Abstract
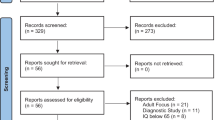
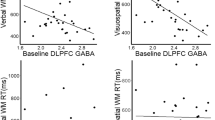
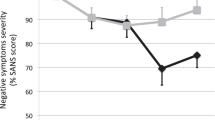
Whether brain stimulation could modulate brain structure in autism remains unknown. This study explored the impact of continuous theta burst stimulation (cTBS) over the left dorsolateral prefrontal cortex (DLPFC) on white matter macro/microstructure in intellectually able children and emerging adults with autism. Sixty autistic participants were randomized (30 active) and received active or sham cTBS for eight weeks twice per week, 16 total sessions using a double-blind (participant-, rater-, analyst-blinded) design. All participants received high-angular resolution diffusion MR imaging at baseline and week 8. Twenty-eight participants in the active group and twenty-seven in the sham group with good imaging quality entered the final analysis. With longitudinal fixel-based analysis and network-based statistics, we found no significant difference between the active and sham groups in changes of white matter macro/microstructure and connections following cTBS. In addition, we found no association between baseline white matter macro/microstructure and autistic symptom changes from baseline to week 8 in the active group. In conclusion, we did not find a significant impact of left DLPFC cTBS on white matter macro/microstructure and connections in children and emerging adults with autism. These findings need to be interpreted in the context that the current intellectually able cohort in a single university hospital site limits the generalizability. Future studies are required to investigate if higher stimulation intensities and/or doses, other personal factors, or rTMS parameters might confer significant brain structural changes visible on MRI in ASD.




Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author upon reasonable request.
References
Altman, D. G. (1985). Comparability of Randomised Groups. Journal of the Royal Statistical Society. Series D (The Statistician), 34(1), 125–136.
Ameis, S. H., Blumberger, D. M., Croarkin, P. E., Mabbott, D. J., Lai, M. C., Desarkar, P., . . . Daskalakis, Z. J. (2020). Treatment of Executive Function Deficits in autism spectrum disorder with repetitive transcranial magnetic stimulation: A double-blind, sham-controlled, pilot trial. Brain Stimul, 13(3), 539–547. https://doi.org/10.1016/j.brs.2020.01.007
Baruth, J. M., Casanova, M. F., El-Baz, A., Horrell, T., Mathai, G., Sears, L., & Sokhadze, E. (2010). Low-Frequency Repetitive Transcranial Magnetic Stimulation (rTMS) Modulates Evoked-Gamma Frequency Oscillations in Autism Spectrum Disorder (ASD). Journal of Neurotherapy, 14(3), 179–194. https://doi.org/10.1080/10874208.2010.501500
Bastiani, M., Cottaar, M., Fitzgibbon, S. P., Suri, S., Alfaro-Almagro, F., Sotiropoulos, S. N., . . . Andersson, J. L. R. (2019). Automated quality control for within and between studies diffusion MRI data using a non-parametric framework for movement and distortion correction. Neuroimage, 184, 801–812. https://doi.org/10.1016/j.neuroimage.2018.09.073
Blumberger, D. M., Maller, J. J., Thomson, L., Mulsant, B. H., Rajji, T. K., Maher, M., . . . Daskalakis, Z. J. (2016). Unilateral and bilateral MRI-targeted repetitive transcranial magnetic stimulation for treatment-resistant depression: a randomized controlled study. J Psychiatry Neurosci, 41(4), E58–66. https://doi.org/10.1503/jpn.150265
Blumberger, D. M., Vila-Rodriguez, F., Thorpe, K. E., Feffer, K., Noda, Y., Giacobbe, P., ... Downar, J. (2018). Effectiveness of theta burst versus high-frequency repetitive transcranial magnetic stimulation in patients with depression (THREE-D): a randomised non-inferiority trial. Lancet, 391(10131), 1683–1692. https://doi.org/10.1016/S0140-6736(18)30295-2
Bourgeron, T. (2015). From the genetic architecture to synaptic plasticity in autism spectrum disorder. Nature Reviews Neuroscience, 16(9), 551–563. https://doi.org/10.1038/nrn3992
Brown, M. S., Singel, D., Hepburn, S., & Rojas, D. C. (2013). Increased glutamate concentration in the auditory cortex of persons with autism and first-degree relatives: A (1)H-MRS study. Autism Research, 6(1), 1–10. https://doi.org/10.1002/aur.1260
Buard, I., Rogers, S. J., Hepburn, S., Kronberg, E., & Rojas, D. C. (2013). Altered oscillation patterns and connectivity during picture naming in autism. Frontiers in Human Neuroscience, 7, 742. https://doi.org/10.3389/fnhum.2013.00742
