Abstract
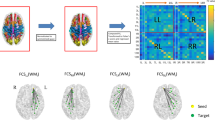
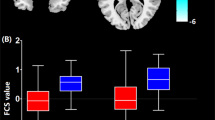
Growing evidence has supported that the nucleus accumbens (NAc), especially its shell portion, has been involved in epileptogenesis. However, relevant studies on vivo human brain are quite limited. In this study, we investigated left mesial temporal lobe epilepsy (MTLE) related function connectivity (FC) changes of NAc subregions using resting-state functional magnetic resonance imaging. We calculated functional connectivity from two NAc subregions to both whole brain and 16 related targets. Two-sample t-test (Alphasim multiple comparisons corrected) was performed to identify the effect of the disease on each seed’s whole brain network. Repeated-measures ANOVA and Post hoc pairwise t test (Bonferroni corrections) were performed to visualize the seed to target FC group differences in each subdivision. In whole brain FC networks, neither the left or right core show different FC changes. The left shell showed decreased FC with a cluster located around the right inferior frontal gyrus. The right shell portion showed increased FC with a cluster located around the left inferior temporal gyrus. The seed to targets results showed that the left shell of LTLE group exhibited lower FC with left posterior-parahippocampal gyrus and right caudate, putamen, thalamus, paracingulate gyrus but higher FC with right subcallosal cortex. The right core of LTLE group exhibited higher FC with right frontal pole and the right shell exhibited lower FC with left thalamus and left anterior-parahippocampal gyrus. This is the first study to investigate the functional connectivity changes of NAc subdivisions of epilepsy in vivo human brain. Our results showed that the left MTLE related FC changes on NAc are mainly on shell portion rather than core. The decrease FC between the left shell and right frontal area and the decrease FC between the right shell and left temporal area suggested they serve vital roles for MTLE.




Similar content being viewed by others
References
Baliki, M. N., Mansour, A., Baria, A. T., Huang, L., Berger, S. E., Fields, H. L., & Apkarian, A. V. (2013). Parceling human accumbens into putative core and shell dissociates encoding of values for reward and pain. The Journal of Neuroscience, 33(41), 16383–16393. https://doi.org/10.1523/jneurosci.1731-13.2013.
Bartolomei, F., Wendling, F., Bellanger, J. J., Regis, J., & Chauvel, P. (2001). Neural networks involving the medial temporal structures in temporal lobe epilepsy. Clinical Neurophysiology, 112(9), 1746–1760. https://doi.org/10.1016/S1388-2457(01)00591-0.
Bartolomei, F., Wendling, F., Regis, J., Gavaret, M., Guye, M., & Chauvel, P. (2004). Pre-ictal synchronicity in limbic networks of mesial temporal lobe epilepsy. Epilepsy Research, 61(1–3), 89–104. https://doi.org/10.1016/j.eplepsyres.2004.06.006.
Bartolomei, F., Chauvel, P., & Wendling, F. (2008). Epileptogenicity of brain structures in human temporal lobe epilepsy: A quantified study from intracerebral EEG. Brain, 131(Pt 7), 1818–1830. https://doi.org/10.1093/brain/awn111.
Basar, K., Sesia, T., Groenewegen, H., Steinbusch, H. W., Visser-Vandewalle, V., & Temel, Y. (2010). Nucleus accumbens and impulsivity. Progress in Neurobiology, 92(4), 533–557. https://doi.org/10.1016/j.pneurobio.2010.08.007.
Bertram, E. H. (2013). Neuronal circuits in epilepsy: Do they matter? Experimental Neurology, 244, 67–74. https://doi.org/10.1016/j.expneurol.2012.01.028.
Bertram, E. H., Zhang, D. X., Mangan, P., Fountain, N., & Rempe, D. (1998). Functional anatomy of limbic epilepsy: A proposal for central synchronization of a diffusely hyperexcitable network. Epilepsy Research, 32(1–2), 194–205. https://doi.org/10.1016/S0920-1211(98)00051-5.
Bettus, G., Guedj, E., Joyeux, F., Confort-Gouny, S., Soulier, E., Laguitton, V., … Guye, M. (2009). Decreased basal fMRI functional connectivity in epileptogenic networks and contralateral compensatory mechanisms. Human Brain Mapping, 30(5), 1580–1591. https://doi.org/10.1002/hbm.20625.
Bettus, G., Ranjeva, J. P., Wendling, F., Benar, C. G., Confort-Gouny, S., Regis, J., … Guye, M. (2011). Interictal functional connectivity of human epileptic networks assessed by intracerebral EEG and BOLD signal fluctuations. PLoS One, 6(5), e20071. https://doi.org/10.1371/journal.pone.0020071.
Brog, J. S., Salyapongse, A., Deutch, A. Y., & Zahm, D. S. (1993). The patterns of afferent innervation of the core and shell in the “accumbens” part of the rat ventral striatum: Immunohistochemical detection of retrogradely transported fluoro-gold. The Journal of Comparative Neurology, 338(2), 255–278. https://doi.org/10.1002/cne.903380209.
