2017;27(11):3064–7.
Yemini R, Nesher E, Winkler J, et al. Bariatric surgery in solid organ transplant patients: long-term follow-up results of outcome, safety, and effect on immunosuppression. Am J Transplant Off J Am Soc Transplant Am Soc Transplant Surg. 2018;18(11):2772–80.
Duchini A, Brunson ME. Roux-en-Y gastric bypass for recurrent nonalcoholic steatohepatitis in liver transplant recipients with morbid obesity. Transplantation. 2001;72(1):156–9.
Golomb I, Winkler J, Ben-Yakov A, et al. Laparoscopic sleeve gastrectomy as a weight reduction strategy in obese patients after kidney transplantation. Am J Transplant. 2014;14(10):2384–90.
Alexander JW, Goodman H. Gastric bypass in chronic renal failure and renal transplant. Nutr Clin Pract. 2007;22(1):16–21.
Al-Sabah S, Christou NV. Laparoscopic gastric bypass after cardiac transplantation. Surg Obes Relat Dis. 2008;4(5):668–70.
Velez JP, Arias RH, Olaya P. Laparoscopic sleeve gastrectomy on heart transplant recipient with body mass index of 34 kg/m2 and metabolic syndrome. Surg Obes Relat Dis. 2012;8(1):e6–7.
Salinas A, Mc Quattie Pimentel AC, Salinas HM. Roux-en-Y gastric bypass as metabolic treatment in a heart transplant patient with type 2 diabetes: 4 years later. Surg Obes Relat Dis. 2014;10(6):e81–3.
Khullar V, Dolganiuc A, Firpi RJ. Pre-and-post transplant considerations in patients with nonalcoholic fatty liver disease. World J Transplant. 2014;4(2):81–92.
Guckelberger O, Mutzke F, Glanemann M, et al. Validation of cardiovascular risk scores in a liver transplant population. Liver Transplant. 2006;12(3):394–401.
Briggs JD. Causes of death after renal transplantation. Nephrol Dial Transplant. 2001;16(8):1545–9.
Gill JS. Cardiovascular disease in transplant recipients: current and future treatment strategies. Clin J Am Soc Nephrol. 2008;3(Supplement 2):S29.
Berthoux F, Mariat C. Cardiovascular death after renal transplantation remains the first cause despite significant quantitative and qualitative changes. Transplantation. 2010;89(7):806.
Watt KD, Pedersen RA, Kremers WK, et al. Evolution of causes and risk factors for mortality post-liver transplant: results of the NIDDK long-term follow-up study. Am J Transplant Off J Am Soc Transplant Am Soc Transplant Surg. 2010;10(6):1420–7.
American College of Surgeons. MBSAQIP Participant Use Data File (PUF). American College of Surgeons; 2019 [cited 2019 September 14]; Available from: https://www.facs.org/quality-programs/mbsaqip/participant-use.
Beckmann S, Nikolic N, Denhaerynck K, Binet I, Koller M, Boely E, et al. Evolution of body weight parameters up to 3 years after solid organ transplantation: the prospective Swiss Transplant Cohort Study. Clin Transplant. 2017;31(3).
Andalib A, Aminian A, Khorgami Z, et al. Early postoperative outcomes of primary bariatric surgery in patients on chronic steroid or immunosuppressive therapy. Obes Surg. 2016;26(7):1479–86.
Hefler J, Dang J, Modasi A, Switzer N, Birch DW, Karmali S. Effects of chronic corticosteroid and immunosuppressant use in patients undergoing bariatric surgery. Obes Surg. 2019;1–7.
Lee Y, Doumouras AG, Yu J, et al. Complete resolution of nonalcoholic fatty liver disease after bariatric surgery: a systematic review and meta-analysis. Clin Gastroenterol Hepatol. 2019;17(6):1040–60.
Verdonk RC, Buis CI, Porte RJ, et al. Anastomotic biliary strictures after liver transplantation: causes and consequences. Liver Transplant. 2006;12(5):726–35.
Wickman C, Kramer H. Obesity and kidney disease: potential mechanisms. Semin Nephrol. 2013;33(1):14–22.
Chagnac A, Weinstein T, Herman M, et al. The effects of weight loss on renal function in patients with severe obesity. J Am Soc Nephrol. 2003;14(6):1480–6.
Ashrafian H, le Roux Carel W, Darzi A, et al. Effects of bariatric surgery on cardiovascular function. Circulation. 2008;118(20):2091–102.
Kindel TL, Strande JL. Bariatric surgery as a treatment for heart failure: review of the literature and potential mechanisms. Surg Obes Relat Dis. 2018;14(1):117–22.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethics Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. For this type of study, formal consent is not required. Informed consent does not apply.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix
Appendix
Data collection definitions
Leak—defined by any of the following: reoperation for anastomotic/staple line leak, readmission for anastomotic/staple line leak, reintervention for anastomotic/staple line leak, drain present 30 days postoperatively, or death caused by anastomotic/staple line leak.
Postoperative VTE—defined by any of the following: reoperation for pulmonary embolism or venous thrombosis, readmission for pulmonary embolism or venous thrombosis, reintervention for pulmonary embolism or venous thrombosis, venous thrombosis requiring anticoagulation therapy, pulmonary embolism requiring anticoagulation therapy, and does not include anticoagulation for presumed/suspected VTE.
Postoperative bleed—defined by any of the following: reoperation for bleed, readmission for bleed, reintervention for bleed, transfusion required in first 72 h of surgery start time, and death caused by bleeding.
Major complication—defined by any of the following: cardiac complications, pneumonia, acute renal failure, reoperation, reintervention, venous thromboembolism, deep surgical site infection, wound disruption, sepsis, unplanned intubation, leak, bleed, coma > 24 h, and cerebral vascular accident.
Rights and permissions
About this article
Cite this article
Verhoeff, K., Dang, J.T., Modasi, A. et al. Bariatric Surgery Outcomes in Patients with Previous Organ Transplant: Scoping Review and Analysis of the MBSAQIP. OBES SURG 31, 508–516 (2021). https://doi.org/10.1007/s11695-020-05042-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-020-05042-w