Abstract
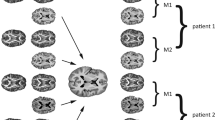
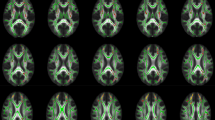
An increasing number of studies suggest chemotherapy for breast cancer may be neurotoxic. Cross-sectional MRI diffusion tensor imaging (DTI) studies suggest a vulnerability of brain white matter to various chemotherapeutic regimens. Up till now, this was confirmed in one prospective DTI study: Deprez et al. (2012) showed a widespread decline in fractional anisotropy (FA) of breast cancer patients after chemotherapy consisting of 5-fluorouracil (5-FU), epirubicin and cyclophosphamide (FEC) +/− taxanes +/− endocrine treatment. Our aim was to evaluate whether similar detrimental effects on white matter integrity would be observed with the currently widely prescribed anthracycline-based chemotherapy for breast cancer (predominantly doxorubicin and cyclophosphamide +/− taxanes +/− endocrine treatment (=BC + SYST; n = 26) compared to no systemic treatment (BC; n = 23) and no-cancer controls (NC; n = 30). Assessment took place before and six months after chemotherapy, and matched intervals for the unexposed groups. DTI data were analyzed using voxel-based tract-based spatial statistics and region of interest (ROI) analysis. Voxel-based analysis did not show an effect of chemotherapy +/− endocrine treatment on white matter integrity. ROI analysis however indicated subtle detrimental effects of chemotherapy +/− endocrine treatment by showing a larger decline in WM integrity in the superior longitudinal fasciculus and corticospinal tract in BC + SYST than BC. Indications for relatively mild neurotoxicity in our study might be explained by patient characteristics and specific aspects of data analysis. The omission of 5-FU in current treatment regimens or the administration of doxorubicin instead of epirubicin is also discussed as an explanation for the observed effects.


Similar content being viewed by others
References
Aaronson, N. K., Ahmedzai, S., Bergman, B., Bullinger, M., Cull, A., Duez, N. J., et al. (1993). The European Organization for Research and Treatment of cancer QLQ-C30: A quality-of-life instrument for use in international clinical trials in oncology. Journal of the National Cancer Institute, 85, 365–376. doi:10.1093/jnci/85.5.365.
Abraham, J., Haut, M. W., Moran, M. T., Filburn, S., Lemiuex, S., & Kuwabara, H. (2008a). Adjuvant chemotherapy for breast cancer: Effects on cerebral white matter seen in diffusion tensor imaging. Clinical Breast Cancer, 8(1), 88–91. doi:10.3816/CBC.2008.n.007.
Abraham, J., Haut, M. W., Moran, M. T., Filburn, S., Lemiuex, S., & Kuwabara, H. (2008b). Adjuvant chemotherapy for breast cancer: Effects on cerebral white matter seen in diffusion tensor imaging. Clinical Breast Cancer, 8(1), 88–91.
Akitake, R., Miyamoto, S., Nakamura, F., Horimatsu, T., Ezoe, Y., Muto, M., & Chiba, T. (2011). Early detection of 5-FU-induced acute leukoencephalopathy on diffusion-weighted MRI. Japanese Journal of Clinical Oncology, 41(1), 121–124. doi:10.1093/jjco/hyq157.
Alpherts, W., Aldenkamp, A. P. (1994). FePsy: The iron psyche. Heemstede.
Assaf, Y., & Pasternak, O. (2008). Diffusion tensor imaging (DTI)-based white matter mapping in brain research: A review. Journal of molecular neuroscience : MN, 34(1), 51–61. doi:10.1007/s12031-007-0029-0.
Bach, M., Laun, F. B., Leemans, A., Tax, C. M. W., Biessels, G. J., Stieltjes, B., & Maier-Hein, K. H. (2014). Methodological considerations on tract-based spatial statistics (TBSS). NeuroImage, 100, 358–369. doi:10.1016/j.neuroimage.2014.06.021.
Barrick, T. R., Charlton, R. A., Clark, C. A., & Markus, H. S. (2010). White matter structural decline in normal ageing: A prospective longitudinal study using tract-based spatial statistics. NeuroImage, 51(2), 565–577. doi:10.1016/j.neuroimage.2010.02.033.