Calabrese, F., Riva, M. A., & Molteni, R. (2016). Synaptic alterations associated with depression and schizophrenia: Potential as a therapeutic target. Expert Opinion on Therapeutic Targets, 20(10), 1195–1207. https://doi.org/10.1080/14728222.2016.1188080
Carlisi, C. O., Norman, L. J., Lukito, S. S., Radua, J., Mataix-Cols, D., & Rubia, K. (2017). Comparative Multimodal Meta-analysis of Structural and Functional Brain Abnormalities in Autism Spectrum Disorder and Obsessive-Compulsive Disorder. Biological Psychiatry, 82(2), 83–102. https://doi.org/10.1016/j.biopsych.2016.10.006
Carper, R. A., & Courchesne, E. (2005). Localized enlargement of the frontal cortex in early autism. Biological Psychiatry, 57(2), 126–133. https://doi.org/10.1016/j.biopsych.2004.11.005
Casanova, M. F., Baruth, J. M., El-Baz, A., Tasman, A., Sears, L., & Sokhadze, E. (2012). Repetitive Transcranial Magnetic Stimulation (rTMS) Modulates Event-Related Potential (ERP) Indices of Attention in Autism. Translational Neuroscience, 3(2), 170–180. https://doi.org/10.2478/s13380-012-0022-0
Casanova, M. F., Hensley, M. K., Sokhadze, E. M., El-Baz, A. S., Wang, Y., Li, X., & Sears, L. (2014). Effects of weekly low-frequency rTMS on autonomic measures in children with autism spectrum disorder. Frontiers in Human Neuroscience, 8, 851. https://doi.org/10.3389/fnhum.2014.00851
Caulfield, K. A., Badran, B. W., Li, X., Bikson, M., & George, M. S. (2020b). Can transcranial electrical stimulation motor threshold estimate individualized tDCS doses over the prefrontal cortex? Evidence from reverse-calculation electric field modeling. Brain Stimulation, 13(4), 1150–1152. https://doi.org/10.1016/j.brs.2020.05.012
Caulfield, K. A., Li, X., & George, M. S. (2021). A reexamination of motor and prefrontal TMS in tobacco use disorder: Time for personalized dosing based on electric field modeling? Clinical Neurophysiology, 132(9), 2199–2207. https://doi.org/10.1016/j.clinph.2021.06.015
Caulfield, K. A., Badran, B. W., DeVries, W. H., Summers, P. M., Kofmehl, E., Li, X., . . . George, M. S. (2020). Transcranial electrical stimulation motor threshold can estimate individualized tDCS dosage from reverse-calculation electric-field modeling. Brain Stimul, 13(4), 961–969. https://doi.org/10.1016/j.brs.2020.04.007
Chang, J. C., Lai, M. C., Chien, Y. L., Cheng, C. Y., Wu, Y. Y., & Gau, S. S. (2023). Psychometric properties of the Mandarin version of the autism diagnostic observation Schedule-Generic. Journal of the Formosan Medical Association, 122(7), 574–583. https://doi.org/10.1016/j.jfma.2023.01.008
Cocchi, L., Naze, S., Robinson, C., Webb, L., Sonkusare, S., Hearne, L. J., . . . Breakspear, M. (2023). Effects of transcranial magnetic stimulation of the rostromedial prefrontal cortex in obsessive–compulsive disorder: a randomized clinical trial. Nature Mental Health, 1(8), 555–563. https://doi.org/10.1038/s44220-023-00094-0
Cohan, R., Rafique, S. A., Stoby, K. S., Gorbet, D. J., & Steeves, J. K. E. (2023). Continuous and intermittent theta burst stimulation of primary visual cortex do not modulate resting state functional connectivity: A sham-controlled multi-echo fMRI study. Brain and Behavior: A Cognitive Neuroscience Perspective, 13(5), e2989. https://doi.org/10.1002/brb3.2989
Cole, E. J., Enticott, P. G., Oberman, L. M., Gwynette, M. F., Casanova, M. F., Jackson, S. L. J., . . . r, T. M. S. i. A. S. D. C. G. (2019). The Potential of Repetitive Transcranial Magnetic Stimulation for Autism Spectrum Disorder: A Consensus Statement. Biol Psychiatry, 85(4), e21-e22. https://doi.org/10.1016/j.biopsych.2018.06.003
Cordero-Grande, L., Christiaens, D., Hutter, J., Price, A. N., & Hajnal, J. V. (2019). Complex diffusion-weighted image estimation via matrix recovery under general noise models. NeuroImage, 200, 391–404. https://doi.org/10.1016/j.neuroimage.2019.06.039
Courchesne, E., Mouton, P. R., Calhoun, M. E., Semendeferi, K., Ahrens-Barbeau, C., Hallet, M. J., . . . Pierce, K. (2011). Neuron number and size in prefrontal cortex of children with autism. JAMA, 306(18), 2001–2010. https://doi.org/10.1001/jama.2011.1638
Desarkar, P., Rajji, T. K., Ameis, S. H., Blumberger, D. M., Lai, M. C., Lunsky, Y., & Daskalakis, Z. J. (2021). Assessing and stabilizing atypical plasticity in autism spectrum disorder using rTMS: Results from a proof-of-principle study. Clinical Neurophysiology. https://doi.org/10.1016/j.clinph.2021.03.046
Dhollander, T., Mito, R., Raffelt, D., & Connelly, A. (2019). Improved white matter response function estimation for 3-tissue constrained spherical deconvolution. In: Proc. Intl. Soc. Mag. Reson. Med. (p. 555). ISMRM.
