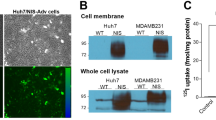
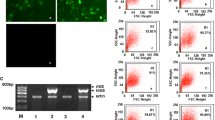
Abstract
Norepinephrine transporter (NET) transfection leads to significant uptake of iodine-131-labeled metaiodobenzylguanidine (131I-MIBG) in non-neuroendocrine tumors. However, the use of 131I-MIBG is limited by its short retention time in target cells. To prolong the retention of 131I-MIBG in target cells, we infected hepatocarcinoma (HepG2) cells with Lentivirus-encoding human NET and vesicular monoamine transporter 2 (VMAT2) genes to obtain NET-expressing, NET-VMAT2-coexpressing, and negative-control cell lines. We evaluated the uptake and efflux of 131I-MIBG both in vitro and in vivo in mice bearing transfected tumors. NET-expressing and NET-VMAT2-coexpressing cells respectively showed 2.24 and 2.22 times higher 131I-MIBG uptake than controls. Two hours after removal of 131I-MIBG-containing medium, 25.4% efflux was observed in NET-VMAT2-coexpressing cells and 38.6% in NET-expressing cells. In vivo experiments were performed in nude mice bearing transfected tumors; results revealed that NET-VMAT2-coexpressing tumors had longer 131I-MIBG retention time than NET-expressing tumors. Meanwhile, NET-VMAT2-coexpressing and NET-expressing tumors displayed 0.54% and 0.19%, respectively, of the injected dose per gram of tissue 24 h after 131I-MIBG administration. Cotransfection of HepG2 cells with NET and VMAT2 resulted in increased 131I-MIBG uptake and retention. However, the degree of increase was insufficient to be therapeutically effective in target cells.
Similar content being viewed by others
References
Dubois SG, Geier E, Batra V, Yee SW, Neuhaus J, Segal M, Martinez D, Pawel B, Yanik G, Naranjo A, London WB, Kreissman S, Baker D, Attiyeh E, Hogarty MD, Maris JM, Giacomini K, Matthay KK. Evaluation of norepinephrine transporter expression and metaiodobenzylguanidine avidity in neuroblastoma: a report from the Children’s Oncology Group. Int J Mol Imaging 2012; 2012:250834
Carlin S, Mairs RJ, McCluskey AG, Tweddle DA, Sprigg A, Estlin C, Board J, George RE, Ellershaw C, Pearson AD, Lunec J, Montaldo PG, Ponzoni M, van Eck-Smit BL, Hoefnagel CA, van den Brug MD, Tytgat GA, Caron HN. Development of a real-time polymerase chain reaction assay for prediction of the uptake of meta-[(131)I]iodobenzylguanidine by neuroblastoma tumors. Clin Cancer Res 2003; 9(9): 3338–3344
Lode HN, Bruchelt G, Seitz G, Gebhardt S, Gekeler V, Niethammer D, Beck J. Reverse transcriptase-polymerase chain reaction (RTPCR) analysis of monoamine transporters in neuroblastoma cell lines: correlations to meta-iodobenzylguanidine (MIBG) uptake and tyrosine hydroxylase gene expression. Eur J Cancer 1995; 31(4): 586–590
Gonias S, Goldsby R, Matthay KK, Hawkins R, Price D, Huberty J, Damon L, Linker C, Sznewajs A, Shiboski S, Fitzgerald P. Phase II study of high-dose [131I]metaiodobenzylguanidine therapy for patients with metastatic pheochromocytoma and paraganglioma. J Clin Oncol 2009; 27(25): 4162–4168
Kang TI, Brophy P, Hickeson M, Heyman S, Evans AE, Charron M, Maris JM. Targeted radiotherapy with submyeloablative doses of 131I-MIBG is effective for disease palliation in highly refractory neuroblastoma. J Pediatr Hematol Oncol 2003; 25(10): 769–773
