Abstract
Purposes
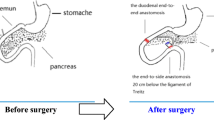
Biliopancreatic diversion could improve type 2 diabetes mellitus. Our aim was to investigate the effects of biliary jejunostomy on the improvement of glucose.
Materials and Methods
Twenty-seven type 2 diabetes patients underwent biliary jejunostomy between January 2013 and January 2018 in our hospital and were followed up. Demographic data, operation details, body weight, food intake, effects on diabetes control, and biomedical parameters were collected and analyzed.
Results
As defined previously, 3 of 27 diabetes patients were “under control,” 8 patients were “in remission,” and 12 patients were “improved.” The fasting glucose decreased from 9.7 ± 2.1 mmol/L before surgery to 7.9 ± 1.8 mmol/L 12 months after surgery (P = 0.001). The level of hemoglobin A1c in these patients was 9.1 ± 2.3% before surgery, and it decreased to 7.2 ± 1.3% 12 months after surgery (P < 0.001). There was no significant difference in the body weight index (P = 0.78) or food intake (P = 0.18) between the time prior to surgery and 12 months afterward. The average level of total bile acids increased significantly after surgery, from 6.7 ± 2.2 μmol/L before surgery to 8.6 ± 2.9 μmol/L 12 months after surgery (P < 0.001).
Conclusions
Fasting glucose in type 2 diabetes patients was improved after biliary jejunostomy. Increasing bile acids levels might play an important role in the remission of type 2 diabetes.



Similar content being viewed by others
References
American DA. Economic costs of diabetes in the U.S. in 2017. Diabetes Care. 2018;41(5):917–28.
Kodama S, Fujihara K, Horikawa C, et al. Network meta-analysis of the relative efficacy of bariatric surgeries for diabetes remission. Obes Rev : Off J Int Assoc Study Obes. 2018;30
Hage MP, Safadi B, Salti I, et al. Role of gut-related peptides and other hormones in the amelioration of type 2 diabetes after roux-en-Y gastric bypass surgery. ISRN endocrinology. 2012;2012:504756.
Flynn CR, Albaugh VL, Cai S, et al. Bile diversion to the distal small intestine has comparable metabolic benefits to bariatric surgery. Nat Commun. 2015;6:7715.
Cipriani S, Mencarelli A, Palladino G, et al. FXR activation reverses insulin resistance and lipid abnormalities and protects against liver steatosis in Zucker (fa/fa) obese rats. J Lipid Res. 2010;51(4):771–84.
Ma K, Saha PK, Chan L, Moore DD. Farnesoid X receptor is essential for normal glucose homeostasis. J Clin Invest 2006;116(4):1102–1109. Pubmed Central PMCID: 1409738.
Potthoff MJ, Boney-Montoya J, Choi M, et al. FGF15/19 regulates hepatic glucose metabolism by inhibiting the CREB-PGC-1alpha pathway. Cell Metab. 2011;13(6):729–38.
Diabetes Prevention Program Research G. The diabetes prevention program (DPP): description of lifestyle intervention. Diabetes Care 2002;25(12):2165–2171. Pubmed Central PMCID: PMC1282458. Epub 2002/11/28.
Chen W, Yan Z, Liu S, et al. The better effect of roux-en-Y gastrointestinal reconstruction on blood glucose of nonobese type 2 diabetes mellitus patients. Am J Surg. 2014;207(6):877–81.
Eh K, McGill M, Wong J, et al. Cultural issues and other factors that affect self-management of type 2 diabetes mellitus (T2D) by Chinese immigrants in Australia. Diabetes Res Clin Pract. 2016;119:97–105. Epub 2016/08/09
Swann JR, Want EJ, Geier FM, Spagou K, Wilson ID, Sidaway JE, et al. Systemic gut microbial modulation of bile acid metabolism in host tissue compartments. Proc Natl Acad Sci U S A 2011;108 Suppl 1:4523–4530. Pubmed Central PMCID: 3063584.
Sinal CJ, Tohkin M, Miyata M, et al. Targeted disruption of the nuclear receptor FXR/BAR impairs bile acid and lipid homeostasis. Cell. 2000;102(6):731–44.
Thomas C, Gioiello A, Noriega L, et al. TGR5-mediated bile acid sensing controls glucose homeostasis. Cell Metab. 2009;10(3):167–77.
Prawitt J, Abdelkarim M, Stroeve JH, et al. Farnesoid X receptor deficiency improves glucose homeostasis in mouse models of obesity. Diabetes. 2011;60(7):1861–71.
Watanabe M, Houten SM, Mataki C, et al. Bile acids induce energy expenditure by promoting intracellular thyroid hormone activation. Nature. 2006;439(7075):484–9.
Staley C, Weingarden AR, Khoruts A, et al. Interaction of gut microbiota with bile acid metabolism and its influence on disease states. Appl Microbiol Biotechnol. 2017;101(1):47–64.
Tremaroli V, Backhed F. Functional interactions between the gut microbiota and host metabolism. Nature. 2012;489(7415):242–9.
Shao Y, Ding R, Xu B, et al. Alterations of gut microbiota after roux-en-Y gastric bypass and sleeve gastrectomy in Sprague-Dawley rats. Obes Surg. 2017;27(2):295–302.
Guo Y, Liu CQ, Shan CX, Chen Y, Li HH, Huang ZP, et al. Gut microbiota after Roux-en-Y gastric bypass and sleeve gastrectomy in a diabetic rat model: increased diversity and associations of discriminant genera with metabolic changes. Diabetes/metabolism research and reviews. 2017 Mar;33(3).
Murphy R, Tsai P, Jullig M, et al. Differential changes in gut microbiota after gastric bypass and sleeve gastrectomy bariatric surgery vary according to diabetes remission. Obes Surg. 2017;27(4):917–25.
Hutchison RL, Hutchison AL. Cesar roux and his original 1893 paper. Obes Surg. 2010;20(7):953–6.
Pories WJ, Albrecht RJ. Etiology of type II diabetes mellitus: role of the foregut. World J Surg. 2001;25(4):527–31.
Peterli R, Steinert RE, Woelnerhanssen B, Peters T, Christoffel-Courtin C, Gass M, Kern B, von Fluee M, Beglinger C Metabolic and hormonal changes after laparoscopic roux-en-Y gastric bypass and sleeve gastrectomy: a randomized, prospective trial. Obes Surg 2012;22(5):740–748. Pubmed Central PMCID: PMC3319900. Epub 2012/02/23.
Drucker DJ, Nauck MA. The incretin system: glucagon-like peptide-1 receptor agonists and dipeptidyl peptidase-4 inhibitors in type 2 diabetes. Lancet. 2006;368(9548):1696–705.
Hussain A, Mahmood H, El-Hasani S. Can roux-en-Y gastric bypass provide a lifelong solution for diabetes mellitus? Can J Surg. 2009;52(6):E269–75.
Funding
This study was funded by the National Natural Science Foundation of China (grant number 81400797/H0713).
Author information
Authors and Affiliations
Contributions
Xiaodong He designed the study. Ning Zhang and Haixin Yin followed up patients and collected the data. Wei Liu analyzed the data, and Weijie Chen wrote the manuscript.
Corresponding author
Ethics declarations
The authors declare that they have no conflict(s) of interest. For this type of study, formal consent is not required.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, N., Chen, W., Yin, H. et al. Biliary Jejunostomy Might Improve Glucose in Type 2 Diabetes Patients. OBES SURG 30, 1446–1451 (2020). https://doi.org/10.1007/s11695-019-04319-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-019-04319-z