Abstract
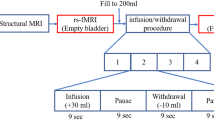
The aim of the study was to elucidate the correlation between spatially distinct brain areas with a full bladder from the perspective of functional connectivity using resting-state functional magnetic resonance imaging (rs-fMRI) with simultaneous urodynamic testing in healthy volunteers. The brain regions with full and empty bladders were reported via rs-fMRI using a 3 T magnetic resonance system. Then, we identified brain regions that are activated during bladder filling by calculating the amplitude of low-frequency fluctuation (ALFF) values using brain imaging software (DPABI and SPM8) and empirically derived six regions of interest (ROI) from analysis of activation were used as seeds for resting-state functional connectivity (rs-FC) analysis with the rest of the brain to examine differences in the two conditions. Statistical analysis was performed with a paired t-test and statistical significance was defined as a P < 0.01. Twenty-two healthy volunteers (11 men and 11 women) 35–64 years of age were enrolled. The rs-fMRI scans of 22 healthy volunteers were analyzed. After motion correction, two subjects were excluded. Meaningful data were obtained on 20 of these subjects. Compared with an empty bladder, functional connection enhancement was noted mainly in the right inferior orbitofrontal cortex and bilateral calcarine gyrus, the left lingual gyrus, left fusiform gyrus, left superior occipital gyrus, right insula, right inferior temporal gyrus, superior parietal lobe, left insula, right lingual gyrus, right fusiform gyrus, left parahippocampal gyrus, right inferior temporal gyrus, superior parietal lobe, left calcarine gyrus, bilateral lingual gyrus, prefrontal cortex, including the middle frontal gyrus and superior frontal gyrus, the right middle temporal gyrus, bilateral posterior cingulate cortex, and right precuneus. The decrease in functional connection was mainly located in the right inferior orbitofrontal cortex, prefrontal cortex, including the superior frontal gyrus, orbitofrontal cortex, and anterior cingulate cortex, the left inferior orbitofrontal cortex, right insula, middle occipital gyrus, angular gyrus, inferior frontal gyrus, right insula, middle temporal gyrus, inferior parietal lobe, middle occipital gyrus, supplementary motor area, superior frontal gyrus, left insula, bilateral posterior cingulate cortex, bilateral precuneus, middle occipital gyrus, and right middle temporal lobe. There were significant changes in the functional connectivity of the brain between full and empty bladders in healthy volunteers, which suggests that the central neural processes involved in storage needs brain areas with integrated control. These findings are strong evidence for physicians to consider brain responses in urine storage and offer the provision of some normative data.



Similar content being viewed by others
References
Andrew, J., & Nathan, P. W. (1964). Lesions on the anterior frontal lobes and disturbances of micturition and defaecation. Brain., 87, 233–262.
Athwal, B. S., Berkley, K. J., Hussain, I., Brennan, A., Craggs, M., Sakakibara, R., Frackowiak, R. S., & Fowler, C. J. (2001). Brain responses to changes in bladder volume and urge to void in healthy men. Brain., 124(2), 369–377.
Bush, G., Luu, P., & Posner, M. I. (2000). Cognitive and emotional influences in anterior cingulate cortex. Trends in Cognitive Sciences, 4(6), 215–222.
Carmichael, S. T., & Price, J. L. (1995). Limbic connections of the orbital and medial prefrontal cortex in macaque monkeys. The Journal of Comparative Neurology, 363, 615–641.
Clarkson, B. D., Karim, H. T., Griffiths, D. J., & Resnick, N. M. (2018). Functional connectivity of the brain in older women with urgency urinary incontinence. Neurourology and Urodynamics, 37(8), 2763–2775.
De Groat, W. C., & Theobald, R. J. (1976). Reflex activation of sympathetic pathways to vesical smooth muscle and parasympathetic ganglia by electrical stimulation of vesical afferents. The Journal of Physiology, 259, 223–237.
Di Gangi Herms, A. M., Veit, R., Reisenauer, C., Herms, A., Grodd, W., Enck, P., Stenzl, A., & Birbaumer, N. (2006). Functional imaging of stress urinary incontinence. Neuroimage., 29(1), 267–275.
Fowler, C. J., & Griffiths, D. J. (2010). A decade of functional brain imaging applied to bladder control. Neurourology and Urodynamics, 29(1), 49–55.
Franzmeier, N., Rubinski, A., Neitzel, J., Kim, Y., Damm, A., Na, D. L., Kim, H. J., Lyoo, C. H., Cho, H., Finsterwalder, S., Duering, M., Seo, S. W., & Ewers, M. (2019). Functional connectivity associated with tau levels in ageing, Alzheimer’s, and small vessel disease. Brain., 142, 1093–1107.
Gjone, R. (1966). Excitatory and inhibitory bladder responses to stimulation of ‘limbic’, diencephalic and mesencephalic structures in the cat. Acta Physiologica Scandinavica, 66, 91–102.
Griffiths, D. (1998). Clinical studies of cerebral and urinary tract function in elderly people with urinary incontinence. Behavioural Brain Research, 92, 151–155.
Jasmin, L., Burkey, A. R., Granato, A., & Ohara, P. T. (2004). Rostral agranular insular cortex and pain areas of the central nervous system: A tracttracing study in the rat. The Journal of Comparative Neurology, 468, 425–440.