Fitzgerald PA, Goldsby RE, Huberty JP, Price DC, Hawkins RA, Veatch JJ, Dela Cruz F, Jahan TM, Linker CA, Damon L, Matthay KK. Malignant pheochromocytomas and paragangliomas: a phase II study of therapy with high-dose 131I-metaiodobenzylguanidine (131I-MIBG). Ann N Y Acad Sci 2006; 1073(1): 465–490
Matthay KK, Panina C, Huberty J, Price D, Glidden DV, Tang HR, Hawkins RA, Veatch J, Hasegawa B. Correlation of tumor and whole-body dosimetry with tumor response and toxicity in refractory neuroblastoma treated with (131)I-MIBG. J Nucl Med 2001; 42(11): 1713–1721
DuBois SG, Messina J, Maris JM, Huberty J, Glidden DV, Veatch J, Charron M, Hawkins R, Matthay KK. Hematologic toxicity of highdose iodine-131-metaiodobenzylguanidine therapy for advanced neuroblastoma. J Clin Oncol 2004; 22(12): 2452–2460
Fullerton NE, Boyd M, Ross SC, Pimlott SL, Babich J, Kirk D, Zalutsky MR, Mairs RJ. Comparison of radiohaloanalogues of meta-iodobenzylguanidine (MIBG) for a combined gene- and targeted radiotherapy approach to bladder carcinoma. Med Chem 2005; 1(6): 611–618
Mairs RJ, Ross SC, McCluskey AG, Boyd M. A transfectant mosaic xenograft model for evaluation of targeted radiotherapy in combination with gene therapy in vivo. J Nucl Med 2007; 48(9): 1519–1526
Jia ZY, Deng HF, Huang R, Yang YY, Yang XC, Qi ZZ, Ou XH. In vitro and in vivo studies of adenovirus-mediated human norepinephrine transporter gene transduction to hepatocellular carcinoma. Cancer Gene Ther 2011; 18(3): 196–205
Altmann A, Kissel M, Zitzmann S, Kübler W, Mahmut M, Peschke P, Haberkorn U. Increased MIBG uptake after transfer of the human norepinephrine transporter gene in rat hepatoma. J Nucl Med 2003; 44(6): 973–980
Fullerton NE, Mairs RJ, Kirk D, Keith WN, Carruthers R, McCluskey AG, Brown M, Wilson L, Boyd M. Application of targeted radiotherapy/gene therapy to bladder cancer cell lines. Eur Urol 2005; 47(2): 250–256
Boyd M, Cunningham SH, Brown MM, Mairs RJ, Wheldon TE. Noradrenaline transporter gene transfer for radiation cell kill by 131I meta-iodobenzylguanidine. Gene Ther 1999; 6(6): 1147–1152
Parsons SM. Transport mechanisms in acetylcholine and monoamine storage. FASEB J 2000; 14(15): 2423–2434
Kölby L, Bernhardt P, Levin-Jakobsen AM, Johanson V, Wängberg B, Ahlman H, Forssell-Aronsson E, Nilsson O. Uptake of metaiodobenzylguanidine in neuroendocrine tumours is mediated by vesicular monoamine transporters. Br J Cancer 2003; 89(7): 1383–1388
Temple W, Mendelsohn L, Kim GE, Nekritz E, Gustafson WC, Lin L, Giacomini K, Naranjo A, van Ryn C, Yanik GA, Kreissman SG, Hogarty M, Matthay KK, Du Bois SG. Vesicular monoamine transporter protein expression correlates with clinical features, tumor biology, and MIBG avidity in neuroblastoma: a report from the Children’s Oncology Group. Eur J Nucl Med Mol Imaging 2016; 43(3): 474–481
Erickson JD, Schafer MK, Bonner TI, Eiden LE, Weihe E. Distinct pharmacological properties and distribution in neurons and endocrine cells of two isoforms of the human vesicular monoamine transporter. Proc Natl Acad Sci USA 1996; 93(10): 5166–5171
Liu Y, Schweitzer ES, Nirenberg MJ, Pickel VM, Evans CJ, Edwards RH. Preferential localization of a vesicular monoamine transporter to dense core vesicles in PC12 cells. J Cell Biol 1994; 127(5): 1419–1433
