Abstract
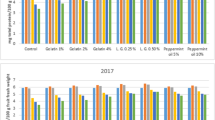
Sap from the trunk of oil palm (Elaeis guineensis) was processed into syrup in this study. Physicochemical properties of syrups produced from the sap of freshly cut oil palm trunks (OPT0syrup) and sap of trunks stored for 60 days (OPT60syrup) at ambient temperature were compared to nipa palm syrup (Nsyrup), maple syrup (Msyrup), and two commercial glucose syrups (G1syrup and G2syrup). The total soluble solids for the syrups (OPT0sy, OPT60sy, Nsy, Msy, G1sy and G2sy) were in the range of 66 to 82 ˚Brix. The sugar analysis indicated that glucose was the main sugar for OPT0sy and OPT60sy as 367 and 300 mg/g, respectively, whereby sucrose was the foremost sugar for Nsy and Msy. The colour of the syrups was ranked from the darkest to the lightest; OPT60syrup > OPT0syrup > Msyrup > Nsyrup > G1syrup > G2syrup. OPT0syrup and OPT60syrup achieved reducing capacity of 825 and 886 mg GAE/100 g, respectively, significantly higher than other syrups. The free radical scavenging activities of OPT0syrup and OPT60syrup were 74% and 70%, respectively. In conclusion, the oil palm trunk syrup has similar physicochemical properties with the commercial syrups, and it has the potential to be an alternative to the commercial sweeteners with added nutritional benefits.

Similar content being viewed by others

References
H. Yamada, R. Tanaka, O. Sulaiman, R. Hashim, Z.A.A. Hamid, M.K.A. Yahya, A. Kosugi, T. Arai, Y. Murata, S. Nirasawa, K. Yamamoto, S. Ohara, M.N.M. Yusof, W.A. Ibrahim, Y. Mori, Old oil palm trunk: a promising source of sugars for bioethanol production. Biomass Bioenerg. 34, 1608–1613 (2010)
S. Jalani, B. Yusof, D. Ariffin, K.W. Chan, N. Rajanaidu, Prospects of elevating national oil palm productivity: a Malaysian perspective. Oil Palm Ind. Econ. J. 2(2), 1–9 (2002)
I. Wan-Asma, S. Mahanim, H. Zulkafli, S. Osman, S.Y. Mori, in Regional Workshop on UNEP/DTIE/IETC in collaboration with GEC (Osaka, Japan, 2010)
K.O. Lim, F.H. Ahmaddin, S.M. Vizhi, A note on the conversion of oil-palm trunks to glucose via acid hydrolysis. Bioresour. Technol. 59(1), 33–35 (1997)
P. Hull, Glucose Syrups: Technology and Applications (John Wiley & Sons, Singapore, 2010), pp. 1–388
A. Keating, R.A. Chez, Ginger syrup as an antiemetic in early pregnancy. Altern. Ther. Health Med. 8(5), 89–91 (2002)
G.A. Bray, S.J. Nielsen, B.M. Popkin, Consumption of high-fructose corn syrup in beverages may play a role in the epidemic of obesity. Am. J. Clin. Nutr. 79(4), 537–543 (2004)
K.A. Strauss, B. Wardley, D. Robinson, C. Hendrickson, N.L. Rider, E.G. Puffenberger, D. Shellmer, A.B. Moser, D.H. Morton, Classical maple syrup urine disease and brain development: principles of management and formula design. Mol. Genet. Metab. 99(4), 333–345 (2010)
A. Kosugi, R. Tanaka, K. Magara, Y. Murata, T. Arai, S. Othman, H. Rokiah, Z.A.H. Aimi, M.A.Y. Khairul, M.M.Y. Nor, I. Wan-Asma, Y. Mori, Ethanol and lactic acid production using sap squeezed from old oil palm trunks felled for replanting. J. Biosci. Bioeng. 110(3), 322–325 (2010)
P. Noparat, P. Prasertsan, Isolation and characterization of high hydrogen producing strain Clostridium beijerinckii PS-3 from fermented oil palm sap. Int. J. Hydrog. Energy 36(21), 14086–14092 (2011)
B.E. Lokesh, Z.A.A. Hamid, T. Arai, A. Kosugi, Y. Murata, R. Hashim, S. Othman, Y. Mori, K. Sudesh, Potential of oil palm trunk sap as a novel inexpensive renewable carbon feedstock for polyhydroxyalkanoate biosynthesis and as a bacterial growth medium. Clean (Weinh) 40(3), 310–317 (2012)