Kavia, R. B., Dasgupta, R., & Fowler, C. J. (2005). Functional imaging and the central control of the bladder. The Journal of Comparative Neurology, 493(1), 27–32.
Ketai, L.H., Komesu, Y.M., Dodd, A.B., Rogers, R.G., Ling, J.M., & Mayer, A.R. (2016). Urgency urinary incontinence and the interoceptive network: A functional magnetic resonance imaging study. Am J Obstet Gynecol. 215(4), 449.e1-449.e17.
Khavari, R., Karmonik, C., Shy, M., Fletcher, S., & Boone, T. (2017). Functional magnetic resonance imaging with concurrent urodynamic testing identifies brain structures involved in micturition cycle in patients with multiple sclerosis. Adult Urology Voiding Dysfunction., 192, 438–444.
Krhut, J., Holy, P., Tintera, J., Zachoval, R., & Zvara, P. (2014). Brain activity during bladder filling and pelvic floor muscle contractions: A study using functional magnetic resonance imaging and synchronous urodynamics. International Journal of Urology, 21(2), 169–174.
Kuhtz-Buschbeck, J. P., Gilster, R., Horst, C., Hamann, M., Wolff, S., & Jansen, O. (2009). Control of bladder sensations: An fMRI study of brain activity and effective connectivity. Neuroimage., 47(1), 18–27.
Matsumoto, S., Ishikawa, A., Matsumoto, S., & Homma, Y. (2011). Brain response provoked by different bladder volumes: A near infrared spectroscopy study. Neurourology and Urodynamics, 30(4), 529–535.
Matsuura, S., Kakizaki, H., Mitsui, T., Shiga, T., Tamaki, N., & Koyanagi, T. (2002). Human brain region response to distention or cold stimulation of the bladder: A positron emission tomography study. The Journal of Urology, 168(5), 2035–2039.
Michels, L., Blok, B. F., Gregorini, F., Kurz, M., Schurch, B., Kessler, T. M., Kollias, S., & Mehnert, U. (2015). Supraspinal control of urine storage and micturition in men—An fMRI study. Cerebral Cortex, 25(10), 3369–3380.
Nardos, R., Gregory, W. T., Krisky, C., Newell, A., Nardos, B., Schlaggar, B., & Fair, D. A. (2014). Examining mechanisms of brain control of bladder function with resting state functional connectivity MRI. Neurourology and Urodynamics, 33(5), 493–501.
Nishijima, S., Sugaya, K., Kadekawa, K., Ashitomi, K., & Yamamoto, H. (2012). Effect of chemical stimulation of the medial frontal lobe on the micturition reflex in rats. The Journal of Urology, 187, 1116–1120.
Sakakibara, R., Fowler, C. J., & Hattori, T. (1999). Voiding and MRI analysis of the brain. Urogynecol J Pelvic Floor Dysfunct., 10(3), 192–199.
Sakakibara, R., Hattori, T., Yasuda, K., & Yamanishi, T. (1996). Micturitional disturbance after acute hemispheric stroke: Analysis of the lesion site by CT and MRI. Journal of the Neurological Sciences, 137(1), 47–56.
Seseke, S., Baudewig, J., Kallenberg, K., Ringert, R. H., Seseke, F., & Dechent, P. (2006). Voluntary pelvic floor muscle control—An fMRI study. Neuroimage., 31(4), 1399–1407.
Shelley, B. P., & Trimble, M. R. (2004). The insular lobe of Reil–its anatamico-functional, behavioural and neuropsychiatric attributes in humans–a review. The World Journal of Biological Psychiatry, 5(4), 176–200.
Shen, Z., Yu, S., Wang, M., She, T., Yang, Y., Wang, Y., Wei, W., Guo, X., Hu, Y., Liang, F., & Yang, J. (2019). Abnormal amygdala resting-state functional connectivity in primary dysmenorrhea. Neuroreport., 30(5), 363–368.
Yamamoto, T., Sakakibara, R., Nakazawa, K., Uchiyama, T., Shimizu, E., Hattori, T., & Kuwabara, S. (2010). Neuronal activities of forebrain structures with respect to bladder contraction in cats. Neuroscience Letters, 473, 42–47.
Yi, G., Limin, L., & Bertil, B. (2015). A resting-state functional MRI study on central control of storage: Brain response provoked by strong desire to void. International Urology and Nephrology, 47, 927–935.
Zare, A., Jahanshahi, A., Rahnama'i, M. S., Schipper, S., & Van, K. G. A. (2019). The role of the periaqueductal gray matter in lower urinary tract function. Molecular Neurobiology, 56(2), 920–934.
Acknowledgments
We wish to express our gratitude to our participants. The study was funded by grants from the National Natural Scientific Foundation of China (No.81570688) and were approved by the appropriate ethics committee of the China Rehabilitation Research Center (IRB: 2017-002-1).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Lingna Zhao, Limin Liao and Yi Gao declare that they have no conflict of interest.
Informed consent
All procedures performed in study involving human participants were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1964, and its later amendments or comparable ethical standards. Informed consent was obtained from all volunteers for being included in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhao, L., Liao, L. & Gao, Y. Brain functional connectivity during storage based on resting state functional magnetic resonance imaging with synchronous urodynamic testing in healthy volunteers. Brain Imaging and Behavior 15, 1676–1684 (2021). https://doi.org/10.1007/s11682-020-00362-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11682-020-00362-y