Abstract
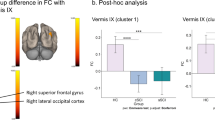
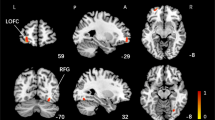
The objective of this study was to investigate the alterations of brain activation and effective connectivity during motor imagery (MI) in complete spinal cord injury (CSCI) patients and to reveal a potential mechanism of MI in motor rehabilitation of CSCI patients. Fifteen CSCI patients and twenty healthy controls underwent the MI task-related fMRI scan, and the motor execution (ME) task only for healthy controls. The brain activation patterns of the two groups during MI, and CSCI patients during the MI task and healthy controls during the ME task were compared. Then the significantly changed brain activation areas in CSCI patients during the MI task were used as regions of interest for effective connectivity analysis, using a voxel-wise granger causality analysis (GCA) method. Compared with healthy controls, increased activations in left primary sensorimotor cortex and bilateral cerebellar lobules IV-VI were detected in CSCI patients during the MI task, and the activation level of these areas even equaled that of healthy controls during the ME task. Furthermore, GCA revealed decreased effective connectivity from sensorimotor related areas (primary sensorimotor cortex and cerebellar lobules IV-VI) to cognitive related areas (prefrontal cortex, precuneus, middle temporal gyrus, and inferior temporal gyrus) in CSCI patients. Our findings demonstrated that motor related brain areas can be functionally preserved and activated through MI after CSCI, it maybe the potential mechanism of MI in the motor rehabilitation of CSCI patients. In addition, Sensorimotor related brain regions have less influence on the cognitive related regions in CSCI patients during MI (The trial registration number: ChiCTR2000032793).




Similar content being viewed by others
Data availability
Research data is confidential given that we do not have participant approval to share data with outside investigators.
Code availability
Not applicable.
Abbreviations
- ADF:
-
Ankle dorsiflexion
- ASIA:
-
American Spinal Injury Association
- EC:
-
Effective connectivity
- FC:
-
Functional connectivity
- fMRI:
-
Functional magnetic resonance imaging
- GCA:
-
Granger causality analysis
- HCs:
-
Healthy controls
- IFGtriang:
-
Triangular part of inferior frontal gyrus
- IPL:
-
Inferior parietal lobule
- ITG:
-
Inferior temporal gyrus
- KVIQ:
-
Kinesthetic and Visual Imagery Questionnaire
- M1:
-
Precentral gyrus
- ME:
-
Motor execution
- MI:
-
Motor imagery
- MOG:
-
Middle occipital gyrus
- MRI:
-
Magnetic resonance imaging
- MTG:
-
Middle temporal gyrus
- oMFC:
-
Orbital region of the medial prefrontal cortex
- PCL:
-
Paracentral lobule
- PSMC:
-
Primary sensorimotor cortex
- ROI:
-
Region of interest
- S1:
-
Postcentral gyrus
- SCI:
-
Spinal cord injury
- SMA:
-
Supplementary motor area
- SMG:
-
Supramarginal
References
Alkadhi, H., Brugger, P., Boendermaker, S. H., Crelier, G., Curt, A., Hepp-Reymond, M. C., & Kollias, S. S. (2005). What Disconnection Tells about Motor Imagery: Evidence from Paraplegic Patients. Cerebral Cortex, 15, 131–140. https://doi.org/10.1093/cercor/bhh116
Battaglia, C., D’Artibale, E., Fiorilli, G., Piazza, M., Tsopani, D., Giombini, A., Calcagno, G., & di Cagno, A. (2014). Use of video observation and motor imagery on jumping performance in national rhythmic gymnastics athletes. Human Movement Science. https://doi.org/10.1016/j.humov.2014.10.001