Diedrichsen, J., Maderwald, S., Küper, M., Thürling, M., Rabe, K., Gizewski, E. R., . . . Timmann, D. (2011). Imaging the deep cerebellar nuclei: A probabilistic atlas and normalization procedure. Neuroimage, 54(3), 1786–1794. https://doi.org/10.1016/j.neuroimage.2010.10.035
Dimond, D., Schuetze, M., Smith, R. E., Dhollander, T., Cho, I., Vinette, S., . . . Bray, S. (2019). Reduced white matter fiber density in autism spectrum disorder. Cerebral Cortex, 29(4), 1778–1788. https://doi.org/10.1093/cercor/bhy348
Enticott, P. G., Fitzgibbon, B. M., Kennedy, H. A., Arnold, S. L., Elliot, D., Peachey, A., . . . Fitzgerald, P. B. (2014). A double-blind, randomized trial of deep repetitive transcranial magnetic stimulation (rTMS) for autism spectrum disorder. Brain Stimulation, 7(2), 206–211. https://doi.org/10.1016/j.brs.2013.10.004
Enticott, P. G., Barlow, K., Guastella, A. J., Licari, M. K., Rogasch, N. C., Middeldorp, C. M., . . . Fitzgerald, P. B. (2021). Repetitive transcranial magnetic stimulation (rTMS) in autism spectrum disorder: protocol for a multicentre randomised controlled clinical trial. BMJ Open, 11(7), e046830. https://doi.org/10.1136/bmjopen-2020-046830
Fatemi, S. H., Reutiman, T. J., Folsom, T. D., & Thuras, P. D. (2009). GABA(A) receptor downregulation in brains of subjects with autism. Journal of Autism and Developmental Disorders, 39(2), 223–230. https://doi.org/10.1007/s10803-008-0646-7
Fitzgerald, P. B., Hoy, K. E., Herring, S. E., McQueen, S., Peachey, A. V., Segrave, R. A., . . . Daskalakis, Z. J. (2012). A double blind randomized trial of unilateral left and bilateral prefrontal cortex transcranial magnetic stimulation in treatment resistant major depression. J Affect Disord, 139(2), 193–198. https://doi.org/10.1016/j.jad.2012.02.017
Fitzsimmons, S. M. D. D., Oostra, E., Postma, T. S., van der Werf, Y. D., & van den Heuvel, O. A. (2023). Repetitive Transcranial Magnetic Stimulation-Induced Neuroplasticity and the Treatment of Psychiatric Disorders: State of the Evidence and Future Opportunities. Biological Psychiatry. https://doi.org/10.1016/j.biopsych.2023.11.016
Friedman, N. P., & Robbins, T. W. (2022). The role of prefrontal cortex in cognitive control and executive function. Neuropsychopharmacology, 47(1), 72–89. https://doi.org/10.1038/s41386-021-01132-0
Godfrey, K. E. M., Muthukumaraswamy, S. D., Stinear, C. M., & Hoeh, N. (2022). Decreased salience network fMRI functional connectivity following a course of rTMS for treatment-resistant depression. Journal of Affective Disorders, 300, 235–242. https://doi.org/10.1016/j.jad.2021.12.129
Guo, Z., Jin, Y., Peng, H., Xing, G., Liao, X., Wang, Y., . . . Mu, Q. (2016). Ipsilesional High Frequency Repetitive Transcranial Magnetic Stimulation Add-On Therapy Improved Diffusion Parameters of Stroke Patients with Motor Dysfunction: A Preliminary DTI Study. Neural Plast, 2016, 6238575. https://doi.org/10.1155/2016/6238575
Hertrich, I., Dietrich, S., Blum, C., & Ackermann, H. (2021). The Role of the Dorsolateral Prefrontal Cortex for Speech and Language Processing. Frontiers in Human Neuroscience, 15, 645209. https://doi.org/10.3389/fnhum.2021.645209
Huang, Y. Z., Edwards, M. J., Rounis, E., Bhatia, K. P., & Rothwell, J. C. (2005). Thetaburst stimulation of the human motor cortex. Neuron, 45(2), 201–206. https://doi.org/10.1016/j.neuron.2004.12.033
Jannati, A., Ryan, M. A., Block, G., Kayarian, F. B., Oberman, L. M., Rotenberg, A., & Pascual-Leone, A. (2021). Modulation of motor cortical excitability by continuous theta-burst stimulation in adults with autism spectrum disorder. Clinical Neurophysiology, 132(7), 1647–1662. https://doi.org/10.1016/j.clinph.2021.03.021
Jannati, A., Block, G., Ryan, M. A., Kaye, H. L., Kayarian, F. B., Bashir, S., . . . Rotenberg, A. (2020). Continuous Theta-Burst Stimulation in Children With High-Functioning Autism Spectrum Disorder and Typically Developing Children. Front Integr Neurosci, 14, 13. https://doi.org/10.3389/fnint.2020.00013
Jenkinson, M., Beckmann, C. F., Behrens, T. E. J., Woolrich, M. W., & Smith, S. M. (2012). FSL. Neuroimage, 62(2), 782–790. https://doi.org/10.1016/j.neuroimage.2011.09.015
Jeurissen, B., Tournier, J. D., Dhollander, T., Connelly, A., & Sijbers, J. (2014). Multi-tissue constrained spherical deconvolution for improved analysis of multi-shell diffusion MRI data. NeuroImage, 103, 411–426. https://doi.org/10.1016/j.neuroimage.2014.07.061
