Abstract
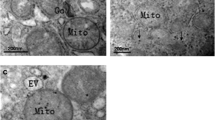
The aim of this paper is to investigate the mechanism of hepatocellular damage in rats caused by low serum selenium. Thirty six rats were randomly divided into 2 groups: group A (fed with low-selenium diet from the Keshan Disease area with the content of selenium being 0.017 mg/kg); group B [fed with selenium-supplemented diet and 0.3 mg/L selenium (Na2SeO3) was added to the drinking water]. Both were respectively fed for 12 weeks. At the end of the 12th week, the levels of serum selenium, glutathione peroxidase (GPX) and malondialdehyde (MDA) in hepatic tissue were measured; the hepatocellular ultrastructure and apoptosis were observed as well. The levels of serum selenium and GPX in group A were markedly lower than those in group B. MDA level in group A was significantly higher than that in group B. Under the electron microscope (EM), the mitochondria were remarkably changed in group A. The rate of liver cell apoptosis appeared much higher in group A as well. It indicated that the damage caused by selenium deficiency was through the process of oxidation. Selenium deficiency led to apoptosis of hepatocytes where oxidative damage to mitochondria might be the cause.
Similar content being viewed by others
References
Jabłońska-Kaszewska I, Swiatkowska-Stodulska R, Łukasiak J, Dejneka W, Dorosz A, Dabrowska E, Falkiewicz B. Serum selenium levels in alcoholic liver disease. Med Sci Monit, 2003, 9(Suppl 3): 15–18
Liu S Y, Li T Y, Zhao Z, Man R Y, Wang F. Effects of selenium and alpha-tocopherol on liver damage induced by feeding grains from an endemic area of Keshan disease in rats. Mol Cell Biochem, 1994, 132(2): 109–115
Thuluvath P J, Triger D R. Selenium in chronic liver disease. J Hepatol, 1992, 14(2–3): 176–182
Jüngst C, Cheng B, Gehrke R, Schmitz V, Nischalke H D, Ramakers J, Schramel P, Schirmacher P, Sauerbruch T, Caselmann W H. Oxidative damage is increased in human liver tissue adjacent to hepatocellular carcinoma. Hepatology, 2004, 39(6): 1663–1672
Mandal A K, Sinha J, Mandal S, Mukhopadhyay S, Das N. Targeting of liposomal flavonoid to liver in combating hepatocellular oxidative damage. Drug Deliv, 2002, 9(3): 181–185
Jeyabal P V, Syed M B, Venkataraman M, Sambandham J K, Sakthisekaran D. Apigenin inhibits oxidative stress-induced macromolecular damage in N-nitrosodiethylamine (NDEA)-induced hepatocellular carcinogenesis in Wistar albino rats. Mol Carcinog, 2005, 44(1): 11–20
Jozanov-Stankov O, Demajo M, Djujić I, Mandić M. Selenium intake as a modulator of responsiveness to oxidative stress. J Environ Pathol Toxicol Oncol, 1998, 17(3–4): 251–257
Doyen P, Bigot A, Vasseur P, Rodius F. Molecular cloning and expression study of pi-class glutathione S-transferase (pi-GST) and selenium-dependent glutathione peroxidase (Se-GPx) transcripts in the freshwater bivalve Dreissena polymorpha. Comp Biochem Physiol C Toxicol Pharmacol, 2008, 147(1): 69–77
Baliga M S, Wang H, Zhuo P, Schwartz J L, Diamond A M. Selenium and GPx-1 overexpression protect mammalian cells against UV-induced DNA damage. Biol Trace Elem Res, 2007, 115(3): 227–242
Turturro F, Friday E, Welbourne T. Hyperglycemia regulates thioredoxin-ROS activity through induction of thioredoxin-interacting protein (TXNIP) in metastatic breast cancer-derived cells MDA-MB-231. BMC Cancer, 2007, 7: 96
Lin C H, Lin P H. Induction of ROS formation, poly(ADP-ribose) polymerase-1 activation, and cell death by PCB126 and PCB153 in human T47D and MDA-MB-231 breast cancer cells. Chem Biol Interact, 2006, 162(2): 181–194
Gawel S, Wardas M, Niedworok E, Wardas P. Malondialdehyde (MDA) as a lipid peroxidation marker. Wiad Lek, 2004, 57(9–10): 453–455
Newmeyer D D, Ferguson-Miller S. Mitochondria: releasing power for life and unleashing the machineries of death. Cell, 2003, 112(4): 481–490
Indo H P, Davidson M, Yen H C, Suenaga S, Tomita K, Nishii T, Higuchi M, Koga Y, Ozawa T, Majima H J. Evidence of ROS generation by mitochondria in cells with impaired electron transport chain and mitochondrial DNA damage. Mitochondrion, 2007, 7(1–2): 106–118
Scherz-Shouval R, Elazar Z. ROS, mitochondria and the regulation of autophagy. Trends Cell Biol, 2007, 17(9): 422–427
Linford N J, Schriner S E, Rabinovitch P S. Oxidative damage and aging: spotlight on mitochondria. Cancer Res, 2006, 66(5): 2497–2499
Pessayre D, Mansouri A, Haouzi D, Fromenty B. Hepatotoxicity due to mitochondrial dysfunction. Cell Biol Toxicol, 1999, 15(6): 367–373
Yang J C, Cortopassi G A. Induction of the mitochondrial permeability transition causes release of the apoptogenic factor cytochrome c. Free Radic Biol Med, 1998, 24(4): 624–631
Reid A B, Kurten R C, McCullough S S, Brock R W, Hinson J A. Mechanisms of acetaminophen-induced hepatotoxicity: role of oxidative stress and mitochondrial permeability transition in freshly isolated mouse hepatocytes. J Pharmacol Exp Ther, 2005, 312(2): 509–516
Kowaltowski A J, Castilho R F, Vercesi A E. Mitochondrial permeability transition and oxidative stress. FEBS Lett, 2001, 495(1–2): 12–15
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lu, Y., Qu, B., Liu, C. et al. Mechanism of hepatocellular damage in rat caused by low serum selenium. Front. Med. China 2, 255–258 (2008). https://doi.org/10.1007/s11684-008-0048-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11684-008-0048-4