Bostan, A. C., Dum, R. P., & Strick, P. L. (2013). Cerebellar networks with the cerebral cortex and basal ganglia. Trends in Cognitive Sciences, 17, 241–254. https://doi.org/10.1016/j.tics.2013.03.003
Brown, R. M., & Palmer, C. (2013). Auditory and motor imagery modulate learning in music performance. Frontiers in Human Neuroscience, 7, 320. https://doi.org/10.3389/fnhum.2013.00320
Butler, A.J., Cazeaux, J., Fidler, A., Jansen, J., Lefkove, N., Gregg, M., Hall, C., Easley, K.A., Shenvi, N., & Wolf SL. (2012). The movement imagery Questionnaire-Revised, second edition (MIQ-RS) is a reliable and valid tool for evaluating motor imagery in stroke populations. Evidence-Based Complementary and Alternative Medicine, 2012, 1-11. https://doi.org/10.1155/2012/497289
Chen, Q., Zheng, W., Chen, X., Li, X., Wang, L., Qin, W., Li, K., & Chen, N. (2019). Reorganization of the somatosensory pathway after subacute incomplete cervical cord injury. NeuroImage: Clinical, 21, 101674. https://doi.org/10.1016/j.nicl.2019.101674
Chen, X., Wan, L., Qin, W., Zheng, W., Qi, Z., Chen, N., & Li, K. (2016). Functional preservation and reorganization of brain during motor imagery in patients with incomplete spinal cord injury: A pilot fMRI study. Frontiers in Human Neuroscience, 10. 10.3389/ fnhum.2016.00046
Cramer, S. C., Lastra, L., Lacourse, M. G., & Cohen, M. J. (2005). Brain motor system function after chronic, complete spinal cord injury. Brain, 128, 2941–2950. https://doi.org/10.1093/brain/awh648
Cramer, S. C., Orr, E. L. R., Cohen, M. J., & Lacourse, M. G. (2007). Effects of motor imagery training after chronic, complete spinal cord injury. Experimental Brain Research, 177, 233–242. https://doi.org/10.1007/s00221-006-0662-9
de Lange, F. P., Roelofs, K., & Toni, I. (2008). Motor imagery: A window into the mechanisms and alterations of the motor system. Cortex, 44, 494–506. https://doi.org/10.1016/j.cortex.2007.09.002
Di Rienzo, F., Joassy, P., Kanthack, T., MacIntyre, T. E., Debarnot, U., Blache, Y., Hautier, C., Collet, C., & Guillot, A. (2019). Effects of action observation and action observation combined with motor imagery on maximal isometric strength. Neuroscience, 418, 82–95. https://doi.org/10.1016/j.neuroscience.2019.08.025
Enzinger, C., Ropele, S., Fazekas, F., Loitfelder, M., Gorani, F., Seifert, T., Reiter, G., Neuper, C., Pfurtscheller, G., & Müller-Putz, G. (2008). Brain motor system function in a patient with complete spinal cord injury following extensive brain–computer interface training. Experimental Brain Research, 190, 215–223. https://doi.org/10.1007/s00221-008-1465-y
Errante, A., Bozzetti, F., Sghedoni, S., Bressi, B., & Fogassi, L. (2019). Explicit motor imagery for grasping actions in children with spastic unilateral cerebral palsy. Frontiers in Neurology, 10. https://doi.org/10.3389/fneur.2019.00837
Fekih, S., Zguira, M.S., Koubaa, A., Masmoudi, L., Bragazzi, N.L., & Jarraya, M. (2020). Effects of Motor Mental Imagery Training on Tennis Service Performance during the Ramadan Fasting: A Randomized, Controlled Trial. Nutrients, 12. https://doi.org/10.3390/nu12041035
Feng, N., Hu, F., Wang, H., & Gouda, M. A. (2020). Decoding of voluntary and involuntary upper-limb motor imagery based on graph fourier transform and cross-frequency coupling coefficients. Journal of Neural Engineering, 17, 56043. https://doi.org/10.1088/1741-2552/abc024