Kellner, E., Dhital, B., Kiselev, V. G., & Reisert, M. (2016). Gibbs-ringing artifact removal based on local subvoxel-shifts. Magnetic Resonance in Medicine, 76(5), 1574–1581. https://doi.org/10.1002/mrm.26054
Kim, J., He, M. J., Widmann, A. K., & Lee, F. S. (2024). The role of neurotrophic factors in novel, rapid psychiatric treatments. Neuropsychopharmacology, 49(1), 227–245. https://doi.org/10.1038/s41386-023-01717-x
Kirkovski, M., Donaldson, P. H., Do, M., Speranza, B. E., Albein-Urios, N., Oberman, L. M., & Enticott, P. G. (2023a). A systematic review of the neurobiological effects of theta-burst stimulation (TBS) as measured using functional magnetic resonance imaging (fMRI). Brain Structure & Function, 228(3–4), 717–749. https://doi.org/10.1007/s00429-023-02634-x
Kirkovski, M., Fuelscher, I., Hyde, C., Donaldson, P. H., Ford, T. C., Rossell, S. L., . . . Enticott, P. G. (2020). Fixel based analysis reveals atypical white matter micro- and macrostructure in adults with autism spectrum disorder: An investigation of the role of biological sex. Frontiers in Integrative Neuroscience, 14. https://doi.org/10.3389/fnint.2020.00040
Kirkovski, M., Singh, M., Dhollander, T., Fuelscher, I., Hyde, C., Albein-Urios, N., . . . Enticott, P. G. (2023). An Investigation of Age-related Neuropathophysiology in Autism Spectrum Disorder Using Fixel-based Analysis of Corpus Callosum White Matter Micro- and Macrostructure. Journal of Autism and Developmental Disorders. https://doi.org/10.1007/s10803-023-05980-1
Kovalchuk, Y., Hanse, E., Kafitz, K. W., & Konnerth, A. (2002). Postsynaptic Induction of BDNF-Mediated Long-Term Potentiation. Science, 295(5560), 1729–1734. https://doi.org/10.1126/science.1067766
Lacadie, C. M., Fulbright, R. K., Rajeevan, N., Constable, R. T., & Papademetris, X. (2008). More accurate Talairach coordinates for neuroimaging using non-linear registration. NeuroImage, 42(2), 717–725. https://doi.org/10.1016/j.neuroimage.2008.04.240
Lasagna, C. A., Taylor, S. F., Lee, T. G., Rutherford, S., Greathouse, T., Gu, P., & Tso, I. F. (2021). Continuous Theta Burst Stimulation to the Secondary Visual Cortex at 80% Active Motor Threshold Does Not Impair Central Vision in Humans During a Simple Detection Task. Frontiers in Human Neuroscience, 15, 709275. https://doi.org/10.3389/fnhum.2021.709275
Lefaucheur, J. P., Aleman, A., Baeken, C., Benninger, D. H., Brunelin, J., Di Lazzaro, V., . . . Ziemann, U. (2020). Evidence-based guidelines on the therapeutic use of repetitive transcranial magnetic stimulation (rTMS): An update (2014–2018). Clin Neurophysiol, 131(2), 474–528. https://doi.org/10.1016/j.clinph.2019.11.002
Li, C. T., Huang, Y. Z., Bai, Y. M., Tsai, S. J., Su, T. P., & Cheng, C. M. (2019). Critical role of glutamatergic and GABAergic neurotransmission in the central mechanisms of theta-burst stimulation. Human Brain Mapping, 40(6), 2001–2009. https://doi.org/10.1002/hbm.24485
Li, X., Zhang, C., Tan, J., Ding, L., Wang, C., Wang, M., & Lin, Y. (2022). Clinical effects of continuous theta burst stimulation for generalized anxiety disorder and a mechanism involving alpha oscillations: a randomized controlled trial. Journal of Psychiatry & Neuroscience, 47(2), E123–E133. https://doi.org/10.1503/jpn.210134
Lord, C., Brugha, T. S., Charman, T., Cusack, J., Dumas, G., Frazier, T., . . . Veenstra-VanderWeele, J. (2020). Autism spectrum disorder. Nat Rev Dis Primers, 6(1), 5. https://doi.org/10.1038/s41572-019-0138-4
Lu, R., Zhang, C., Liu, Y., Wang, L., Chen, X., & Zhou, X. (2018). The effect of bilateral low-frequency rTMS over dorsolateral prefrontal cortex on serum brain-derived neurotropic factor and serotonin in patients with generalized anxiety disorder. Neuroscience Letters, 684, 67–71. https://doi.org/10.1016/j.neulet.2018.07.008
Masuda, F., Nakajima, S., Miyazaki, T., Yoshida, K., Tsugawa, S., Wada, M., . . . Noda, Y. (2019). Motor cortex excitability and inhibitory imbalance in autism spectrum disorder assessed with transcranial magnetic stimulation: a systematic review. Transl Psychiatry, 9(1), 110. https://doi.org/10.1038/s41398-019-0444-3
Matsuta, H., Shimomura, T., Kouchiyama, T., & Fujiki, M. (2022). Continuous theta-burst stimulation to the sensorimotor cortex affects contralateral gamma-aminobutyric acid level and resting-state networks. PLoS ONE, 17(8), e0272268. https://doi.org/10.1371/journal.pone.0272268
