Abstract
Background
During illness, thyroid parameters undergo acute changes, which are known as non-thyroidal illness syndrome, the cause of which has not been elucidated. In vitro and in vivo data demonstrate that leptin regulates the expression of thyrotropin-releasing hormone (TRH)—mRNA in the paraventricular nucleus as well as the secretion of thyrotropin (TSH) in response to fasting in humans and animals. Moreover, in healthy adults, TSH and leptin have almost identical circadian rhythms. Our aim was to investigate the secretion of leptin and TSH, and their probable interaction, during the acute stress that is induced by surgery.
Methods
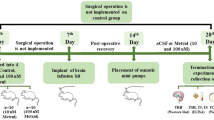
We studied 18 severely obese but otherwise healthy men. All participants were admitted to the hospital in the morning after an overnight fast. On the following day, 14 of the participants underwent bariatric surgery at 0900. The remaining four participants did not undergo surgery and served as controls. Serum samples to measure the levels of TSH and leptin were collected from all participants, as follows: upon admission to the hospital (baseline values) and on the following day at 0900 and every 10 min, thereafter for 9 h.
Results
The serum TSH increased during the first hour after skin incision (si) and then decreased gradually throughout the rest of the observation period. In contrast, during the first hour after si, the leptin levels remained unaltered. The leptin levels then decreased and reached a nadir at 4 h and 10 min post si after which they remained constant for approximately 1 h. Thereafter, while TSH continued to decrease, leptin started to increase and reached baseline values at 9 h post si. In control subjects, the TSH and leptin profiles seemed parallel each other.
Conclusions
During acute surgical stress, the secretion of TSH and leptin in severely obese men is asynchronous and causality could not be proven.



Similar content being viewed by others
References
Van den Berghe G, de Zegher F, Bouillon R. Clinical review 95: acute and prolonged critical illness as different neuroendocrine paradigms. J Clin Endocrinol Metab. 1998;83:1827–34.
Kaptein EM, Grieb DA, Spencer CA, et al. Thyroxine metabolism in the low thyroxine state of critical nonthyroidal illnesses. J Clin Endocrinol Metab. 1981;53:764–71.
Adler SM, Wartofsky L. The nonthyroidal illness syndrome. Endocrinol Metab Clin North Am. 2007;36:657–72.
Romijn JA, Wiersinga WM. Decreased nocturnal surge of thyrotropin in nonthyroidal illness. J Clin Endocrinol Metab. 1990;70:35–42.
Michalaki M, Vagenakis AG, Makri M, et al. Dissociation of the early decline in serum T(3) concentration and serum IL-6 rise and TNFalpha in nonthyroidal illness syndrome induced by abdominal surgery. J Clin Endocrinol Metab. 2001;86:4198–205.
Fekete C, Lechan RM. Negative feedback regulation of hypophysiotropic thyrotropin-releasing hormone (TRH) synthesizing neurons: role of neuronal afferents and type 2 deiodinase. Front Neuroendocrinol. 2007;28:97–114.
Fliers E, Alkemade A, Wiersinga WM, et al. Hypothalamic thyroid hormone feedback in health and disease. Prog Brain Res. 2006;153:189–207.
Blake NG, Eckland DJ, Foster OJ, et al. Inhibition of hypothalamic thyrotropin-releasing hormone messenger ribonucleic acid during food deprivation. Endocrinology. 1991;129:2714–8.
Arancibia S, Rage F, Astier H, et al. Neuroendocrine and autonomous mechanisms underlying thermoregulation in cold environment. Neuroendocrinology. 1996;64:257–67.
Boelen A, Wiersinga WM, Fliers E. Fasting-induced changes in the hypothalamus–pituitary–thyroid axis. Thyroid. 2008;18:123–9.
Mantzoros CS, Ozata M, Negrao A, et al. Synchronicity of frequently sampled thyrotropin (TSH) and leptin concentrations in healthy adults and leptin-deficient subjects: evidence for possible partial TSH regulation by leptin in humans. J Clin Endocrinol Metab. 2001;86:3284–91.
Bornstein SR. Is leptin a stress related peptide? Nat Med. 1997;3:937.
Ahima RS, Prabakaran D, Mantzoros C, et al. Role of leptin in the neuroendocrine response to fasting. Nature. 1996;382:250–2.