Buckner, R. L., Krienen, F. M., & Yeo, B. T. (2013). Opportunities and limitations of intrinsic functional connectivity MRI. Nature Neuroscience, 16(7), 832–837. https://doi.org/10.1038/nn.3423.
Calhoun, V. D., Wager, T. D., Krishnan, A., Rosch, K. S., Seymour, K. E., Nebel, M. B., … Kiehl, K. (2017). The impact of T1 versus EPI spatial normalization templates for fMRI data analyses. Human Brain Mapping, 38(11), 5331–5342. https://doi.org/10.1002/hbm.23737.
Chan, N. K., Kim, J., Shah, P., Brown, E. E., Plitman, E., Carravaggio, F., … Graff-Guerrero, A. (2019). Resting-state functional connectivity in treatment response and resistance in schizophrenia: A systematic review. Schizophrenia Research, 211, 10–20. https://doi.org/10.1016/j.schres.2019.07.020.
de Vasconcelos, A. P., Mazarati, A. M., Wasterlain, C. G., & Nehlig, A. (1999). Self-sustaining status epilepticus after a brief electrical stimulation of the perforant path: A 2-deoxyglucose study. Brain Research, 838(1–2), 110–118. https://doi.org/10.1016/s0006-8993(99)01729-1.
Floresco, S. B. (2015). The nucleus accumbens: An interface between cognition, emotion, and action. Annual Review of Psychology, 66, 25–52. https://doi.org/10.1146/annurev-psych-010213-115159.
Francis, T. C., Chandra, R., Friend, D. M., Finkel, E., Dayrit, G., Miranda, J., … Lobo, M. K. (2015). Nucleus accumbens medium spiny neuron subtypes mediate depression-related outcomes to social defeat stress. Biological Psychiatry, 77(3), 212–222. https://doi.org/10.1016/j.biopsych.2014.07.021.
Groenewegen, H. J., Vermeulen-Van der Zee, E., te Kortschot, A., & Witter, M. P. (1987). Organization of the projections from the subiculum to the ventral striatum in the rat. A study using anterograde transport of Phaseolus vulgaris leucoagglutinin. Neuroscience, 23(1), 103–120. https://doi.org/10.1016/0306-4522(87)90275-2.
Haber, S. N., & Knutson, B. (2010). The reward circuit: Linking primate anatomy and human imaging. Neuropsychopharmacology, 35(1), 4–26. https://doi.org/10.1038/npp.2009.129.
Heimer, L., Zahm, D. S., Churchill, L., Kalivas, P. W., & Wohltmann, C. (1991). Specificity in the projection patterns of accumbal core and shell in the rat. Neuroscience, 41(1), 89–125. https://doi.org/10.1016/0306-4522(91)90202-y.
Leung, L. S., Ma, J., & McLachlan, R. S. (2000). Behaviors induced or disrupted by complex partial seizures. Neuroscience and Biobehavioral Reviews, 24(7), 763–775. https://doi.org/10.1016/S0149-7634(00)00035-X.
Liao, W., Zhang, Z., Pan, Z., Mantini, D., Ding, J., Duan, X., … Chen, H. (2011). Default mode network abnormalities in mesial temporal lobe epilepsy: A study combining fMRI and DTI. Human Brain Mapping, 32(6), 883–895. doi:https://doi.org/10.1002/hbm.21076.
Liu, F., Guo, W., Liu, L., Long, Z., Ma, C., Xue, Z., … Chen, H. (2013). Abnormal amplitude low-frequency oscillations in medication-naive, first-episode patients with major depressive disorder: A resting-state fMRI study. Journal of Affective Disorders, 146(3), 401–406. https://doi.org/10.1016/j.jad.2012.10.001.
Liu, F., Guo, W., Fouche, J. P., Wang, Y., Wang, W., Ding, J., … Chen, H. (2015). Multivariate classification of social anxiety disorder using whole brain functional connectivity. Brain Structure & Function, 220(1), 101–115. https://doi.org/10.1007/s00429-013-0641-4.
Liu, F., Wang, Y., Li, M., Wang, W., Li, R., Zhang, Z., … Chen, H. (2017). Dynamic functional network connectivity in idiopathic generalized epilepsy with generalized tonic-clonic seizure. Human Brain Mapping, 38(2), 957–973. https://doi.org/10.1002/hbm.23430.
Loscher, W., Ebert, U., & Lehmann, H. (1996). Kindling induces a lasting, regionally selective increase of kynurenic acid in the nucleus accumbens. Brain Research, 725(2), 252–256. https://doi.org/10.1016/0006-8993(96)00341-1.
Lothman, E. W., Hatlelid, J. M., & Zorumski, C. F. (1985). Functional mapping of limbic seizures originating in the hippocampus: A combined 2-deoxyglucose and electrophysiologic study. Brain Research, 360(1–2), 92–100. https://doi.org/10.1016/0006-8993(85)91224-7.
Lucas-Neto, L., Reimao, S., Oliveira, E., Rainha-Campos, A., Sousa, J., Nunes, R. G., . . . Campos, J. G. (2015). Advanced MR imaging of the human nucleus accumbens--additional guiding tool for deep brain stimulation. Neuromodulation, 18(5), 341–348. https://doi.org/10.1111/ner.12289.