Y. Murata, R. Tanaka, K. Fujimoto, A. Kosugi, T. Arai, E. Togawa, T. Takano, W.A. Ibrahim, P. Elham, O. Sulaiman, R. Hashim, Y. Mori, Development of sap compressing systems from oil palm trunk. Biomass Bioenerg. 51, 8–16 (2013)
I.B. Thabet, S. Besbes, M. Masmoudi, H. Attia, C. Deroanne, C. Blecker, Compositional, physical, antioxidant and sensory characteristics of novel syrup from date palm (Phoenix dactylifera L.). Food Sci. Technol. Int. 15(6), 583–590 (2009)
M. Al-Farsi, C. Alasalvar, A. Morris, M. Baron, F. Shahidi, Comparison of antioxidant activity, anthocyanins, carotenoids, and phenolics of three native fresh and sun-dried date (Phoenix dactylifera L.) varieties grown in Oman. J. Agric. Food Chem. 53(19), 7592–7599 (2005)
L. Li, N.P. Seeram, Maple syrup phytochemicals include lignans, coumarins, a stilbene, and other previously unreported antioxidant phenolic compounds. J. Agric. Food Chem. 58(22), 11673–11679 (2010)
W. Brand-Williams, M.E. Cuvelier, C.L.W.T. Berset, Use of a free radical method to evaluate antioxidant activity. LWT-Food Sci. Technol. 28(1), 25–30 (1995)
C.G.A. Davies, T.P. Labuza, The Maillard Reaction: Application to Confectionery Products (Penn State University Press, Minnesota, 1997), pp. 35–66
M.M. Theron, J.R. Lues, Organic Acids and Food Preservation (CRC Press, Boca Raton, 2010), pp. 1–340
A. Etienne, M. Génard, P. Lobit, D. Mbeguié-A-Mbéguié, C. Bugaud, What controls fleshy fruit acidity? A review of malate and citrate accumulation in fruit cells. J. Exp. Bot. 64(6), 1451–1469 (2013)
F.K. Alanazi, Utilization of date syrup as a tablet binder, comparative study. Saudi Pharm. J. 18(2), 81–89 (2010)
S.I. Martins, W.M. Jongen, M.A.V. Boekel, A review of Maillard reaction in food and implications to kinetic modelling. Trends Food Sci. Technol. 11(9), 364–373 (2001)
O.O. James, M.A. Mesubi, L.A. Usman, S.O. Yeye, K.O. Anjaku, K.O. Ogunniran, O.O. Ajani, T.O. Siyanbola, Physical characterisation of some honey samples from North-Central Nigeria. Int. J. Phys. Sci. 4(9), 464–470 (2009)
W.P. Schellart, Rheology and density of glucose syrup and honey: determining their suitability for usage in analogue and fluid dynamic models of geological processes. J. Struct. Geol. 33(6), 1079–1088 (2011)
T.D. Perkins, A.K.V.D. Berg, Chap. 4 Maple syrup—production, composition, chemistry, and sensory characteristics. Adv. Food. Nutr. Res. 56, 101–143 (2009)
A. Mondal, R.L. Buchanan, Y.M. Lo, Computational fluid dynamics approaches in quality and hygienic production of semisolid low-moisture foods: a review of critical factors. J. Food. Sci. 79(10), 1861–1870 (2014)
D.W. Ball, The chemical composition of maple syrup. J. Chem. Educ. 84(10), 1647–1650 (2007)
J.M. deMan, Principles of Food Chemistry, 3rd edn. (Aspen Publishers, Inc., Maryland, 1999), pp. 163–208
E. Kim, Relationship between viscosity and sugar concentration in aqueous sugar solution using the Stokes’ Law and Newton’s First Law of Motion. Science One Program (The University of British Columbia, Canada, 2010). https://circle.ubc.ca/bitstream/handle/2429/25322/Kim_Evelyn_Science_. Accessed 19 July 2018
C.W. Chang, R.D. Ryan, Effects of water stress on starch and sucrose metabolism in cotton leaves. Starch-Stärke 39(3), 84–87 (1987)
K. Maruyama, M. Takeda, S. Kidokoro, K. Yamada, Y. Sakuma, K. Urano, M. Fujita, K. Yoshiwara, S. Matsukura, Y. Morishita, R. Sasaki, H. Suzuki, K. Saito, D. Shibata, K. Shinozaki, K. Yamaguchi-Shinozaki, Metabolic pathways involved in cold acclimation identified by integrated analysis of metabolites and transcripts regulated by DREB1A and DREB2A. Plant Physiol. 150(4), 1972–1980 (2009)