Licinio J, Mantzoros C, Negrao AB, et al. Human leptin levels are pulsatile and inversely related to pituitary–adrenal function. Nat Med. 1997;3:575–9.
Kain ZN, Zimolo Z, Heninger G. Leptin and the perioperative neuroendocrinological stress response. J Clin Endocrinol Metab. 1999;84:2438–42.
Harris M, Aschkenasi C, Elias CF, et al. Transcriptional regulation of the thyrotropin-releasing hormone gene by leptin and melanocortin signaling. J Clin Invest. 2001;107:111–20.
Nillni EA, Vaslet C, Harris M, et al. Leptin regulates prothyrotropin-releasing hormone biosynthesis. Evidence for direct and indirect pathways. J Biol Chem. 2000;275:36124–33.
Fekete C, Singru PS, Sanchez E, et al. Differential effects of central leptin, insulin, or glucose administration during fasting on the hypothalamic-pituitary-thyroid axis and feeding-related neurons in the arcuate nucleus. Endocrinology. 2006;147:520–9.
Morton GJ, Cummings DE, Baskin DG, et al. Central nervous system control of food intake and body weight. Nature. 2006;443:289–95.
Legradi G, Emerson CH, Ahima RS, et al. Leptin prevents fasting-induced suppression of prothyrotropin-releasing hormone messenger ribonucleic acid in neurons of the hypothalamic paraventricular nucleus. Endocrinology. 1997;138:2569–76.
Cho YM, Kim MS, Shin CS, et al. Dynamic change in plasma leptin level during the perioperative period. Horm Res. 2003;59:100–4.
Montalban C, Del Moral I, Garcia-Unzueta MT, et al. Perioperative response of leptin and the tumor necrosis factor alpha system in morbidly obese patients. Influence of cortisol inhibition by etomidate. Acta Anaesthesiol Scand. 2001;45:207–12.
Elimam A, Tjader I, Norgren S, et al. Total parenteral nutrition after surgery rapidly increases serum leptin levels. Eur J Endocrinol. 2001;144:123–8.
Boden G, Chen X, Mozzoli M, et al. Effect of fasting on serum leptin in normal human subjects. J Clin Endocrinol Metab. 1996;81:3419–23.
Considine RV. Regulation of leptin production. Rev Endocr Metab Disord. 2001;2:357–63.
Scriba D, Aprath-Husmann I, Blum WF, et al. Catecholamines suppress leptin release from in vitro differentiated subcutaneous human adipocytes in primary culture via beta1- and beta2-adrenergic receptors. Eur J Endocrinol. 2000;143:439–45.
Couillard C, Mauriege P, Prud'homme D, et al. Plasma leptin response to an epinephrine infusion in lean and obese women. Obes Res. 2002;10:6–13.
Fellander G, Nordenstrom J, Tjader I, et al. Lipolysis during abdominal surgery. J Clin Endocrinol Metab. 1994;78:150–5.
Faggioni R, Feingold KR, Grunfeld C. Leptin regulation of the immune response and the immunodeficiency of malnutrition. Faseb J. 2001;15:2565–71.
Kok P, Roelfsema F, Frolich M, et al. Spontaneous diurnal thyrotropin secretion is enhanced in proportion to circulating leptin in obese premenopausal women. J Clin Endocrinol Metab. 2005;90:6185–91.
Chan JL, Heist K, DePaoli AM, et al. The role of falling leptin levels in the neuroendocrine and metabolic adaptation to short-term starvation in healthy men. J Clin Invest. 2003;111:1409–21.
Schurgin S, Canavan B, Koutkia P, et al. Endocrine and metabolic effects of physiologic r-metHuLeptin administration during acute caloric deprivation in normal-weight women. J Clin Endocrinol Metab. 2004;89:5402–9.
Börner U, Klimek M, Schoengen H, et al. The influence of various anesthetics on the release and metabolism of thyroid hormones: results of two clinical studies. Anesth Analg. 1995;81:612–8.
Marana E, Scambia G, Colicci S, et al. Leptin and perioperative neuroendocrine stress response with two different anaesthetic techniques. Acta Anaesthesiol Scand. 2008;52:541–6.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Michalaki, M., Vagenakis, A.G., Argentou, M. et al. Dissociation of Thyrotropin and Leptin Secretion in Acute Surgical Stress in Severely Obese Patients. OBES SURG 19, 1424–1429 (2009). https://doi.org/10.1007/s11695-009-9852-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-009-9852-4