Peng, S. J., Harnod, T., Tsai, J. Z., Ker, M. D., Chiou, J. C., Chiueh, H., … Hsin, Y. L. (2014). Evaluation of subcortical grey matter abnormalities in patients with MRI-negative cortical epilepsy determined through structural and tensor magnetic resonance imaging. BMC Neurology, 14, 104. https://doi.org/10.1186/1471-2377-14-104.
Pittau, F., Grova, C., Moeller, F., Dubeau, F., & Gotman, J. (2012). Patterns of altered functional connectivity in mesial temporal lobe epilepsy. Epilepsia, 53(6), 1013–1023. https://doi.org/10.1111/j.1528-1167.2012.03464.x.
Salgado, S., & Kaplitt, M. G. (2015). The nucleus accumbens: A comprehensive review. Stereotactic and Functional Neurosurgery, 93(2), 75–93. https://doi.org/10.1159/000368279.
Scholl, E. A., Dudek, F. E., & Ekstrand, J. J. (2013). Neuronal degeneration is observed in multiple regions outside the hippocampus after lithium pilocarpine-induced status epilepticus in the immature rat. Neuroscience, 252, 45–59. https://doi.org/10.1016/j.neuroscience.2013.07.045.
Tong, X., An, D., Xiao, F., Lei, D., Niu, R., Li, W., … Zhou, D. (2019). Real-time effects of interictal spikes on hippocampus and amygdala functional connectivity in unilateral temporal lobe epilepsy: An EEG-fMRI study. Epilepsia, 60(2), 246–254. https://doi.org/10.1111/epi.14646.
Voets, N. L., Beckmann, C. F., Cole, D. M., Hong, S., Bernasconi, A., & Bernasconi, N. (2012). Structural substrates for resting network disruption in temporal lobe epilepsy. Brain, 135(Pt 8), 2350–2357. https://doi.org/10.1093/brain/aws137.
Xia, X., Fan, L., Cheng, C., Eickhoff, S. B., Chen, J., Li, H., & Jiang, T. (2017). Multimodal connectivity-based parcellation reveals a shell-core dichotomy of the human nucleus accumbens. Human Brain Mapping, 38(8), 3878–3898. https://doi.org/10.1002/hbm.23636.
Yan, C. G., Wang, X. D., Zuo, X. N., & Zang, Y. F. (2016). DPABI: Data processing & analysis for (Resting-state) brain imaging. Neuroinformatics, 14(3), 339–351. https://doi.org/10.1007/s12021-016-9299-4.
Yuan, K., Yu, D., Zhao, M., Li, M., Wang, R., Li, Y., … Tian, J. (2018). Abnormal frontostriatal tracts in young male tobacco smokers. Neuroimage, 183, 346–355. https://doi.org/10.1016/j.neuroimage.2018.08.046.
Yuan, K., Zhao, M., Yu, D., Manza, P., Volkow, N. D., Wang, G.-J., & Tian, J. (2019). Correction: Striato-cortical tracts predict 12-h abstinence-induced lapse in smokers. Neuropsychopharmacology, 44(7), 1338–1338. https://doi.org/10.1038/s41386-019-0330-y.
Zhao, X., Yang, R., Wang, K., Zhang, Z., Wang, J., Tan, X., et al. (2017). Connectivity-based parcellation of the nucleus accumbens into core and shell portions for stereotactic target localization and alterations in each NAc subdivision in mTLE patients. Human Brain Mapping, 39. https://doi.org/10.1002/hbm.23912.
Zhou, C. F. W. K. Q. S. (2014). Effect of high frequency deep brain stimulation to nucleus accumben on lithium-pilocarpine seizures in acute phase rats. Chinese Journal of Neurosurgery, 30(3). https://doi.org/10.3760/cma.j.issn.1001-2346.2014.03.026.
Funding sources
This study was funded by National Natural Science Foundation Guangdong United Found (grant number U1501256), the science and technology project of Guangdong Province (grant number 2015B010131011), the science and technology project of Guangdong Province (grant number 2015B010106008) and the National Natural Science Foundation of China (grant number 61971275).
Author information
Authors and Affiliations
Contributions
Qianjin Feng, YiKai Xu and Xixi Zhao designed the study; Xixi Zhao collected the data; Ru Yang analyzed the data; Ru Yang and Feng Hou undertook the statistical analysis; Xixi Zhao and Ru Yang wrote the manuscript; Xufeng Yao and Jun Liu contributed to the final manuscript.
Corresponding authors
Ethics declarations
Ethical approval
All procedures performed involving human participants in the present study were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Conflict of interest
All authors declared that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yang, R., Zhao, X., Liu, J. et al. Functional connectivity changes of nucleus Accumbens Shell portion in left mesial temporal lobe epilepsy patients. Brain Imaging and Behavior 14, 2659–2667 (2020). https://doi.org/10.1007/s11682-019-00217-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11682-019-00217-1