Benedict, R. H. B., Schretlen, D., Groninger, L., & Brandt, J. (1998). Hopkins verbal learning test – Revised: Normative data and analysis of inter-form and test-retest reliability. The Clinical Neuropsychologist, 12, 43–55. doi:10.1076/clin.12.1.43.1726.
Briones, T. L., & Woods, J. (2014). Dysregulation in myelination mediated by persistent neuroinflammation: Possible mechanisms in chemotherapy-related cognitive impairment. Brain, Behavior, and Immunity, 35, 23–32. doi:10.1016/j.bbi.2013.07.175.
Cheung, Y. T., Lim, S. R., Ho, H. K., & Chan, A. (2013). Cytokines as mediators of chemotherapy-associated cognitive changes: Current evidence, limitations and directions for future research. PloS One, 8(12). doi:10.1371/journal.pone.0081234.
Choi, S. M., Lee, S. H., Yang, Y. S., Kim, B. C., Kim, M. K., & Cho, K. H. (2001). 5-fluorouracil-induced leukoencephalopathy in patients with breast cancer. Journal of Korean Medical Science. doi:10.3346/jkms.2001.16.3.328.
Cohen, S., Kamarck, T., & Mermelstein, R. (1983). A global measure of perceived stress. Journal of Health and Social Behavior, 24(4), 385–396.
De Ruiter, M. B., Reneman, L., Boogerd, W., Veltman, D. J., Caan, M., Douaud, G., et al. (2012). Late effects of high-dose adjuvant chemotherapy on white and gray matter in breast cancer survivors: Converging results from multimodal magnetic resonance imaging. Human Brain Mapping, 33(12), 2971–2983. doi:10.1002/hbm.21422.
Dekkers, A. M. M., Olff, M., & Näring, G. W. B. (2010). Identifying persons at risk for PTSD after trauma with TSQ in the Netherlands. Community Mental Health Journal, 46(1), 20–25. doi:10.1007/s10597-009-9195-6.
Deprez, S., Amant, F., Yigit, R., Porke, K., Verhoeven, J., Van den Stock, J., et al. (2011). Chemotherapy-induced structural changes in cerebral white matter and its correlation with impaired cognitive functioning in breast cancer patients. Human Brain Mapping, 32(3), 480–493. doi:10.1002/hbm.21033.
Deprez, S., Amant, F., Smeets, A., Peeters, R., Leemans, A., Van Hecke, W., et al. (2012). Longitudinal assessment of chemotherapy-induced structural changes in cerebral white matter and its correlation with impaired cognitive functioning. Journal of Clinical Oncology, 30(3), 274–281. doi:10.1200/JCO.2011.36.8571.
Eriksen, B. A., & Eriksen, C. W. (1974). Effects of noise letters upon the identification of a target letter in a nonsearch task. Perception & Psychophysics. doi:10.3758/BF03203267.
Fan, Y., Lin, K., Liu, H., Chen, Y., & Wu, C. (2015). Changes in structural integrity are correlated with motor and functional recovery after post-stroke rehabilitation. Restorative Neurology and Neuroscience, 33(6), 835–844. doi:10.3233/RNN-150523.
Fazekas, F., Chawluk, J. B., Alavi, A., Hurtig, H. I., & Zimmerman, R. A. (1987). MR signal abnormalities at 1. 5 T in Alzheimer ’ s dementia and normal aging deficiency. AJNR. American Journal of Neuroradiology, 149(August), 351–356. doi:10.2214/ajr.149.2.351.
Gosling, S. D., Rentfrow, P. J., & Swann, W. B. (2003). A very brief measure of the big-five personality domains. Journal of Research in Personality, 37, 504–528. doi:10.1016/S0092-6566(03)00046-1.
Han, R., Yang, Y. M., Dietrich, J., Luebke, A., Mayer-Pröschel, M., & Noble, M. (2008). Systemic 5-fluorouracil treatment causes a syndrome of delayed myelin destruction in the central nervous system. Journal of Biology, 7(4), 12. doi:10.1186/jbiol69.
Hesbacher, P. T., Rickels, K., Morris, R. J., Newman, H., & Rosenfeld, H. (1980). Psychiatric illness in family practice. The Journal of Clinical Psychiatry, 41(1), 6–10.
Israel, Z. H., Lossos, A., Barak, V., Soffer, D., & Siegal, T. (2000). Multifocal demyelinative leukoencephalopathy associated with 5-fluorouracil and levamisole. Acta Oncologica, 39(1), 117–120.
