Abstract
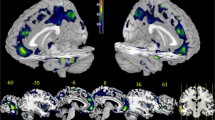
Gray matter (GM) atrophy is well documented in patients with major depressive disorder (MDD), but its underlying mechanism remains unknown. This study aimed to examine the GM atrophy in MDD patients with diverse suicidal ideations (SIs) and to explore whether those alterations were driven by connections. GM volume was estimated in 163 patients with recurrent MDD (comprising 122 with SI [MDDSI] and 41 without SI [MDDNSI]) and 134 health controls (HCs). A two-sample t-test was used to identify GM volume abnormalities in MDD patients and their subgroups. Functional connectivity was computed between pairs of aberrant GM in both patients and HCs, which were further compared with the connectivity of random brain regions. A permutation test was performed to assess its significance. Propensity score matching (PSM) was further performed to validate the main results. Compared with HCs, the MDDNSI group exhibited GM atrophy in 24 regions, with the largest effect sizes found in the frontal and parietal lobes, while the MDDSI group exhibited more widespread GM atrophy involving 49 regions, with the largest effect sizes in the frontal lobe, parietal lobe, temporal lobe, and the limbic system. Furthermore, patients and HCs exhibited significantly increased functional connectivity between regions with GM atrophy compared with randomly selected regions (p < 0.05). PSM analysis presented similar results to the main analysis. MDD patients had diverse GM atrophy features according to their SI tendency. Moreover, connectome architecture modulates the GM atrophy in MDD patients, implying the possibility that connections drive these pathological changes.


Similar content being viewed by others
Data availability
The deidentified data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy and ethical restrictions.
References
Amidfar, M., Quevedo, J., & Kim, Y. K. (2021). Grey matter volume abnormalities in the first depressive episode of medication-naive adult individuals: A systematic review of Voxel based morphometric studies. International Journal of Psychiatry in Clinical Practice, 25(4), 407–420. https://doi.org/10.1080/13651501.2020.1861632
Avena-Koenigsberger, A., Misic, B., & Sporns, O. (2017). Communication dynamics in complex brain networks. Nature Reviews Neuroscience, 19(1), 17–33. https://doi.org/10.1038/nrn.2017.149
Bora, E., Fornito, A., Pantelis, C., & Yucel, M. (2012). Gray Matter abnormalities in major depressive disorder: A meta-analysis of Voxel based morphometry studies. Journal of Affective Disorders, 138(1–2), 9–18. https://doi.org/10.1016/j.jad.2011.03.049
Cauda, F., Nani, A., Manuello, J., Premi, E., Palermo, S., Tatu, K., & Costa, T. (2018). Brain structural alterations are distributed following functional, anatomic and genetic connectivity. Brain, 141(11), 3211–3232. https://doi.org/10.1093/brain/awy252
Chao-Gan, Y., & Yu-Feng, Z. (2010). DPARSF: A MATLAB toolbox for pipeline data analysis of resting-state fMRI. Frontiers in Systems Neuroscience, 4,. https://doi.org/10.3389/fnsys.2010.00013
Chen, L., Wang, Y., Niu, C., Zhong, S., Hu, H., Chen, P., & Huang, R. (2018). Common and distinct abnormal frontal-limbic system structural and functional patterns in patients with major depression and bipolar disorder. Neuroimage Clinical, 20, 42–50. https://doi.org/10.1016/j.nicl.2018.07.002
Chen, Z., Xia, M., Zhao, Y., Kuang, W., Jia, Z., & Gong, Q. (2021). Characteristics of intrinsic brain functional connectivity alterations in major depressive disorder patients with Suicide behavior. Journal of Magnetic Resonance Imaging, 54(6), 1867–1875. https://doi.org/10.1002/jmri.27784
Ciric, R., Wolf, D. H., Power, J. D., Roalf, D. R., Baum, G. L., Ruparel, K., & Satterthwaite, T. D. (2017). Benchmarking of participant-level confound regression strategies for the control of motion artifact in studies of functional connectivity. NeuroImage, 154, 174–187. https://doi.org/10.1016/j.neuroimage.2017.03.020
Di Biase, M. A., Cropley, V. L., Baune, B. T., Olver, J., Amminger, G. P., Phassouliotis, C., & Zalesky, A. (2017). White matter connectivity disruptions in early and chronic schizophrenia. Psychological Medicine, 47(16), 2797–2810. https://doi.org/10.1017/S0033291717001313
Eickhoff, S. B., Yeo, B. T. T., & Genon, S. (2018). Imaging-based parcellations of the human brain. Nature Reviews Neuroscience, 19(11), 672–686. https://doi.org/10.1038/s41583-018-0071-7
Fornito, A., Zalesky, A., & Breakspear, M. (2015). The connectomics of brain disorders. Nature Reviews Neuroscience, 16(3), 159–172. https://doi.org/10.1038/nrn3901