Moxon-Emre, I., Daskalakis, Z. J., Blumberger, D. M., Croarkin, P. E., Lyon, R. E., Forde, N. J., . . . Ameis, S. H. (2021). Modulation of Dorsolateral Prefrontal Cortex Glutamate/Glutamine Levels Following Repetitive Transcranial Magnetic Stimulation in Young Adults With Autism. Front Neurosci, 15, 711542. https://doi.org/10.3389/fnins.2021.711542
Mylius, V., Ayache, S. S., Ahdab, R., Farhat, W. H., Zouari, H. G., Belke, M., . . . Lefaucheur, J. P. (2013). Definition of DLPFC and M1 according to anatomical landmarks for navigated brain stimulation: inter-rater reliability, accuracy, and influence of gender and age. Neuroimage, 78, 224–232. https://doi.org/10.1016/j.neuroimage.2013.03.061
Nejati, V., Majdi, R., Salehinejad, M. A., & Nitsche, M. A. (2021). The role of dorsolateral and ventromedial prefrontal cortex in the processing of emotional dimensions. Science and Reports, 11(1), 1971. https://doi.org/10.1038/s41598-021-81454-7
Ni, H. C., Hung, J., Wu, C. T., Wu, Y. Y., Chang, C. J., Chen, R. S., & Huang, Y. Z. (2017). The Impact of Single Session Intermittent Theta-Burst Stimulation over the Dorsolateral Prefrontal Cortex and Posterior Superior Temporal Sulcus on Adults with Autism Spectrum Disorder. Frontiers in Neuroscience, 11, 255. https://doi.org/10.3389/fnins.2017.00255
Ni, H. C., Chen, Y. L., Chao, Y. P., Wu, C. T., Wu, Y. Y., Liang, S. H., . . . Lin, H. Y. (2021). Intermittent theta burst stimulation over the posterior superior temporal sulcus for children with autism spectrum disorder: A 4-week randomized blinded controlled trial followed by another 4-week open-label intervention. Autism, 25(5), 1279–1294. https://doi.org/10.1177/1362361321990534
Ni, H. C., Chao, Y. P., Tseng, R. Y., Wu, C. T., Cocchi, L., Chou, T. L., . . . Lin, H. Y. (2023a). Lack of effects of four-week theta burst stimulation on white matter macro/microstructure in children and adolescents with autism. Neuroimage Clin, 37, 103324. https://doi.org/10.1016/j.nicl.2023.103324
Ni, H. C., Chen, Y. L., Chao, Y. P., Wu, C. T., Chen, R. S., Chou, T. L., . . . Lin, H. Y. (2023b). A lack of efficacy of continuous theta burst stimulation over the left dorsolateral prefrontal cortex in autism: A double blind randomized sham-controlled trial. Autism Res, 16(6), 1247–1262. https://doi.org/10.1002/aur.2954
Ning, L., Rathi, Y., Barbour, T., Makris, N., & Camprodon, J. A. (2022). White matter markers and predictors for subject-specific rTMS response in major depressive disorder. Journal of Affective Disorders, 299, 207–214. https://doi.org/10.1016/j.jad.2021.12.005
Oberman, L. M., & Benussi, A. (2023). Transcranial magnetic stimulation across the lifespan: Impact of developmental and degenerative processes. Biological Psychiatry. https://doi.org/10.1016/j.biopsych.2023.07.012
Peng, H., Zheng, H., Li, L., Liu, J., Zhang, Y., Shan, B., . . . Zhang, Z. (2012). High-frequency rTMS treatment increases white matter FA in the left middle frontal gyrus in young patients with treatment-resistant depression. J Affect Disord, 136(3), 249–257. https://doi.org/10.1016/j.jad.2011.12.006
Pietropaolo, S., & Provenzano, G. (2022). Editorial: Targeting excitation-inhibition imbalance in neurodevelopmental and autism spectrum disorders. Frontiers in Neuroscience, 16, 968115. https://doi.org/10.3389/fnins.2022.968115
Raffelt, D. A., Tournier, J. D., Smith, R. E., Vaughan, D. N., Jackson, G., Ridgway, G. R., & Connelly, A. (2017). Investigating white matter fibre density and morphology using fixel-based analysis. NeuroImage, 144(Pt A), 58–73. https://doi.org/10.1016/j.neuroimage.2016.09.029
Raffelt, D. A., Smith, R. E., Ridgway, G. R., Tournier, J. D., Vaughan, D. N., Rose, S., . . . Connelly, A. (2015). Connectivity-based fixel enhancement: Whole-brain statistical analysis of diffusion MRI measures in the presence of crossing fibres. Neuroimage, 117, 40–55. https://doi.org/10.1016/j.neuroimage.2015.05.039
Sampaio-Baptista, C., & Johansen-Berg, H. (2017). White Matter Plasticity in the Adult Brain. Neuron, 96(6), 1239–1251. https://doi.org/10.1016/j.neuron.2017.11.026