Khasraw, M., Bell, R., & Dang, C. (2012). Epirubicin: Is it like doxorubicin in breast cancer? A clinical review. Breast, 21(2), 142–149. doi:10.1016/j.breast.2011.12.012.
Koppelmans, V., de Groot, M., de Ruiter, M. B., Boogerd, W., Seynaeve, C., Vernooij, M. W., et al. (2014). Global and focal white matter integrity in breast cancer survivors 20 years after adjuvant chemotherapy. Human Brain Mapping, 35(3), 889–899. doi:10.1002/hbm.22221.
Mangin, J.-F., Poupon, C., Clark, C., Le Bihan, D., & Bloch, I. (2002). Distortion correction and robust tensor estimation for MR diffusion imaging. Medical Image Analysis, 6(3), 191–198.
McDonald, B. C., Conroy, S. K., Ahles, T. A., West, J. D., & Saykin, A. J. (2010). Gray matter reduction associated with systemic chemotherapy for breast cancer: A prospective MRI study. Breast Cancer Research and Treatment, 123(3), 819–828. doi:10.1007/s10549-010-1088-4.
Menning, S., de Ruiter, M., Veltman, D., Koppelmans, V., Kirschbaum, C., Boogerd, W., et al. (2015). Multimodal MRI and cognitive function in patients with breast cancer prior to adjuvant treatment - the role of fatigue. NeuroImage: Clinical, 7, 547–554.
Menning, S., de Ruiter, M. B., & Kieffer, J. M. Agelink van Rentergem, J., Veltman, D. J., Fruijtier, A., et al. (2016). Cognitive Impairment in a Subset of Breast Cancer Patients After Systemic Therapy - Results From a Longitudinal Study. Journal of Pain and Symptom Management, 52(4), 560–569.e1. doi:10.1016/j.jpainsymman.2016.04.012.
Nichols, T. E., & Holmes, A. P. (2002). Nonparametric permutation tests for functional neuroimaging: A primer with examples. Human Brain Mapping, 15(1), 1–25.
Pfefferbaum, A., Rosenbloom, M. J., Chu, W., Sassoon, S. A., Rohlfing, T., Pohl, K. M., et al. (2014). White matter microstructural recovery with abstinence and decline with relapse in alcohol dependence interacts with normal ageing: A controlled longitudinal DTI study. The Lancet. Psychiatry, 1(3), 202–212. doi:10.1016/S2215-0366(14)70301-3.
Pierpaoli, C., & Basser, P. J. (1996). Toward a quantitative assessment of diffusion anisotropy. Magnetic Resonance in Medicine, 36(6), 893–906. doi:10.1002/mrm.1910360612.
Pomykala, K. L., de Ruiter, M. B., Deprez, S., McDonald, B. C., & Silverman, D. H. S. (2013). Integrating imaging findings in evaluating the post-chemotherapy brain. Brain Imaging and Behavior, 7(4), 436–452. doi:10.1007/s11682-013-9239-y.
Schilder, C. M., Seynaeve, C., Beex, L. V., Boogerd, W., Linn, S. C., Gundy, C. M., et al. (2010a). Effects of tamoxifen and exemestane on cognitive functioning of postmenopausal patients with breast cancer: Results from the neuropsychological side study of the tamoxifen and exemestane adjuvant multinational trial. Journal of Clinical Oncology, 28(8), 1294–1300. doi:10.1200/JCO.2008.21.3553.
Schilder, C. M. T., Seynaeve, C., Linn, S. C., Boogerd, W., Beex, LVAM., Gundy, C. M., et al. (2010b). Cognitive functioning of postmenopausal breast cancer patients before adjuvant systemic therapy, and its association with medical and psychological factors. Critical Reviews in Oncology/Hematology, 76(2), 133–141. doi:10.1016/j.critrevonc.2009.11.001
Schmand, B., Lindeboom, J., & van Harskamp, F. (1992). De Nederlandse Leestest Voor Volwassenen. Lisse: Swets & Zeitlinger.
Schwarz, C. G., Reid, R. I., Gunter, J. L., Senjem, M. L., Przybelski, S. A., Zuk, S. M., et al. (2014). Improved DTI registration allows voxel-based analysis that outperforms tract-based spatial statistics. NeuroImage, 94, 65–78. doi:10.1016/j.neuroimage.2014.03.026.
