Abstract
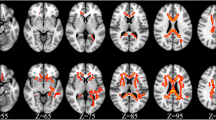
White matter hyperintensities (WMHs) have been associated with neurological complications including cognitive impairment. WMHs have been often described in HIV positive subjects and they have been linked to neurocognitive impairment, cerebrospinal fluid (CSF) residual viral replication and biomarkers of monocyte activation. Aim of this study was to grade WMHs in HIV-positive individuals using a simple visual scale and to explore their severity with clinical, neurocognitive and biomarker characteristics. Brain MRIs were retrospectively evaluated by two reviewers who rated WMHs following the “age-related white matter changes (ARWMC)” scale. 107 adult HIV-positive patients receiving lumbar punctures for clinical reasons were included. 70 patients (66.6%) were diagnosed with WMHs. Average WMH scores were higher in treated [7 (1–11)] vs. naïve individuals [3 (0–6)] (p = 0.008). Higher WHMs scores were observed in patients with chronic renal impairment along with chronic hepatitis (naïve) and longer HIV duration (treated participants). No consistent associations between plasma, CSF biomarkers and WMHs scores were found. 45 patients underwent full neurocognitive tests and WMHs scores were non-significantly higher in patients diagnosed with HAND [6.5 (0.5–8.3) vs. 1.5 (0–7), p = 0.165]; screening (IHDS and FAB), visuo-spatial (Corsi’s) and auditory-verbal memory (disillabic words repetition) tests scored worse in patients with higher WMHs. In our population of HIV-positive patients with low CD4 nadir and partial CD4 cell recovery the burden of WMHs was associated with the duration of HIV infection and with commonly observed comorbidities (such as renal and hepatic impairment). Given the association with worse neurocognition, further studies on tailored interventions are needed.



Similar content being viewed by others

References
Ancuta, P., Kamat, A., Kunstman, K. J., Kim, E.-Y., Autissier, P., Wurcel, A., et al. (2008). Microbial translocation is associated with increased monocyte activation and dementia in AIDS patients. PLoS One, 3(6), e2516.
Antinori, A., Arendt, G., Becker, J. T., Brew, B. J., Byrd, D. A., Cherner, M., et al. (2007). Updated research nosology for HIV-associated neurocognitive disorders. Neurology, 69(18), 1789–1799.
Appollonio, I., Leone, M., Isella, V., Piamarta, F., Consoli, T., Villa, M. L., Forapani, E., Russo, A., & Nichelli, P. (2005). The frontal assessment battery (FAB): Normative values in an Italian population sample. Neurological Sciences: Official Journal of the Italian Neurological Society and of the Italian Society of Clinical Neurophysiology, 26(2), 108–116.
Bairwa, D., Kumar, V., Vyas, S., Das, B. K., Srivastava, A. K., Pandey, R. M., et al. (2016). Case control study: Magnetic resonance spectroscopy of brain in HIV infected patients. BMC Neurology, 16, 99.
Burdo, T. H., Lo, J., Abbara, S., Wei, J., DeLelys, M. E., Preffer, F., et al. (2011). Soluble CD163, a novel marker of activated macrophages, is elevated and associated with noncalcified coronary plaque in HIV-infected patients. The Journal of Infectious Diseases, 204(8), 1227–1236.
Burdo, T. H., Weiffenbach, A., Woods, S. P., Letendre, S., Ellis, R. J., & Williams, K. C. (2013). Elevated sCD163 in plasma but not cerebrospinal fluid is a marker of neurocognitive impairment in HIV infection. AIDS (London, England), 27(9), 1387–1395.
Calcagno, A., Di Perri, G., & Bonora, S. (2017). Treating HIV infection in the central nervous system. Drugs, 77(2), 145–157.
Caplan, L. R., & van Gijn, J. (2012). Stroke Syndromes. Boston: Cambridge Core.
Clifford, K. M., Samboju, V., Cobigo, Y., Milanini, B., Marx, G. A., Hellmuth, J. M., et al. (2017). Progressive brain atrophy despite persistent viral suppression in HIV patients older than 60 years. Journal of Acquired Immune Deficiency Syndromes (1999), 76(3), 289–297.