Frässle, S., Manjaly, Z. M., Do, C. T., Kasper, L., Pruessmann, K. P., & Stephan, K. E. (2021). Whole-brain estimates of directed connectivity for human connectomics. NeuroImage, 225, 117491. https://doi.org/10.1016/j.neuroimage.2020.117491
Friston, K. J. (2011). Functional and effective connectivity: A review. Brain Connectivity, 1, 13–36. https://doi.org/10.1089/brain.2011.0008
Gao, Q., Duan, X., & Chen, H. (2011). Evaluation of effective connectivity of motor areas during motor imagery and execution using conditional Granger causality. NeuroImage, 54, 1280–1288. https://doi.org/10.1016/j.neuroimage.2010.08.071
González, B., Rodríguez, M., Ramirez, C., & Sabaté, M. (2005). Disturbance of motor imagery after cerebellar stroke. Behavioral Neuroscience, 119, 622–626. https://doi.org/10.1037/0735-7044.119.2.622
Grangeon, M., Revol, P., Guillot, A., Rode, G., & Collet, C. (2012). Could motor imagery be effective in upper limb rehabilitation of individuals with spinal cord injury? A case study. Spinal Cord, 50, 766–771. https://doi.org/10.1038/sc.2012.41
Grealy, M. A., & Lee, D. N. (2011). An automatic-voluntary dissociation and mental imagery disturbance following a cerebellar lesion. Neuropsychologia, 49, 271–275. https://doi.org/10.1016/j.neuropsychologia.2010.09.031
Grush, R. (2004). The emulation theory of representation: Motor control, imagery, and perception. The Behavioral and Brain Sciences, 27(377–396), 396–442. https://doi.org/10.1017/s0140525x04000093
Hétu, S., Grégoire, M., Saimpont, A., Coll, M., Eugène, F., Michon, P. E., & Jackson, P. L. (2013). The neural network of motor imagery: An ALE meta-analysis. Neuroscience & Biobehavioral Reviews, 37, 930–949. https://doi.org/10.1016/j.neubiorev.2013.03.017
Hotz-Boendermaker, S., Funk, M., Summers, P., Brugger, P., Hepp-Reymond, M. C., Curt, A., & Kollias, S. S. (2008). Preservation of motor programs in paraplegics as demonstrated by attempted and imagined foot movements. NeuroImage, 39, 383–394. https://doi.org/10.1016/j.neuroimage.2007.07.065
Karunakaran, K. D., Yuan, R., He, J., Zhao, J., Cui, J. L., Zang, Y. F., Zhang, Z., Alvarez, T. L., & Biswal, B. B. (2020). Resting-State functional connectivity of the thalamus in complete spinal cord injury. Neurorehabilitation and Neural Repair, 34, 122–133. https://doi.org/10.1177/1545968319893299
Kelly, R. M., & Strick, P. L. (2003). Cerebellar loops with motor cortex and prefrontal cortex of a nonhuman primate. The Journal of Neuroscience, 23, 8432–8444. https://doi.org/10.1523/jneurosci.23-23-08432.2003
Killgore, W. D., Olson, E. A., & Weber, M. (2013). Physical exercise habits correlate with gray matter volume of the hippocampus in healthy adult humans. Scientific Reports-UK, 3, 3457. https://doi.org/10.1038/srep03457
Kirk-Sanchez, N. J., & McGough, E. L. (2014). Physical exercise and cognitive performance in the elderly: Current perspectives. Clinical Interventions in Aging, 9, 51–62. https://doi.org/10.2147/CIA.S39506
Krüger, B., Hettwer, M., Zabicki, A., de Haas, B., & Munzert, J., et al. (2020). Practice modality of motor sequences impacts the neural signature of motor imagery. Scientific Reports, 10. https://doi.org/10.1038/s41598-020-76214-y
Laure, C.D., Alexandra, P., Sylvie, B., Frédéric, B., & Gilles, K. (2018). Effects of simultaneous aerobic and cognitive training on executive functions, cardiovascular fitness and functional abilities in older adults with mild cognitive impairment. Mental Health & Physical Activity, S1755296617301114. https://doi.org/10.1016/j.mhpa.2018.06.001.