Saturnino, G. B., Puonti, O., Nielsen, J. D., Antonenko, D., Madsen, K. H., & Thielscher, A. (2019). SimNIBS 2.1: A Comprehensive Pipeline for Individualized Electric Field Modelling for Transcranial Brain Stimulation. In S. Makarov, M. Horner, & G. Noetscher (Eds.), Brain and Human Body Modeling: Computational Human Modeling at EMBC 2018 (pp. 3–25). https://doi.org/10.1007/978-3-030-21293-3_1
Schaefer, A., Kong, R., Gordon, E. M., Laumann, T. O., Zuo, X.-N., Holmes, A. J., . . . Yeo, B. T. T. (2017). Local-Global Parcellation of the Human Cerebral Cortex from Intrinsic Functional Connectivity MRI. Cerebral Cortex, 28(9), 3095–3114. https://doi.org/10.1093/cercor/bhx179
Schilling, K. G., Blaber, J., Hansen, C., Cai, L., Rogers, B., Anderson, A. W., . . . Landman, B. A. (2020). Distortion correction of diffusion weighted MRI without reverse phase-encoding scans or field-maps. PLOS ONE, 15(7), e0236418. https://doi.org/10.1371/journal.pone.0236418
Schur, R. R., Draisma, L. W., Wijnen, J. P., Boks, M. P., Koevoets, M. G., Joels, M., . . . Vinkers, C. H. (2016). Brain GABA levels across psychiatric disorders: A systematic literature review and meta-analysis of (1) H-MRS studies. Hum Brain Mapp, 37(9), 3337–3352. https://doi.org/10.1002/hbm.23244
Schwippel, T., Schroeder, P. A., Fallgatter, A. J., & Plewnia, C. (2019). Clinical review: The therapeutic use of theta-burst stimulation in mental disorders and tinnitus. Progress in Neuro-Psychopharmacology and Biological Psychiatry, 92, 285–300. https://doi.org/10.1016/j.pnpbp.2019.01.014
Scott, D. N., & Frank, M. J. (2023). Adaptive control of synaptic plasticity integrates micro- and macroscopic network function. Neuropsychopharmacology, 48(1), 121–144. https://doi.org/10.1038/s41386-022-01374-6
Seewoo, B. J., Feindel, K. W., Won, Y., Joos, A. C., Figliomeni, A., Hennessy, L. A., & Rodger, J. (2022). White Matter Changes Following Chronic Restraint Stress and Neuromodulation: A Diffusion Magnetic Resonance Imaging Study in Young Male Rats. Biol Psychiatry Glob Open Sci, 2(2), 153–166. https://doi.org/10.1016/j.bpsgos.2021.08.006
Smith, R. E., Tournier, J.-D., Calamante, F., & Connelly, A. (2012). Anatomically-constrained tractography: Improved diffusion MRI streamlines tractography through effective use of anatomical information. NeuroImage, 62(3), 1924–1938. https://doi.org/10.1016/j.neuroimage.2012.06.005
Smith, R. E., Tournier, J.-D., Calamante, F., & Connelly, A. (2013). SIFT: Spherical-deconvolution informed filtering of tractograms. NeuroImage, 67, 298–312. https://doi.org/10.1016/j.neuroimage.2012.11.049
Smith, R. E., Tournier, J. D., Calamante, F., & Connelly, A. (2015). SIFT2: Enabling dense quantitative assessment of brain white matter connectivity using streamlines tractography. NeuroImage, 119, 338–351. https://doi.org/10.1016/j.neuroimage.2015.06.092
Sokhadze, E. M., El-Baz, A., Baruth, J., Mathai, G., Sears, L., & Casanova, M. F. (2009). Effects of low frequency repetitive transcranial magnetic stimulation (rTMS) on gamma frequency oscillations and event-related potentials during processing of illusory figures in autism. Journal of Autism and Developmental Disorders, 39(4), 619–634. https://doi.org/10.1007/s10803-008-0662-7
Sokhadze, E. M., El-Baz, A. S., Sears, L. L., Opris, I., & Casanova, M. F. (2014a). rTMS neuromodulation improves electrocortical functional measures of information processing and behavioral responses in autism. Frontiers in Systems Neuroscience, 8, 134. https://doi.org/10.3389/fnsys.2014.00134
Sokhadze, E. M., El-Baz, A. S., Tasman, A., Sears, L. L., Wang, Y., Lamina, E. V., & Casanova, M. F. (2014b). Neuromodulation integrating rTMS and neurofeedback for the treatment of autism spectrum disorder: An exploratory study. Applied Psychophysiology and Biofeedback, 39(3–4), 237–257. https://doi.org/10.1007/s10484-014-9264-7
Sokhadze, E. M., Lamina, E. V., Casanova, E. L., Kelly, D. P., Opris, I., Tasman, A., & Casanova, M. F. (2018). Exploratory Study of rTMS Neuromodulation Effects on Electrocortical Functional Measures of Performance in an Oddball Test and Behavioral Symptoms in Autism. Frontiers in Systems Neuroscience, 12, 20. https://doi.org/10.3389/fnsys.2018.00020
Sokhadze, E., Baruth, J., Tasman, A., Mansoor, M., Ramaswamy, R., Sears, L., . . . Casanova, M. F. (2010). Low-frequency repetitive transcranial magnetic stimulation (rTMS) affects event-related potential measures of novelty processing in autism. Applied Psychophysiology and Biofeedback, 35(2), 147–161. https://doi.org/10.1007/s10484-009-9121-2