Covic, A., Vervloet, M., Massy, Z. A., Torres, P. U., Goldsmith, D., Brandenburg, V., et al. (2017). Bone and mineral disorders in chronic kidney disease: Implications for cardiovascular health and ageing in the general population. The lancet. Diabetes & Endocrinology.
Cysique, L. A., Soares, J. R., Geng, G., Scarpetta, M., Moffat, K., Green, M., et al. (2017). White matter measures are near normal in controlled HIV infection except in those with cognitive impairment and longer HIV duration. Journal of Neurovirology, 23(4), 539–547.
Dufouil, C., Chalmers, J., Coskun, O., Besançon, V., Bousser, M.-G., Guillon, P., et al. (2005). Effects of blood pressure lowering on cerebral white matter hyperintensities in patients with stroke: The PROGRESS (perindopril protection against recurrent stroke study) magnetic resonance imaging substudy. Circulation, 112(11), 1644–1650.
Dufouil, C., Godin, O., Chalmers, J., Coskun, O., MacMahon, S., Tzourio-Mazoyer, N., et al. (2009). Severe cerebral white matter hyperintensities predict severe cognitive decline in patients with cerebrovascular disease history. Stroke, 40(6), 2219–2221.
EACS Guidelines. (2017). http://www.eacsociety.org/guidelines/eacs-guidelines/eacs-guidelines.html. Accessed 21 Feb 2018.
Haddow, L. J., Dudau, C., Chandrashekar, H., Cartledge, J. D., Hyare, H., Miller, R. F., & Jäger, H. R. (2014). Cross-sectional study of unexplained white matter lesions in HIV positive individuals undergoing brain magnetic resonance imaging. AIDS Patient Care and STDs, 28(7), 341–349.
Hammond, E. R., Crum, R. M., Treisman, G. J., Mehta, S. H., Marra, C. M., Clifford, D. B., et al. (2014). The cerebrospinal fluid HIV risk score for assessing central nervous system activity in persons with HIV. American Journal of Epidemiology, 180(3), 297–307.
Hermann, P., Romero, C., Schmidt, C., Reis, C., & Zerr, I. (2014). CSF biomarkers and neuropsychological profiles in patients with cerebral small-vessel disease. PLoS One, 9(8), e105000.
Hogg, R. S., Eyawo, O., Collins, A. B., Zhang, W., Jabbari, S., Hull, M. W., et al. (2017). Health-adjusted life expectancy in HIV-positive and HIV-negative men and women in British Columbia, Canada: A population-based observational cohort study. The lancet. HIV, 4(6), e270–e276.
Jespersen, S., Pedersen, K. K., Anesten, B., Zetterberg, H., Fuchs, D., Gisslén, M., et al. (2016). Soluble CD14 in cerebrospinal fluid is associated with markers of inflammation and axonal damage in untreated HIV-infected patients: A retrospective cross-sectional study. BMC Infectious Diseases, 16, 176.
Jonsson, M., Zetterberg, H., van Straaten, E., Lind, K., Syversen, S., Edman, A., et al. (2010). Cerebrospinal fluid biomarkers of white matter lesions - cross-sectional results from the LADIS study. European Journal of Neurology, 17(3), 377–382.
Kalayjian, R. C., Wu, K., Evans, S., Clifford, D. B., Pallaki, M., Currier, J. S., & Smryzynski, M. (2014). Proteinuria is associated with neurocognitive impairment in antiretroviral therapy treated HIV-infected individuals. Journal of Acquired Immune Deficiency Syndromes (1999), 67(1), 30–35.
Kugathasan, R., Collier, D. A., Haddow, L. J., El Bouzidi, K., Edwards, S. G., Cartledge, J. D., et al. (2017). Diffuse white matter signal abnormalities on magnetic resonance imaging are associated with human immunodeficiency virus type 1 viral escape in the central nervous system among patients with neurological symptoms. Clinical Infectious Diseases: An Official Publication of the Infectious Diseases Society of America, 64(8), 1059–1065.
