Abstract
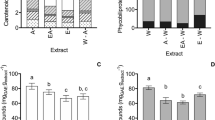
To the best of the author’s knowledge, so far, the utilization of industrial solid Ganoderma lucidum waste was not investigated as a potential source of remaining valuable bioactive compounds. The aim of our study was to chemically characterize the extract prepared with 96% and 70% ethanol utilizing G. lucidum waste from industrial ethanol (GE) and water (GW) extraction. The phenolic profile was assessed by UHPLC-MS/MS, while the amino acid content of selected extracts was done by GC-MS. The chemical composition of the extracts was obtained by ATP-FTIR analysis, and the peak at 1637 cm− 1 of spectra originating from C = O stretching vibration of amide I, was exclusively detected in samples prepared with 70% ethanol. Among eleven amino acids detected in G. lucidum residues, the most abundant was essential amino acid phenylalanine. Phenolic profile revealed p-hydroxybenzoic acid and chlorogenic acid as dominant in all the samples analyzed. The cytotoxic activity against two human cancer cell lines (colon carcinoma (HCT116) and melanoma (Hs294T)), photoprotective activity, and antioxidant activity of G. lucidum residues ethanol extracts (70% and 96%) were examined. The most prominent cytotoxic effect on HCT116 and Hs294T cells was attributed to extract prepared from the water extract production. The sun protection factor (SPF) values of extracts analyzed were found to be in the range from 1.86 (GE96) to 4.80 (GW70). Therefore, G. lucidum solid waste appeared to be a valuable source of bioactive compounds with antioxidant, photoprotective and cytotoxic activities.



Similar content being viewed by others
References
S. Veljović, N. Nikićević, M. Nikšić, in Alcoholic Beverages, ed. by A.M. Grumezescu and A.M. Holban, (Woodhead Publishing, Cambridge, MA, 2019), p. 161–197
K.K. Hapuarachchi, W.A. Elkhateeb, S.C. Karunarathna, C.R. Cheng, A.R. Bandara, P. Kakumyan, K.D. Hyde, G.M. Daba, T.C. Wen, Mycosphere 9(5), 1025–1052 (2018)
S. Wachtel-Galor, J. Yuen, J.A. Buswell, I.F.F. Benzie, in Herbal Medicine: Biomolecular and Clinical Aspects, 2nd edition, ed by I.F.F. Benzie, S. Wachtel-Galor, (CRC Press/Taylor & Francis, Boca Raton (FL), 2011)
Y. Cao, X. Xu, S. Liu, L. Huang, J. Gu, Front. Pharmacol. 9, 121 (2018)
J. Lu, R. He, P. Sun, F. Zhang, R.J. Linhardt, A. Zhang, Int. J. Biol. Macromol. 150, 765–774 (2020)
A. Barbieri, V. Quagliariello, V. Del Vecchio, M. Falco, A. Luciano, N.J. Amruthraj, G. Nasti, A. Ottaiano, M. Berretta, R.V. Iaffaioli, C. Arra, Nutrients 9(3), 210 (2017)
J. Sheng, W. Wang, Q. Ma, R. Yang, R, Nord. Pulp. Pap. Res. J 35(1), 43–49 (2020)
Y.H. Liu, Y.S. Lin, K.L. Lin, Y.L. Lu, C.H. Chen, M.Y. Chien, H.F. Shang, S.Y. Lin, W.C. Hou, WC Bot. Stud. 56(1), 17 (2016)
M. Petrović, M. Jovanović, S. Lević, V. Nedović, D. Mitić-Ćulafić, T. Živković, S. Semren, Veljović, Waste Biomass. Valor 13, 1639–1651 (2022)
A.A.B. Badawy, C.J. Morgan, J.A. Turne, Amino Acids 34(4), 587–596 (2008)
M. Pantelić, D. Dabić Zagorac, I. Ćirić, M. Pergal, D. Relić, S. Todić, M. Natić, M J. Food Compos. Anal. 62, 76–83 (2017)