Sokhadze, E. M., Baruth, J. M., Sears, L., Sokhadze, G. E., El-Baz, A. S., & Casanova, M. F. (2012). Prefrontal neuromodulation using rTMS improves error monitoring and correction function in autism. Applied Psychophysiology and Biofeedback, 37(2), 91–102. https://doi.org/10.1007/s10484-012-9182-5
Stoby, K. S., Rafique, S. A., Oeltzschner, G., & Steeves, J. K. E. (2022). Continuous and intermittent theta burst stimulation to the visual cortex do not alter GABA and glutamate concentrations measured by magnetic resonance spectroscopy. Brain and Behavior: A Cognitive Neuroscience Perspective, 12(2), e2478. https://doi.org/10.1002/brb3.2478
Tan, T., Wang, W., Xu, H., Huang, Z., Wang, Y. T., & Dong, Z. (2018). Low-Frequency rTMS Ameliorates Autistic-Like Behaviors in Rats Induced by Neonatal Isolation Through Regulating the Synaptic GABA Transmission. Frontiers in Cellular Neuroscience, 12, 46. https://doi.org/10.3389/fncel.2018.00046
Tateishi, H., Nishihara, M., Kawaguchi, A., Matsushima, J., Murakawa, T., Haraguchi, Y., . . . Monji, A. (2019). Improvement Of Frontal Lobe Dysfunction And White Matter Integrity By rTMS In Treatment-Resistant Depression. Neuropsychiatr Dis Treat, 15, 3079–3087. https://doi.org/10.2147/NDT.S228501
Tian, Y., Margulies, D. S., Breakspear, M., & Zalesky, A. (2020). Topographic organization of the human subcortex unveiled with functional connectivity gradients. Nature Neuroscience, 23(11), 1421–1432. https://doi.org/10.1038/s41593-020-00711-6
Tournier, J. D., Yeh, C. H., Calamante, F., Cho, K. H., Connelly, A., & Lin, C. P. (2008). Resolving crossing fibres using constrained spherical deconvolution: Validation using diffusion-weighted imaging phantom data. NeuroImage, 42(2), 617–625. https://doi.org/10.1016/j.neuroimage.2008.05.002
Tournier, J. D., Smith, R., Raffelt, D., Tabbara, R., Dhollander, T., Pietsch, M., . . . Connelly, A. (2019). MRtrix3: A fast, flexible and open software framework for medical image processing and visualisation. Neuroimage, 202, 116137. https://doi.org/10.1016/j.neuroimage.2019.116137
Tustison, N. J., Avants, B. B., Cook, P. A., Zheng, Y., Egan, A., Yushkevich, P. A., & Gee, J. C. (2010). N4ITK: Improved N3 Bias Correction. IEEE Transactions on Medical Imaging, 29(6), 1310–1320. https://doi.org/10.1109/TMI.2010.2046908
Ueda, R., Yamada, N., Abo, M., & Senoo, A. (2019). Relationship between motor function improvements and white matter structure after low-frequency repetitive transcranial magnetic stimulation plus intensive occupational therapy in chronic subcortical stroke patients. NeuroReport, 30(7), 485–490. https://doi.org/10.1097/WNR.0000000000001227
van Rooij, S. J. H., Arulpragasam, A. R., McDonald, W. M., & Philip, N. S. (2023). Accelerated TMS - moving quickly into the future of depression treatment. Neuropsychopharmacology. https://doi.org/10.1038/s41386-023-01599-z
Vekony, T., Nemeth, V. L., Holczer, A., Kocsis, K., Kincses, Z. T., Vecsei, L., & Must, A. (2018). Continuous theta-burst stimulation over the dorsolateral prefrontal cortex inhibits improvement on a working memory task. Science and Reports, 8(1), 14835. https://doi.org/10.1038/s41598-018-33187-3
Villafuerte, G., Miguel-Puga, A., & Arias-Carrion, O. (2019). Continuous Theta Burst Stimulation Over the Right Orbitofrontal Cortex Impairs Conscious Olfactory Perception. Frontiers in Neuroscience, 13, 555. https://doi.org/10.3389/fnins.2019.00555
Voineskos, A. N., Blumberger, D. M., Schifani, C., Hawco, C., Dickie, E. W., Rajji, T. K., . . . Daskalakis, Z. J. (2021). Effects of Repetitive Transcranial Magnetic Stimulation on Working Memory Performance and Brain Structure in People With Schizophrenia Spectrum Disorders: A Double-Blind, Randomized, Sham-Controlled Trial. Biol Psychiatry Cogn Neurosci Neuroimaging, 6(4), 449–458. https://doi.org/10.1016/j.bpsc.2020.11.011
Walsh, M. J. M., Wallace, G. L., Gallegos, S. M., & Braden, B. B. (2021). Brain-based sex differences in autism spectrum disorder across the lifespan: A systematic review of structural MRI, fMRI, and DTI findings. NeuroImage: Clinical, 31, 102719. https://doi.org/10.1016/j.nicl.2021.102719