Marin-Webb, V., Jessen, H., Kopp, U., Jessen, A. B., & Hahn, K. (2016). Validation of the international HIV dementia scale as a screening tool for HIV-associated neurocognitive disorders in a German-speaking HIV outpatient clinic. PLoS One, 11(12), e0168225.
McMurtray, A., Nakamoto, B., Shikuma, C., & Valcour, V. (2007). Small-vessel vascular disease in human immunodeficiency virus infection: The Hawaii aging with HIV cohort study. Cerebrovascular Diseases (Basel, Switzerland), 24(2–3), 236–241.
Mok, V., & Kim, J. S. (2015). Prevention and Management of Cerebral Small Vessel Disease. Journal of Stroke, 17(2), 111–122.
Moulignier, A., Savatovsky, A., Godin, O., Valin, N., Lescure, FX., Tubiana, R., et al. (2017) Cerebral small vessel disease in cART well-controlled HIV-infected patients ANRS EP51 microbreak. (n.d.). http://www.natap.org/2017/CROI/croi_125.htm. Accessed 21 Feb 2018.
Moulignier, A., Savatovsky, J., Assoumou, L., Lescure, F.-X., Lamirel, C., Godin, O., et al. (2018). Silent cerebral small-vessel disease is twice as prevalent in middle-aged individuals with well-controlled, combination antiretroviral therapy-treated human immunodeficiency virus (HIV) than in HIV-uninfected individuals. Clinical Infectious Diseases: An Official Publication of the Infectious Diseases Society of America, 66(11), 1762–1769. https://doi.org/10.1093/cid/cix1075.
Nightingale, S., Winston, A., Letendre, S., Michael, B. D., McArthur, J. C., Khoo, S., & Solomon, T. (2014). Controversies in HIV-associated neurocognitive disorders. The Lancet. Neurology, 13(11), 1139–1151.
Ricci, M., Pigliautile, M., D’Ambrosio, V., Ercolani, S., Bianchini, C., Ruggiero, C., et al. (2016). The clock drawing test as a screening tool in mild cognitive impairment and very mild dementia: A new brief method of scoring and normative data in the elderly. Neurological Sciences: Official Journal of the Italian Neurological Society and of the Italian Society of Clinical Neurophysiology, 37(6), 867–873.
Robinson-Papp, J., Navis, A., Dhamoon, M. S., Clark, U. S., Gutierrez-Contreras, J., & Morgello, S. (2018). The use of visual rating scales to quantify brain MRI lesions in patients with HIV infection. Journal of Neuroimaging, 28(2), 217–224.
Schmidt, R., Hayn, M., Fazekas, F., Kapeller, P., & Esterbauer, H. (1996). Magnetic resonance imaging white matter hyperintensities in clinically normal elderly individuals. Correlations with plasma concentrations of naturally occurring antioxidants. Stroke, 27(11), 2043–2047.
Schouten, J., Cinque, P., Gisslen, M., Reiss, P., & Portegies, P. (2011). HIV-1 infection and cognitive impairment in the cART era: A review. AIDS (London, England), 25(5), 561–575.
Seider, T. R., Gongvatana, A., Woods, A. J., Chen, H., Porges, E. C., Cummings, T., et al. (2016). Age exacerbates HIV-associated white matter abnormalities. Journal of Neurovirology, 22(2), 201–212.
Sheppard, D. P., Woods, S. P., Bondi, M. W., Gilbert, P. E., Massman, P. J., Doyle, K. L., & HIV Neurobehavioral Research Program Group. (2015). Does older age confer an increased risk of incident neurocognitive disorders among persons living with HIV disease? The Clinical Neuropsychologist, 29(5), 656–677.
Sinha, A., Ma, Y., Scherzer, R., Hur, S., Li, D., Ganz, P., et al. (2016). Role of T-cell dysfunction, inflammation, and coagulation in microvascular disease in HIV. Journal of the American Heart Association, 5(12), e004243. https://doi.org/10.1161/JAHA.116.004243.
Smit, M., Brinkman, K., Geerlings, S., Smit, C., Thyagarajan, K., van Sighem, A., et al. (2015). Future challenges for clinical care of an ageing population infected with HIV: A modelling study. The lancet. Infectious Diseases, 15(7), 810–818.