J.F. Fox, Harvest of the Palm, Ecological Change in Eastern Indonesia (Harvard University Press, Cambridge, 1977), pp. 1–290
C. Dalibard, Overall view on the tradition of tapping palm trees and prospects for animal production. Livest. Res. Rural Dev. 11(1), 1–37 (1999)
M. Chisari, R.N. Barbagallo, G. Spagna, Characterization and role of polyphenol oxidase and peroxidase in browning of fresh-cut melon. J. Agric. Food Chem. 56(1), 132–138 (2008)
R. Banerji, P. Singh, S.I. Anwar, S. Solomon, Effect of reducing sugars on non-enzymic browning during thermo-evaporation of sugarcane juice for making jaggery. Sugar Tech. 14(4), 428–431 (2012)
L. Hobbs, Starch: Chemistry and Technology, 3rd edn. (Academic Press, Elsevier, London, 2009), pp. 6–7
Z.A.H. Aimi, T. Arai, M.R.S. Fatimah, A. Kosugi, O. Sulaiman, R. Hashim, S. Nirasawa, T. Ryokei, B.E. Lokesh, K. Sudesh, Y. Murata, Analysis of free sugar and starch in oil palm trunks (Elaeis guineensis Jacq.) from various cultivars as a feedstock for bioethanol production. Int. J. Green Energy 1, 37–41 (2015)
S. Kermasha, M. Goetghebeur, J. Dumont, Determination of phenolic compound profiles in maple products by high-performance liquid chromatography. J. Agric. Food Chem. 43(3), 708–716 (1995) (1995)
M. Boscaiu, I. Bautista, P. Donat, J. Llinares, L.L.U.L. Cristina, O. Mayoral, O. Vicente, Phenolic compounds as stress markers in plants from gypsum habitats. Bull. Univ. Agric. Sci. Vet. Med. Cluj-Napoca Hortic. 67(1), 44–49 (2010)
M. Thériault, S. Caillet, S. Kermasha, M. Lacroix, Antioxidant, antiradical and antimutagenic activities of phenolic compounds present in maple products. Food Chem. 98(3), 490–501 (2006)
J.B. Harborne, Phytochemical Methods: A Guide to Modern Techniques of Plant Analysis, 3rd edn. (Chapman and Hall, London, 1984), pp. 1–286
J. Gruz, F.A. Ayaz, H. Torun, M. Strnad, Phenolic acid content and radical scavenging activity of extracts from medlar (Mespilus germanica L.) fruit at different stages of ripening. Food Chem. 124(1), 271–277 (2011)
N. Gheldof, X.H. Wang, N.J. Engeseth, Identification and quantification of antioxidant compounds of honeys form various floral sources. J. Agric. Food Chem. 50, 5870–5877 (2002) (2002)
D.D. Schramm, M. Karim, H.R. Schrader, R.R. Holt, M. Cardetti, C.L. Keen, Honey with high levels of antioxidants can provide protection to healthy human subjects. J. Agric. Food Chem. 51(6), 1732–1735 (2003)
J. Legault, K. Girard-Lalancette, C. Grenon, C. Dussault, A. Pichette, Antioxidant activity, inhibition of nitric oxide overproduction, and in vitro antiproliferative effect of maple sap and syrup from Acer saccharum. J. Med. Food. 13(2), 460–468 (2010)
P. Górnaś, E. Šnē, A. Siger, D. Segliņa, Sea buckthorn (Hippophae rhamnoides L.) leaves as valuable source of lipophilic antioxidants: the effect of harvest time, sex, drying and extraction methods. Ind. Crops. Prod. 60, 1–7 (2014)
Acknowledgements
The authors acknowledge the support from the FRGS Grants (203/PTEKIND/6711370) and (203/PTEKIND/6711530) for the current research project, and the Forest Research Institute of Malaysia for supplying the oil palm trunk samples. Syazana, S. thanks MyBrain for financial assistance during the period of the study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sulaiman, S., Kamilah, H., Mahmood, K. et al. Physicochemical characterisation of oil palm (Elaeis guineensis) trunk syrup from the sap of different storage period as potential sweetener. Food Measure 13, 1011–1019 (2019). https://doi.org/10.1007/s11694-018-0015-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11694-018-0015-y