Abstract
Background
Roux-en-Y bariatric surgery has been shown to have a remarkable and sustainable improvement in type 2 diabetes. Recent clinical studies have shown that bariatric surgery can improve or halt the development of diabetic microvascular complications such as nephropathy. However, the exact underlying mechanisms of surgical procedures are unknown. Here, we have investigated the effects of Roux-en-Y esophagojejunostomy (RYEJ) on renal function and inflammation and fibrosis biomarkers for renal injury in type 2 diabetic rats.
Methods
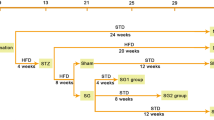
Sprague–Dawley rats with high fat diet and streptozotocin (STZ)-induced diabetes were randomly assigned into four groups: diabetic nephropathy (DN), DN treated with food restriction (DN-FR), DN treated with RYEJ surgery (DN-RYEJ), and DN-RYEJ sham (n = 6/group). Age-matched normal rats were assigned as control group. RYEJ and sham surgeries were performed. Hyperinsulinemic–euglycemic clamps with tracer infusion were completed to assess insulin sensitivity. Twenty-four hour urine albumin excretion rate (UAER) and glomerular filtration rate (GFR) were measured. The renal pathological injury was assessed by hematoxylin and eosin (HE) staining. Kidney messenger RNA (mRNA) and/or protein content/distribution of phospho-c-Jun NH2-terminal kinase (JNK), monocyte chemoattractant protein (MCP)-1, transforming growth factor (TGF)-β1, and mitogen-activated protein kinase phosphatase 5 (MKP5) were evaluated by real-time PCR and/or Western blotting/immunohistochemistry.
Results
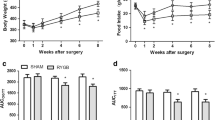
Roux-en-Y esophagojejunostomy improved insulin sensitivity. RYEJ ameliorated renal function by improving UAER and GFR and attenuated glomerular hypertrophy after surgery. RYEJ also significantly downregulated the levels of JNK-mediated inflammatory response and upregulated the level of the anti-inflammatory mediator MKP5.
Conclusion
Roux-en-Y esophagojejunostomy alleviates insulin resistance. RYEJ surgery ameliorated renal function and attenuated glomerular hypertrophy in a DN rat model. The considerable nephroprotective function may be mainly attributed to the reduced inflammatory and fibrotic biomarkers after RYEJ. The improvements in renal function and inflammation are not wholly dependent on the magnitude of weight loss.








Similar content being viewed by others
References
Postma MJ, de Zeeuw D. The economic benefits of preventing endstage renal disease in patients with type 2 diabetes mellitus. Nephrol Dial Transplant. 2009;24:2975–83.
Collins AJ, Foley RN, Chavers B, et al. United states renal data system 2011 annual data report: atlas of chronic kidney disease and end-stage renal disease in the united states. Am J Kidney Dis. 2012;59(1 Suppl 1):A7. e1–420.
Thomson SC, Vallon V, Blantz RC. Kidney function in early diabetes: the tubular hypothesis of glomerular filtration. Am J Physiol Renal Physiol. 2004;286(1):F8–15.
Leon CA, Raij L. Interaction of haemodynamic and metabolic pathways in the genesis of diabetic nephropathy. Hypertension. 2005;23(11):1931–7.
Caramori ML, Mauer M. Diabetes and nephropathy. Curr Opin Nephrol Hypertens. 2003;12(3):273–82.
Docherty NG, LeRoux CW, Jackson S. Bariatric surgery and microvascular complications of type 2 diabetes mellitus. Curr Atheroscler Rep. 2014;16:453.
Fenske WK, Dubb S, Bueter M, et al. Effect of bariatric surgery induced weight loss on renal and systemic inflammation and blood pressure: a 12-month prospective study. Surg Obes Relat Dis. 2013;9(4):559–68.
Miras AD, Chuah LL, Lascaratos G, et al. Bariatric surgery does not exacerbate and may be beneficial for the microvascular complications of type 2 diabetes. Diabetes Care. 2012;35:e81.
Ouchi N, Parker JL, Lugus JJ, et al. Adipokines in inflammation and metabolic disease. Nat Rev Immunol. 2011;11:85–97.
Yang F, Chung AC, Ru Huang X, et al. Angiotensin II induces connective tissue growth factor and collagen I expression via transforming growth factor-beta-dependent and -independent smad pathways: the role of Smad3. Hypertension. 2009;54:877–84.
Murphy M, Docherty NG, Griffin B, et al. IHG-1 amplifies TGF-beta1 signaling and is increased in renal fibrosis. Am Soc Nephrol. 2008;19:1672–80.
Moustakas A, Heldin CH. Dynamic control of TGF-beta signaling and its links to the cytoskeleton. FEBS Lett. 2008;582:2051–65.
Kyriakis JM, Avruch J. Mammalian mitogen-activated protein kinase signal transduction pathways activated by stress and inflammation. Physiol Rev. 2001;81:807–69.