Erickson JD, Eiden LE, Hoffman BJ. Expression cloning of a reserpine-sensitive vesicular monoamine transporter. Proc Natl Acad Sci USA 1992; 89(22): 10993–10997
Liu Y, Peter D, Roghani A, Schuldiner S, Privé GG, Eisenberg D, Brecha N, Edwards RH. A cDNA that suppresses MPP + toxicity encodes a vesicular amine transporter. Cell 1992; 70(4): 539–551
Erickson JD, Eiden LE. Functional identification and molecular cloning of a human brain vesicle monoamine transporter. J Neurochem 1993; 61(6): 2314–2317
Gasnier B, Krejci E, Botton D, Massoulié J, Henry JP. Expression of a bovine vesicular monoamine transporter in COS cells. FEBS Lett 1994; 342(3): 225–229
Zan LB, Yang YY, Jin JN, Ou XH. Synthesis of 3-trimethylsilylbenzylguanidine as precursor for 125I labelling. Atomic Energy Sci Technol (Yuan Zi Neng Ke Xue Ji Shu) 2007; 41(6): 689–693 (in Chinese)
Moroz MA, Serganova I, Zanzonico P, Ageyeva L, Beresten T, Dyomina E, Burnazi E, Finn RD, Doubrovin M, Blasberg RG. Imaging hNET reporter gene expression with 124I-MIBG. J Nucl Med 2007; 48(5): 827–836
Bomanji J, Levison DA, Flatman WD, Horne T, Bouloux PM, Ross G, Britton KE, Besser GM. Uptake of iodine-123 MIBG by pheochromocytomas, paragangliomas, and neuroblastomas: a histopathological comparison. J Nucl Med 1987; 28(6): 973–978
Robson JA, Sidell N. Ultrastructural features of a human neuroblastoma cell line treated with retinoic acid. Neuroscience 1985; 14(4): 1149–1162
Iavarone A, Lasorella A, Servidei T, Riccardi R, Mastrangelo R. Uptake and storage of m-iodobenzylguanidine are frequent neuronal functions of human neuroblastoma cell lines. Cancer Res 1993; 53(2): 304–309
Taupenot L, Harper KL, O’Connor DT. The chromograninsecretogranin family. N Engl J Med 2003; 348(12): 1134–1149
Stettler H, Beuret N, Prescianotto-Baschong C, Fayard B, Taupenot L, Spiess M. Determinants for chromogranin A sorting into the regulated secretory pathway are also sufficient to generate granulelike structures in non-endocrine cells. Biochem J 2009; 418(1): 81–91
Huh YH, Jeon SH, Yoo SH. Chromogranin B-induced secretory granule biogenesis: comparison with the similar role of chromogranin A. J Biol Chem 2003; 278(42): 40581–40589
Beuret N, Stettler H, Renold A, Rutishauser J, Spiess M. Expression of regulated secretory proteins is sufficient to generate granule-like structures in constitutively secreting cells. J Biol Chem 2004; 279(19): 20242–20249
Acknowledgements
We thank Yuanyou Yang, PhD, for helping in the preparation of 131I-MIBG. This study was funded by the National Natural Science Foundation of China (No. 81271602).
Author information
Authors and Affiliations
Corresponding author
Additional information
Yanlin Zhao and Xiao Zhong contributed equally to this work.
Rights and permissions
About this article
Cite this article
Zhao, Y., Zhong, X., Ou, X. et al. Cotransfecting norepinephrine transporter and vesicular monoamine transporter 2 genes for increased retention of metaiodobenzylguanidine labeled with iodine 131 in malignant hepatocarcinoma cells. Front. Med. 11, 120–128 (2017). https://doi.org/10.1007/s11684-017-0501-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11684-017-0501-3