Fries, P. (2015). Rhythms for cognition: Communication through coherence. Neuron, 88(1), 220–235. https://doi.org/10.1016/j.neuron.2015.09.034
Guo, W., Liu, F., Liu, J., Yu, L., Zhang, Z., Zhang, J., & Xiao, C. (2013). Is there a cerebellar compensatory effort in first-episode, treatment-naive major depressive disorder at rest? Progress in Neuropsychopharmacology and Biological Psychiatry, 46, 13–18. https://doi.org/10.1016/j.pnpbp.2013.06.009
Hamilton, M. (1960). A rating scale for depression. Journal of Neurology, Neurosurgery and Psychiatry, 23, 56–62. https://doi.org/10.1136/jnnp.23.1.56
Harenski, C. L., Harenski, K. A., Calhoun, V. D., & Kiehl, K. A. (2020). Source-based morphometry reveals gray matter differences related to suicidal behavior in criminal offenders. Brain Imaging and Behavior, 14(1), 1–9. https://doi.org/10.1007/s11682-018-9957-2
He, M., Ping, L., Chu, Z., Zeng, C., Shen, Z., & Xu, X. (2022). Identifying changes of brain regional homogeneity and cingulo-opercular network connectivity in first-episode, drug-naive depressive patients with suicidal ideation. Frontiers in Neuroscience, 16, 856366. https://doi.org/10.3389/fnins.2022.856366
Jiang, Y., Wang, Y., Huang, H., He, H., Tang, Y., Su, W., & Luo, C. (2022). Antipsychotics effects on network-level reconfiguration of cortical morphometry in First-Episode Schizophrenia. Schizophrenia Bulletin, 48(1), 231–240. https://doi.org/10.1093/schbul/sbab082
Jucker, M., & Walker, L. C. (2018). Propagation and spread of pathogenic protein assemblies in neurodegenerative Diseases. Nature Neuroscience, 21(10), 1341–1349. https://doi.org/10.1038/s41593-018-0238-6
Kelly, S., Jahanshad, N., Zalesky, A., Kochunov, P., Agartz, I., Alloza, C., & Donohoe, G. (2018). Widespread white matter microstructural differences in schizophrenia across 4322 individuals: Results from the ENIGMA Schizophrenia DTI Working Group. Molecular Psychiatry, 23(5), 1261–1269. https://doi.org/10.1038/mp.2017.170
Liu, X., He, C., Fan, D., Zang, F., Zhu, Y., Zhang, H., & Xie, C. (2021). Alterations of core structural network connectome associated with suicidal ideation in major depressive disorder patients. Translational Psychiatry, 11(1), 243. https://doi.org/10.1038/s41398-021-01353-3
Liu, Z., Rolls, E. T., Liu, Z., Zhang, K., Yang, M., Du, J., & Feng, J. (2019). Brain annotation toolbox: Exploring the functional and genetic associations of neuroimaging results. Bioinformatics, 35(19), 3771–3778. https://doi.org/10.1093/bioinformatics/btz128
Murphy, K., & Fox, M. D. (2017). Towards a consensus regarding global signal regression for resting state functional connectivity MRI. NeuroImage, 154, 169–173. https://doi.org/10.1016/j.neuroimage.2016.11.052
Nakano, M., Matsuo, K., Nakashima, M., Matsubara, T., Harada, K., Egashira, K., & Watanabe, Y. (2014). Gray matter volume and rapid decision-making in major depressive disorder. Progress in Neuropsychopharmacology and Biological Psychiatry, 48, 51–56. https://doi.org/10.1016/j.pnpbp.2013.09.011
Power, J. D., Barnes, K. A., Snyder, A. Z., Schlaggar, B. L., & Petersen, S. E. (2012). Spurious but systematic correlations in functional connectivity MRI networks arise from subject motion. NeuroImage, 59(3), 2142–2154. https://doi.org/10.1016/j.neuroimage.2011.10.018
Renteria, M. E., Schmaal, L., Hibar, D. P., Couvy-Duchesne, B., Strike, L. T., Mills, N. T., & Hickie, I. B. (2017). Subcortical brain structure and suicidal behaviour in major depressive disorder: A meta-analysis from the ENIGMA-MDD working group. Translational Psychiatry, 7(5), e1116. https://doi.org/10.1038/tp.2017.84
Rizk, M. M., Rubin-Falcone, H., Lin, X., Keilp, J. G., Miller, J. M., Milak, M. S., & Mann, J. J. (2019). Gray matter volumetric study of major depression and suicidal behavior. Psychiatry Research Neuroimaging, 283, 16–23. https://doi.org/10.1016/j.pscychresns.2018.11.007
Shafiei, G., Markello, R. D., Makowski, C., Talpalaru, A., Kirschner, M., Devenyi, G. A., & Misic, B. (2020). Spatial patterning of tissue volume loss in Schizophrenia reflects Brain Network Architecture. Biological Psychiatry, 87(8), 727–735. https://doi.org/10.1016/j.biopsych.2019.09.031
Shen, Z., Cheng, Y., Yang, S., Dai, N., Ye, J., Liu, X., & Xu, X. (2016). Changes of grey matter volume in first-episode drug-naive adult major depressive disorder patients with different age-onset. Neuroimage Clinical, 12, 492–498. https://doi.org/10.1016/j.nicl.2016.08.016
Staffa, S. J., & Zurakowski, D. (2018). Five steps to successfully implement and evaluate propensity score matching in Clinical Research studies. Anesthesia and Analgesia, 127(4), 1066–1073. https://doi.org/10.1213/ANE.0000000000002787