Levy, L. M., Ziemann, U., Chen, R., & Cohen, L. G. (2002). Rapid modulation of GABA in sensorimotor cortex induced by acute deafferentation. Annals of Neurology, 52, 755–761. https://doi.org/10.1002/ana.10372
Li, F., Zhang, T., Li, B. J., Zhang, W., Zhao, J., & Song, L. P. (2018). Motor imagery training induces changes in brain neural networks in stroke patients. Neural Regeneration Research, 13, 1771–1781. https://doi.org/10.4103/1673-5374.238616
Li, H., Hu, X., Gao, Y., Cao, L., Zhang, L., Bu, X., Lu, L., Wang, Y., Tang, S., Li, B., Yang, Y., Biswal, B.B., Gong, Q., & Huang, X. (2020a). Neural primacy of the dorsolateral prefrontal cortex in patients with obsessive-compulsive disorder. NeuroImage: Clinical, 28. 10.1016/ j.nicl. 2020a.102432
Li, X., Wang, L., Chen, Q., Hu, Y., Du, J., et al. (2020b). The reorganization of insular subregions in individuals with Below-Level neuropathic pain following incomplete spinal cord injury. Neural Plasticity, 2020, 1–9. https://doi.org/10.1155/2020/2796571
Lorey, B., Pilgramm, S., Walter, B., Stark, R., Munzert, J., & Zentgraf, K. (2010). Your mind’s hand: Motor imagery of pointing movements with different accuracy. NeuroImage, 49, 3239–3247. https://doi.org/10.1016/j.neuroimage.2009.11.038
Malouin, F., Richards, C. L., Jackson, P. L., Lafleur, M. F., Durand, A., & Doyon, J. (2007). The kinesthetic and visual imagery questionnaire (KVIQ) for assessing motor imagery in persons with physical disabilities: A reliability and construct validity study. Journal of Neurologic Physical Therapy, 31, 20–29. https://doi.org/10.1097/01.NPT.0000260567.24122.64
Melo, M. C., Macedo, D. R., & Soares, A. B. (2020). Divergent findings in brain reorganization after spinal cord injury: A review. Journal of Neuroimaging, 30, 410–427. https://doi.org/10.1111/jon.12711
Menicucci, D., Di Gruttola, F., Cesari, V., Gemignani, A., Manzoni, D., & Sebastiani, L. (2020). Task-independent electrophysiological correlates of motor imagery ability from kinaesthetic and visual perspectives. Neuroscience, 443, 176–187. https://doi.org/10.1016/j.neuroscience.2020.07.038
Nascimento, I. A. P. D., Santiago, L. M. D. M., Souza, A. A. D., Pegado, C. D. L., Ribeiro, T. S., & Lindquist, A. R. R. (2019). Effects of motor imagery training of Parkinson's disease: A protocol for a randomized clinical trial. Trials, 20. https://doi.org/10.1186/s13063-019-3694-8
Page, S. J., Levine, P., Sisto, S., & Johnston, M. V. (2001). A randomized efficacy and feasibility study of imagery in acute stroke. Clinical Rehabilitation, 15, 233–240. https://doi.org/10.1191/026921501672063235
Porro, C. A., Francescato, M. P., Cettolo, V., Diamond, M. E., Baraldi, P., Zuiani, C., Bazzocchi, M., & di Prampero, P. E. (1996). Primary motor and sensory cortex activation during motor performance and motor imagery: A functional magnetic resonance imaging study. Journal of Neuroence, 16, 7688–7698. https://doi.org/10.1523/jneurosci.16-23-07688.1996
Proville, R. D., Spolidoro, M., Guyon, N., Dugué, G. P., Selimi, F., Isope, P., Popa, D., & Léna, C. (2014). Cerebellum involvement in cortical sensorimotor circuits for the control of voluntary movements. Nature Neuroscience, 17, 1233–1239. https://doi.org/10.1038/nn.3773