Soontornniyomkij, V., Umlauf, A., Chung, S. A., Cochran, M. L., Soontornniyomkij, B., Gouaux, B., et al. (2014). HIV protease inhibitor exposure predicts cerebral small vessel disease. AIDS (London, England), 28(9), 1297–1306.
Su, T., Wit, F. W. N. M., Caan, M. W. A., Schouten, J., Prins, M., Geurtsen, G. J., et al. (2016). White matter hyperintensities in relation to cognition in HIV-infected men with sustained suppressed viral load on combination antiretroviral therapy. AIDS (London, England), 30(15), 2329–2339.
van Dijk, E. J., Breteler, M. M. B., Schmidt, R., Berger, K., Nilsson, L.-G., Oudkerk, M., et al. (2004). The association between blood pressure, hypertension, and cerebral white matter lesions: Cardiovascular determinants of dementia study. Hypertension (Dallas, Tex.: 1979), 44(5), 625–630.
van Zoest, R. A., Underwood, J., De Francesco, D., Sabin, C. A., Cole, J. H., Wit, F. W., et al. (2017). Structural brain abnormalities in successfully treated HIV infection: Associations with disease and cerebrospinal fluid biomarkers. The Journal of Infectious Diseases, 217(1), 69–81.
Vuorinen, M., Solomon, A., Rovio, S., Nieminen, L., Kåreholt, I., Tuomilehto, J., et al. (2011). Changes in vascular risk factors from midlife to late life and white matter lesions: A 20-year follow-up study. Dementia and Geriatric Cognitive Disorders, 31(2), 119–125.
Watson, C., Busovaca, E., Foley, J. M., Allen, I. E., Schwarz, C. G., Jahanshad, N., et al. (2017). White matter hyperintensities correlate to cognition and fiber tract integrity in older adults with HIV. Journal of Neurovirology, 23(3), 422–429.
Wright, E. J., Grund, B., Robertson, K., Brew, B. J., Roediger, M., Bain, M. P., et al. (2010). Cardiovascular risk factors associated with lower baseline cognitive performance in HIV-positive persons. Neurology, 75(10), 864–873.
Wu, M., Fatukasi, O., Yang, S., Alger, J., Barker, PB., Hetherington, H., et al. (2018). Brain white matter hyperintensities, hiv disease, cognition and diabetes 435 poster session http://www.croiconference.org/download-croi-2018-abstract-book
Xiong, Y., Yang, J., Wong, A., Wong, C. H. K., Chan, S. S. W., Li, H. H. S., et al. (2011). Operational definitions improve reliability of the age-related white matter changes scale. European Journal of Neurology, 18(5), 744–749.
Yuen, T., Brouillette, M.-J., Fellows, L. K., Ellis, R. J., Letendre, S., Heaton, R., et al. (2017). Personalized risk index for neurocognitive decline among people with well-controlled HIV infection. Journal of Acquired Immune Deficiency Syndromes (1999), 76(1), 48–54.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
A. Calcagno. has received honoraria from Gilead, Janssen-Cilag, MSD and ViiV, and is currently receiving research grants from ViiV. G. Di Perri and S. Bonora have received honoraria from AbbVie, BMS, Gilead, Janssen-Cilag, MSD and ViiV. G.Nunnari has received speaker’s honoraria and consultancy fees from Viiv, Gilead, MSD. A. Trentalange declares that she has no conflict of interest. A. Prochet declares that he has no conflict of interest. D. Imperiale declares that he has no conflict of interest. M. Tettoni declares that she has no conflict of interest. A. Barco declares that she has no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. This article does not contain any studies with animals performed by any of the authors.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Electronic supplementary material
ESM 1
(DOC 37 kb)
Rights and permissions
About this article
Cite this article
Trentalange, A., Prochet, A., Imperiale, D. et al. Cerebral white matter Hyperintensities in HIV–positive patients. Brain Imaging and Behavior 14, 10–18 (2020). https://doi.org/10.1007/s11682-018-9966-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11682-018-9966-1