Yamada, N., Ueda, R., Kakuda, W., Momosaki, R., Kondo, T., Hada, T., . . . Abo, M. (2018). Diffusion Tensor Imaging Evaluation of Neural Network Development in Patients Undergoing Therapeutic Repetitive Transcranial Magnetic Stimulation following Stroke. Neural Plast, 2018, 3901016. https://doi.org/10.1155/2018/3901016
Yang, Y., & Calakos, N. (2013). Presynaptic Long-Term Plasticity. Front Synaptic Neurosci, 5, 8. https://doi.org/10.3389/fnsyn.2013.00008
Yang, W., Liu, T. T., Song, X. B., Zhang, Y., Li, Z. H., Cui, Z. H., . . . Liu, J. (2015). Comparison of different stimulation parameters of repetitive transcranial magnetic stimulation for unilateral spatial neglect in stroke patients. J Neurol Sci, 359(1–2), 219–225. https://doi.org/10.1016/j.jns.2015.08.1541
Yeh, C.-H., Smith, R. E., Dhollander, T., Calamante, F., & Connelly, A. (2019). Connectomes from streamlines tractography: Assigning streamlines to brain parcellations is not trivial but highly consequential. NeuroImage, 199, 160–171. https://doi.org/10.1016/j.neuroimage.2019.05.005
Yeh, C.-H., Tseng, R.-Y., Ni, H.-C., Cocchi, L., Chang, J.-C., Hsu, M.-Y., . . . Lin, H.-Y. (2022). White matter microstructural and morphometric alterations in autism: implications for intellectual capabilities. Molecular Autism, 13(1), 21. https://doi.org/10.1186/s13229-022-00499-1
Zalesky, A., Fornito, A., & Bullmore, E. T. (2010). Network-based statistic: Identifying differences in brain networks. NeuroImage, 53(4), 1197–1207. https://doi.org/10.1016/j.neuroimage.2010.06.041
Zhao, X., Li, Y., Tian, Q., Zhu, B., & Zhao, Z. (2019). Repetitive transcranial magnetic stimulation increases serum brain-derived neurotrophic factor and decreases interleukin-1β and tumor necrosis factor-α in elderly patients with refractory depression. Journal of International Medical Research, 47(5), 1848–1855. https://doi.org/10.1177/0300060518817417
Acknowledgements
The authors would like to thank all of our participants and their family members for partaking in this study and the anonymous reviewers for comments that significantly improved the manuscript. We want to pay tribute to the late Professor Ying-Zu Huang, who guided and inspired this study with wisdom, insight, and tenacity.
Funding
This work is supported by grants from the Ministry of Science and Technology of Taiwan (108–2628-B-182A-006-; 109–2628-B-182A-010-; 110–2628-B-182A-016-; 112–2221-E-182–029). Hsiang-Yuan Lin is supported by the Azrieli Adult Neurodevelopmental Centre at Centre for Addiction and Mental Health, and an Academic Scholar Award from the Department of Psychiatry, University of Toronto. The funders had no role in the study's design, in the collection, analyses, or interpretation of data, in the writing of the manuscript, or in the decision to publish the results.
Author information
Authors and Affiliations
Contributions
All authors read and approved the final manuscript. Conceptualization and design: Hsing-Chang Ni, Hsiang-Yuan Lin; Methodology: Yi-Ping Chao, Chen-Te Wu, Rou-Shayn Chen, Chun-Hung Yeh; Clinical data collection: Hsing-Chang Ni; Neuroimaging data collection: Hsing-Chang Ni, Yi-Ping Chao, Chen-Te Wu; Formal analysis: Hsiang-Chang Ni, Rung-Yu Tseng, Chun-Hung Yeh, Hsiang-Yuan Lin; Interpretation: Hsiang-Chang Ni, Po-Chun Lin, Chun-Hung Yeh, and Hsiang-Yuan Lin; Writing—original draft preparation: Hsiang-Chang Ni, Po-Chun Lin, Chun-Hung Yeh, and Hsiang-Yuan Lin; Figures: Hsiang-Chang Ni, Rung-Yu Tseng, Chun-Hung Yeh; Writing—review and editing: All authors; Funding acquisition: Hsing-Chang Ni; Resources: Tai-Li Chou and Susan Shur-Fen Gau; Supervision: Hsiang-Yuan Lin.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. The Research Ethics Committee approved the study at Chang Gung Memorial Hospital, Linkou, Taiwan (201802246A0), and registered with ClinicalTrials.gov (NCT04993144).
Consent to participate
All participants provided informed consent.
Consent for publication
All participants provided informed consent to publish the data.
Conflict of interests
All authors have no financial or non-financial interests relevant to the content of this article to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yeh, CH., Lin, PC., Tseng, RY. et al. Lack of effects of eight-week left dorsolateral prefrontal theta burst stimulation on white matter macro/microstructure and connection in autism. Brain Imaging and Behavior (2024). https://doi.org/10.1007/s11682-024-00874-x
Accepted:
Published:
DOI: https://doi.org/10.1007/s11682-024-00874-x