Sabbah, P., De, S. S., Leveque, C., Gay, S., Pfefer, F., Nioche, C., Sarrazin, J. L., Barouti, H., Tadie, M., & Cordoliani, Y. S. (2002). Sensorimotor cortical activity in patients with complete spinal cord injury: A functional magnetic resonance imaging study. Journal of Neurotrauma, 19, 53–60. https://doi.org/10.1089/089771502753460231
Saetia, S., Yoshimura, N., & Koike, Y. (2021). Constructing brain connectivity model using causal network reconstruction approach. Frontiers in Neuroinformatics, 15, 619557. https://doi.org/10.3389/fninf.2021.619557
Salmi, J., Pallesen, K. J., Neuvonen, T., Brattico, E., Korvenoja, A., Salonen, O., & Carlson, S. (2010). Cognitive and motor loops of the human cerebro-cerebellar system. Journal of Cognitive Neuroscience, 22, 2663–2676. https://doi.org/10.1162/jocn.2009.21382
Saturno, E., Bonato, C., Miniussi, C., Lazzaro, V., & Callea, L. (2008). Motor cortex changes in spinal cord injury: A TMS study. Neurological Research, 30, 1084–1085. https://doi.org/10.1179/174313208X332968
Schuch, F. B., Vancampfort, D., Firth, J., Rosenbaum, S., Ward, P. B., Silva, E. S., Hallgren, M., Ponce De Leon, A., Dunn, A. L., Deslandes, A. C., Fleck, M. P., Carvalho, A. F., & Stubbs, B. (2018). Physical Activity and Incident Depression: A Meta-Analysis of Prospective Cohort Studies. The American Journal of Psychiatry, 175, 631–648. https://doi.org/10.1176/appi.ajp.2018.17111194
Sebastián-Romagosa, M., Cho, W., Ortner, R., Murovec, N., Von Oertzen, T., Kamada, K., Allison, B. Z., & Guger, C. (2020). Brain computer interface treatment for motor rehabilitation of upper extremity of stroke Patients-A feasibility study. Frontiers in Neuroscience, 14, 591435. https://doi.org/10.3389/fnins.2020.591435
Shi, Y., Liu, W., Liu, R., Zeng, Y., Wu, L., Huang, S., Cai, G., Yang, J., & Wu, W. (2019). Investigation of the emotional network in depression after stroke: A study of multivariate Granger causality analysis of fMRI data. Journal of Affective Disorders, 249, 35–44. https://doi.org/10.1016/j.jad.2019.02.020
Silva, S. T. D., Borges, L. R. D. M., Santiago, L. M. M., Lucena, L. C., Raquel Rodrigues Lindquist, A., & Ribeiro, T. S. (2020). Motor imagery for gait rehabilitation after stroke. Stroke, 6, CD013019. https://doi.org/10.1161/STROKEAHA.120.033098
Stoodley, C. J., & Schmahmann, J. D. (2010). Evidence for topographic organization in the cerebellum of motor control versus cognitive and affective processing. Cortex, 46, 831–844. https://doi.org/10.1016/j.cortex.2009.11.008
Tian, X., Zarate, J. M., & Poeppel, D. (2016). Mental imagery of speech implicates two mechanisms of perceptual reactivation. Cortex, 77, 1–12. https://doi.org/10.1016/j.cortex.2016.01.002
Tian, Y., Ma, L., Xu, W., & Chen, S. (2020). The influence of listening to music on adults with left-behind experience revealed by EEG-based connectivity. Scientific Reports, 10, 7575. https://doi.org/10.1038/s41598-020-64381-x
Varsehi, H., & Firoozabadi, S. M. P. (2021). An EEG channel selection method for motor imagery based brain-computer interface and neurofeedback using Granger causality. Neural Networks, 133, 193–206. https://doi.org/10.1016/j.neunet.2020.11.002
Wang, L., Zhang, Y., Zhang, J., Sang, L., Li, P., Yan, R., Qiu, M., & Liu, C. (2019). Aging changes effective connectivity of motor networks during motor execution and motor imagery. Frontiers in Aging Neuroscience, 11, 312. https://doi.org/10.3389/fnagi.2019.00312