Tsypes, A., Burkhouse, K. L., & Gibb, B. E. (2016). Classification of facial expressions of emotion and risk for suicidal ideation in children of depressed mothers: Evidence from cross-sectional and prospective analyses. Journal of Affective Disorders, 197, 147–150. https://doi.org/10.1016/j.jad.2016.03.037
Tzourio-Mazoyer, N., Landeau, B., Papathanassiou, D., Crivello, F., Etard, O., Delcroix, N., & Joliot, M. (2002). Automated anatomical labeling of activations in SPM using a macroscopic anatomical parcellation of the MNI MRI single-subject brain. NeuroImage, 15(1), 273–289. https://doi.org/10.1006/nimg.2001.0978
Wannan, C. M. J., Cropley, V. L., Chakravarty, M. M., Bousman, C., Ganella, E. P., Bruggemann, J. M., & Zalesky, A. (2019). Evidence for Network-based cortical thickness reductions in Schizophrenia. American Journal of Psychiatry, 176(7), 552–563. https://doi.org/10.1176/appi.ajp.2019.18040380
Warren, J. D., Rohrer, J. D., Schott, J. M., Fox, N. C., Hardy, J., & Rossor, M. N. (2013). Molecular nexopathies: A new paradigm of neurodegenerative Disease. Trends in Neurosciences, 36(10), 561–569. https://doi.org/10.1016/j.tins.2013.06.007
Watanabe, K., Kakeda, S., Yoshimura, R., Abe, O., Ide, S., Hayashi, K., & Korogi, Y. (2015). Relationship between the catechol-O-methyl transferase Val108/158Met genotype and brain volume in treatment-naive major depressive disorder: Voxel-based morphometry analysis. Psychiatry Research, 233(3), 481–487. https://doi.org/10.1016/j.pscychresns.2015.07.024
Wise, T., Radua, J., Via, E., Cardoner, N., Abe, O., Adams, T. M., & Arnone, D. (2017). Common and distinct patterns of grey-matter volume alteration in major depression and bipolar disorder: Evidence from voxel-based meta-analysis. Molecular Psychiatry, 22(10), 1455–1463. https://doi.org/10.1038/mp.2016.72
Zhang, R., Wei, S., Chang, M., Jiang, X., Tang, Y., & Wang, F. (2020a). Dorsolateral and ventrolateral prefrontal cortex structural changes relative to suicidal ideation in patients with depression. Acta Neuropsychiatr, 32(2), 84–91. https://doi.org/10.1017/neu.2019.45
Zhang, T., Zhao, B., Shi, C., Nie, B., Liu, H., Yang, X., … Shan, B. (2020b). Subthreshold depression may exist on a spectrum with major depressive disorder: Evidence from gray matter volume and morphological brain network. Journal of Affective Disorders, 266, 243–251. https://doi.org/10.1016/j.jad.2020.01.135
Zheng, R., Zhang, Y., Yang, Z., Han, S., & Cheng, J. (2021). Reduced Brain Gray Matter volume in patients with First-Episode Major depressive disorder: A quantitative Meta-analysis. Frontiers in Psychiatry, 12,. https://doi.org/10.3389/fpsyt.2021.671348
Acknowledgements
We thank LetPub (https://www.letpub.com) for its linguistic assistance during the preparation of this manuscript.
Funding
This study has received funding by Natural Scientific Foundation of China (grant numbers: 81560283 and 81201084) and Guangdong Basic and Applied Basic Research Foundation (grant numbers: 2022A1515012503), the Shenzhen Key Medical Discipline Construction Fund (No.SZXK041), the Shenzhen Fund for Guangdong Provincial High-level Clinical Key Specialties (No. SZGSP013) and Nanshan District Health System Technology Major Project (NSZD2023025), and Scientific and Technological Projects of Nanshan (NS2022007).
Author information
Authors and Affiliations
Contributions
Conception and study design (Yingwei Qiu and Gangqiang Hou), data collection or acquisition (Yingli Zhang, Ziyun Xu, Yanqing Li and Manxi Xu), statistical analysis (Shengli Chen and Shiwei Lin), interpretation of results (Shengli Chen and Xiaojing Zhang), drafting the manuscript work or revising it critically for important intellectual content (Shengli Chen, Yingli Zhang, Gangqiang Hou and Yingwei Qiu) and approval of final version to be published and agreement to be accountable for the integrity and accuracy of all aspects of the work (All authors).
Corresponding authors
Ethics declarations
Ethical approval
This retrospective study was approved by the local Research Ethics Committee. Written informed consent was obtained from each participant.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 4.59 MB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, S., Zhang, X., Lin, S. et al. Connectome architecture modulates the gray matter atrophy in major depression disorder patients with diverse suicidal ideations. Brain Imaging and Behavior (2023). https://doi.org/10.1007/s11682-023-00826-x
Accepted:
Published:
DOI: https://doi.org/10.1007/s11682-023-00826-x