Cohen MP, Shea E, Chen S, et al. Glycated albumin increases oxidative stress, activates NF-kB and extracellular signal-regulated kinase (ERK), and stimulates ERK-dependent transforming growth factor-B1 production in macrophage RAW cells. J Lab Clin Med. 2003;141:242–9.
Evans JL, Goldfine ID, Maddux BA, et al. Oxidative stress and stretch-activated signaling pathways: a unifying hypothesis of type 2 diabetes. Endocr Rev. 2002;23:599–622.
Theodosiou A, Smith A, Gillieron C, et al. MKP5, a new member of the MAP kinase phosphatase family, which selectively dephosphorylates stress-activated kinases. Oncogene. 1999;18:6981–8.
Zhang Y, Blattman JN, Kennedy NJ, et al. Regulation of innate and adaptive immune responses by MAP kinase phosphatase 5. Nature. 2004;430:793–7.
Ma FY et al. The role of stress-activated protein kinase signaling in renal pathophysiology. Braz J Med Biol Res. 2009;42(1):29–37.
Hofso D, Nordstrand N, Johnson LK, et al. Obesity-related cardiovascular risk factors after weight loss: a clinical trial comparing gastric bypass surgery and intensive lifestyle intervention. Eur J Endocrinol. 2010;163:735–45.
Navaneethan SD, Kelly KR, Sabbagh F, et al. Urinary albumin excretion, HMW adiponectin, and insulin sensitivity in type 2 diabetic patients undergoing bariatric surgery. Obes Surg. 2010;20:308–15.
Amor A, Jimenez A, Moize V, et al. Weight loss independently predicts urinary albumin excretion normalization in morbidly obese type 2 diabetic patients undergoing bariatric surgery. Surg Endosc. 2013;27:2046–51.
Agrawal V, Khan I, Rai B, et al. The effect of weight loss after bariatric surgery on albuminuria. Clin Nephrol. 2008;70:194–202.
He B, Yu C, Runyu D, et al. Roux-en-Y esophagojejunostomy reduces serum and aortic inflammatory biomarkers in type 2 diabetic rats. Obes Surg. 2014;24:916–26.
Srinivasan K, Viswanad B, Asrat L, et al. Combination of high-fat diet-fed and low-dose streptozotocin-treated rat: a model for type 2 diabetes and pharmacological screening. Pharmacol Res. 2005;52(4):313–20.
Bing H, Sheng Z, Zhang Wei L, et al. Salicylate prevents hepatic oxidative stress activation caused by short-term elevation of free fatty acids in vivo. Diabetes Res Clin Pract. 2010;89(2):150–6.
Matsuda M, DeFronzo RA. Insulin sensitivity indices obtained from oral glucose tolerance testing: comparison with the euglycemic insulin clamp. Diabetes Care. 1999;22(9):1462–70.
Dunn JP, Abumrad NN, Breitman I, et al. Hepatic and peripheral insulin sensitivity and diabetes remission at 1 month after Roux-en-Y gastric bypass surgery in patients randomized to omentectomy. Diabetes Care. 2012;35(1):137–42.
Mima A, Matsubara T, Arai H, et al. Angiotensin II-dependent Src and Smad1 signaling pathway is crucial for the development of diabetic nephropathy. Lab Investig. 2006;86:927–39.
Flatt PR, Day C, Bailey CJ. Review: bariatric surgery: to treat diabesity. Br J Diab Vasc Dis. 2009;9(3):103–7.
Buchwald H, Oien DM. Metabolic/bariatric surgery worldwide 2011. Obes Surg. 2013;23(4):427–36.
Lanzarini E, Csendes A, Lembach H, et al. Evolution of type 2diabetes mellitus in non morbid obese gastrectomized patients with Roux en-Y reconstruction: retrospective study. World J Surg. 2010;34:2098–102.
Lee W. Sang Hoon Ahn, Jue Hee Lee, et al. Comparative study of diabetes mellitus resolution according to reconstruction type after gastrectomy in gastric cancer patients with diabetes mellitus. Obes Surg. 2012;22:1238–43.
Wang KC, Huang KH, Lan YT, et al. Outcome after curative surgery for gastric cancer patients with type 2 diabetes. World J Surg. 2013.
Navaneethan SD, Yehnert H, Moustarah F, et al. Weight loss interventions in chronic kidney disease: a systematic review and meta-analysis. Clin J Am Soc Nephrol. 2009;4:1565–74.
Meijer RI, van Wagensveld BA, Siegert CE, et al. Bariatric surgery as a novel treatment for type 2 diabetes mellitus: a systematic review. Arch Surg. 2011;146:744–50.
Buchwald H, Oien DM. Metabolic/bariatric surgery worldwide 2008. Obes Surg. 2009;19:1605–11.
Rao SR. Inflammatory markers and bariatric surgery: a meta analysis. Inflamm Res. 2012;61:789–807.
He B, Piao D, Yu C, et al. Amelioration in hepatic insulin sensitivity by reduced hepatic lipid accumulation at short-term after Roux-en-Y gastric bypass surgery in type 2 diabetic rats. Obes Surg. 2013;23(12):2033–41.