V.L. Singleton, R. Orthofer, R.M. Lamuela-Ravetós, in Methods in Enzymology, ed by L. Packer, (Academic Press, Cambridge, MA, 1999), p. 152–178
M.S. Blois, Nature 181(4617), 1199–1200 (1985)
I.F.F. Benzie, J.J. Strain, Anal. Biochem. 239(1), 70–76 (1966)
J.S. Mansur, M.N.R. Breder, M.D.A. Mansur, R.D. Azulay, RD An. Bras. Dermatol. 61(2), 167–172 (1986)
Y. Zhu, T.L. Tan, Spectrochim. Acta A. Mol. Biomol. Spectrosc 159, 68–77 (2016)
B. Sangeetha, A.S. Krishnamoorthy, D. Amirtham, D.J. Sundara Sharmila, P. Renukadevi, V.G. Malathi, Curr. J. Appl. Sci. Technol. 38(6), 1–8 (2020)
M. Kozarski, A. Klaus, M. Niksic, D. Jakovljevic, J.P.F.G. Helsper, L.J.L.D. Van Griensven, Food Chem. 129(4), 1667–1675 (2011)
X.-P. Li, J. Li, H. Liu, Y.-Z. Wang, Int. J. Food Prop. 23(1), 227–240 (2020)
Y. Zhu, T.L. Tan, Am. J. Anal. Chem. 6, 10 (2015)
Y.-Y. Wang, J.-Q. Li, H.-G. Liu, Y.-Z. Wang, Molecules 24, 2210 (2019)
A. Anghileri, R. Lantto, K. Kruus, C. Arosio, G. Freddi, J. Biotechnol. 127(3), 508–519 (2007)
Y. Li, J. Zhang, T. Li, T. Yang, Y. Wang, H. Liu, J. Spectr. 7813405 (2016)
Y.C. Lo, S.C. Chien, D.O. Mishchuk, C.M. Slupsky, J.L. Mau, Int. J. Med. Mushrooms 18(5), 413–424 (2016)
H. Zhang, H. Jiang, X. Zhang, J. Yan, Curr. Pharm. Anal. 14(6), 562–570 (2018)
B.U. Shamaki, F.I. Abdulrahman, U.K. Sandabe, K.A. Sanda, KA, Savannah Vet. J 2, 1–15 (2019)
K. Kumakura, C. Hori, H. Matsuoka, K. Igarashi, M. Samejima, J. Sci. Food Agric. 99(2), 529–535 (2019)
S. Veljović, M. Veljović, N. Nikićević, S. Despotović, S. Radulović, M. Nikšić, L. Filipović, J. Food Sci. Technol. Mysore 54, 1312–1320 (2017)
S. Pecić, N. Nikićević, M. Veljović, M. Jadranin, V. Tešević, M. Belović, M. Nikšić, Chem. Ind. Chem. Eng. Q. 22(2), 181–189 (2016)
Z. Liang, Y. Yi, Y. Guo, R. Wang, Q. Hu, X. Xiong, Int. J. Mol. Sci. 15(5), 9103–9116 (2014)
H. He, A. Li, S. Li, J. Tang, L. Li, L. Xiong, Biomed. Pharmacother 134, 111161 (2021)
W.Y. Cheng, X.Q. Wei, K.C. Siu, A.X. Song, J.Y. Wu, Int. J. Med. Mushrooms 20(7), 623–636 (2018)
K. Kawabata, M. Kanoh, M. Okazaki, R. Maeda, S. Mori, S. Akimoto, M. Inagaki, H. Nishi, Pharmaceuticals 13, 135 (2020)
W. Wong, J. Wu, I. Benzie, Brit J. Dermatol. 164(5), 980–986 (2011)
K. Sułkowska-Ziaja, K. Grabowska, A. Apola, A. Kryczyk-Poprawa, B. Muszyńska, Biotechnol. Lett. 43(5), 1051–1061 (2021)
Acknowledgements
Authors acknowledge financial support Ministry of Education, Science, and Technological Development, Republic of Serbia (Grant Nos. 200051 and 200168). The authors thank Steva Lević (Faculty of Agriculture, University of Belgrade) for obtaining ATR-FTIR spectra. Authors wish to express gratitude to “Ganoherb Technology (Fujian) Corporation” for providing G. lucidum residues investigated in this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
None.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Veljović, S., Petrović, M., Jovanović, M. et al. Industrial solid wastes from Ganoderma lucidum extract production: chemical characterization and investigation of antioxidant, photoprotective and cytotoxic activities. Food Measure 17, 3673–3682 (2023). https://doi.org/10.1007/s11694-023-01897-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11694-023-01897-6