Wang, S. S., Kloth, A. D., & Badura, A. (2014). The cerebellum, sensitive periods, and autism. Neuron, 83, 518–532. https://doi.org/10.1016/j.neuron.2014.07.016
Welniarz, Q., Worbe, Y., & Gallea, C. (2021). The forward model: A unifying theory for the role of the cerebellum in motor control and sense of agency. Frontiers in Systems Neuroscience, 15, 644059. https://doi.org/10.3389/fnsys.2021.644059
Xue, J., Guo, H., Gao, Y., Wang, X., Cui, H., Chen, Z., Wang, B., & Xiang, J. (2019). Altered directed functional connectivity of the hippocampus in mild cognitive impairment and alzheimer’s disease: A Resting-State fMRI study. Frontiers in Aging Neuroscience, 11, 326. https://doi.org/10.3389/fnagi.2019.00326
Yang, Y. J., Jeon, E. J., Kim, J. S., & Chung, C. (2021). Characterization of kinesthetic motor imagery compared with visual motor imageries. Scientific Reports, 11, 3751. https://doi.org/10.1038/s41598-021-82241-0
Zhang, C., Kim, Y. K., & Eskandarian, A. (2021a). EEG-inception: An accurate and robust end-to-end neural network for EEG-based motor imagery classification. Journal of Neural Engineering. https://doi.org/10.1088/1741-2552/abed81
Zhang, W., Song, A., Zeng, H., Xu, B., & Miao, M. (2021b). Closed-Loop Phase-Dependent vibration stimulation improves motor Imagery-Based Brain-Computer interface performance. Frontiers in Neuroscience, 15, 638638. https://doi.org/10.3389/fnins.2021.638638
Acknowledgements
The authors thank the patients and healthy volunteers who participated in this study and generously gave their time.
Funding
This study was supported by the National Natural Science Foundation of China (Nos. 81871339 and 81271556), the Beijing Municipal Natural Science Foundation (No. 7113155), and the Science Foundation of Beijing Municipal Commission of Education (No. KM201210025013).
Author information
Authors and Affiliations
Contributions
Author contributions included conception (LW, NC), data collection, acquisition, and processing (LW, XJL, WMZ, XC, QC,YSH, LC, JR, JL), statistical analysis (LW, WQ), interpretation of results (LW, NC), drafting the manuscript work or revising it critically for important intellectual content (LW, NC, XJL, WMZ, WQ) and approval of final version to be published and agreement to be accountable for the integrity and accuracy of all aspects of the work (all authors).
Corresponding author
Ethics declarations
Ethical approval
The study protocol was approved by the Ethics Committee of Xuanwu Hospital of Capital Medical University. All procedures performed in this study involving human participants were in accordance with the ethical standards of the institutional research committee and with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
This article does not contain any studies with animals performed by any of the authors.
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Consent for publication
All authors consent to publish this manuscript.
Conflict of interest
The authors declare that they do not have any conflicts of interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, L., Li, X., Zheng, W. et al. Motor imagery evokes strengthened activation in sensorimotor areas and its effective connectivity related to cognitive regions in patients with complete spinal cord injury. Brain Imaging and Behavior 16, 2049–2060 (2022). https://doi.org/10.1007/s11682-022-00675-0
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11682-022-00675-0