Kraynak AR, Storer RD, Jensen RD, et al. Extentand persistence of streptozotocin induced DNA damage and cell proliferation in rat kidney as determined by in vivo alkaline elution and BrdUrd labeling assays. Toxicol Appl Pharmacol. 1995;135:279–86.
Fowler S, Kon V, Ma L, et al. Obesity-related focal and segmental glomerulosclerosis: normalization of proteinuria in an adolescent after bariatric surgery. Pediatr Nephrol. 2009;24(4):851–5.
Agnani S, Vachharajani VT, Gupta R, et al. Does treating obesity stabilize chronic kidney disease? BMC Nephrol. 2005;6:7.
Navaneethan SD, Yehnert H. Bariatric surgery and progression of chronic kidney disease. Surg Obes Relat Dis. 2009;5(6):662–5.
Navarro-Diaz M, Serra A, Romero R, et al. Effect of drastic weight loss after bariatric surgery on renal parameters in extremely obese patients: long-term follow up. JASN. 2006;17(12 Suppl 3):S213–7.
Chagnac A, Weinstein T, Herman M, et al. The effects of weight loss on renal function in patients with severe obesity. JASN. 2003;14(6):1480–6.
Alexander JW, Goodman HR, Hawver LR, et al. Improvement and stabilization of chronic kidney disease after gastric bypass. Surg Obes Relat Dis. 2009;5(2):237–41.
Afshinnia F, Wilt TJ, Duval S, et al. Weight loss and proteinuria: systematic review of clinical trials and comparative cohorts. Nephrol Dial Transplant. 2010;25(4):1173–83.
Izzedine H, Coupaye M, Reach I, et al. Gastric bypass and resolution of proteinuria in an obese diabetic patient. Diabet Med. 2005;22(12):1761–2.
Perez G, Devaud N, Escalona A, et al. Resolution of early stage diabetic nephropathy in an obese diabetic patient after gastric bypass. Obes Surg. 2006;16(10):1388–91.
Zhiqing W, Jing W, Haili X, et al. Renal function is ameliorated in a diabetic nephropathy rat model through a duodenal-jejunal bypass. Diabetes Res Clin Pract. 2014;103:26–34.
Hopkins PN. Molecular biology of atherosclerosis. Physiol Rev. 2013;93(3):1317–542.
Lee JY, Yu BP, Chung HY. Activation mechanisms of endothelial NF-kappaB, IKK, and MAP kinase by tertbutyl hydroperoxide. Free Radic Res. 2005;39(4):399–409.
Kim YW, West XZ, Byzova TV. Inflammation and oxidative stress in angiogenesis and vascular disease. Mol Med. 2013;91(3):323–8.
Farooq A, Zhou MM. Structure and regulation of MAPK phosphatases. Cell Signal. 2004;16:769–79.
Bueter M, Dubb SS, Gill A, et al. Renal cytokines improve early after bariatric surgery. Br J Surg. 2010;97:1838–44.
Monte SV, Caruana JA, Ghanim H, et al. Reduction in endotoxemia, oxidative and inflammatory stress, and insulin resistance after Roux-en-Y gastric bypass surgery in patients with morbid obesity and type 2 diabetes mellitus. Surgery. 2012;151:587–93.
He B, Liu L, Yu C, et al. Roux-en-Y gastric bypass reduces lipid overaccumulation in liver by upregulating hepatic autophagy in obese diabetic rats. Obes Surg. 2015;25:109–18.
Knop FK. Resolution of type 2 diabetes following gastric bypass surgery: involvement of gut-derived glucagon and glucagonotropic signalling? Diabetologia. 2009;52(11):2270–6.
Mingrone G, Castagneto-Gissey L. Mechanisms of early improvement/resolution of type 2 diabetes after bariatric surgery. Diabetes Metab. 2009;35(6 Pt 2):518–23.
Luk AO, So WY, Ma RC, et al. Metabolic syndrome predicts new onset of chronic kidney disease in 5,829 patients with type 2 diabetes: a 5-year prospective analysis of the Hong Kong diabetes registry. Diabetes Care. 2008;31(12):2357–61.
Rutkowski P, Klassen A, Sebekova K, et al. Renal disease in obesity: the need for greater attention. J Ren Nutr. 2006;16:216–23.
Acknowledgments
This study was supported by a Research Fund for the Doctoral Program of Higher Education of China (grant no. 20112104110014) and an Outstanding Scientific Fund of Shengjing Hospital (grant no. 201101).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
All contributing authors declare that they have no conflicts of interest.
Ethical Approval
My study is about animal experiment, and the following statement has been included: “All applicable national and international guidelines for the care and use of animals were followed.”
Informed Consent
This does not apply to my submission, the statement: “Does not apply” has been included.
Rights and permissions
About this article
Cite this article
Wang, C., He, B., Piao, D. et al. Roux-en-Y Esophagojejunostomy Ameliorates Renal Function Through Reduction of Renal Inflammatory and Fibrotic Markers in Diabetic Nephropathy. OBES SURG 26, 1402–1413 (2016). https://doi.org/10.1007/s11695-015-1947-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-015-1947-5