Seigers, R., Loos, M., Van Tellingen, O., Boogerd, W., Smit, A. B., & Schagen, S. B. (2015). Cognitive impact of cytotoxic agents in mice. Psychopharmacology, 232(1), 17–37. doi:10.1007/s00213-014-3636-9.
Smith, S. M., Jenkinson, M., Johansen-Berg, H., Rueckert, D., Nichols, T. E., Mackay, C. E., et al. (2006). Tract-based spatial statistics: Voxelwise analysis of multi-subject diffusion data. NeuroImage, 31(4), 1487–1505. doi:10.1016/j.neuroimage.2006.02.024.
Stewart, A. L., Ware, J. E., Sherbourne, C. D., & Wells, K. B. (1992). Psychological distress/well-being and cognitive functioning measures. In A. L. Stewart & J. E. Ware (Eds.), Measuring functioning and well-being: The medical outcomes study approach (pp. 102–142). Durham, NC: Duke University Press.
Stouten-Kemperman, M. M., de Ruiter, M. B., Koppelmans, V., Boogerd, W., Reneman, L., & Schagen, S. B. (2014). Neurotoxicity in breast cancer survivors ≥10 years post-treatment is dependent on treatment type. Brain Imaging and Behavior, 275–284. doi:10.1007/s11682-014-9305-0.
Sullivan, E. V., Rohlfing, T., & Pfefferbaum, A. (2010). Longitudinal study of callosal microstructure in the normal adult aging brain using quantitative DTI fiber tracking. Developmental Neuropsychology, 35(3), 233–256. doi:10.1080/87565641003689556.
Wald, F. D. M., & Mellenbergh, G. J. (1990). De verkorte versie van de Nederlandse vertaling van de Profile of Mood States (POMS). Nederlands Tijdschrift voor de Psychologie, 45, 86–90.
Wechsler, D. (2000). WAIS-III Nederlandstalige bewerking. Technische Handleiding. Lisse: Swets & Zeitlinger.
Wefel, J. S., Kesler, S. R., Noll, K. R., & Schagen, S. B. (2015). Clinical characteristics, pathophysiology, and management of noncentral nervous system cancer-related cognitive impairment in adults. CA: A Cancer Journal for Clinicians, 65(2), 123–138. doi:10.3322/caac.21258.
Weiss, D., & Marmar, C. (1997). The impact of event scale - revised. In J. Wilson & T. Keane (Eds.), Assessing psychological trauma and PTSD (pp. 399–411). New York: Guilford Press.
Weng, Q., Tan, B., Wang, J., Wang, J., Zhou, H., Shi, J., et al. (2014). 5-fluorouracil causes severe CNS demyelination by disruption of TCF7L2/HDAC1/HDAC2 complex in adolescent mice. Toxicology, 325, 144–150. doi:10.1016/j.tox.2014.08.011.
Winocur, G., Vardy, J., Binns, M. A., Kerr, L., & Tannock, I. (2006). The effects of the anti-cancer drugs, methotrexate and 5-fluorouracil, on cognitive function in mice. Pharmacology Biochemistry and Behavior, 85(1), 66–75. doi:10.1016/j.pbb.2006.07.010.
Zwart, W., Terra, H., Linn, S. C., & Schagen, S. B. (2015). Cognitive effects of endocrine therapy for breast cancer: Keep calm and carry on? Nature reviews. Clinical Oncology, 12(10), 597–606. doi:10.1038/nrclinonc.2015.124.
Acknowledgements
This study was funded by the Dutch Cancer Society (KWF 2009-4284). We thank Epie Boven, MD, PhD, Suzan van der Meij, MD, Vera Lustig, MD, and Monique Bos, MD, PhD, for their contributions to the patient recruitment. We are indebted to all patients and controls, as well as physicians and nurses of the Netherlands Cancer Institute-Antoni van Leeuwenhoek Hospital, VU University Medical Center, Flevoziekenhuis, Reinier de Graaf Gasthuis and Academic Medical Center, for providing patients for this study and the research assistants for helping collecting the data.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was funded by the Dutch Cancer Society (grant number KWF 2009–4284).
Conflict of interest
Author Menning declares that she has no conflict of interest. Author de Ruiter declares that he has no conflict of interest. Author Veltman declares that he has no conflict of interest. Author Boogerd declares that he has no conflict of interest. Author Oldenburg declares that she has no conflict of interest. Author Reneman declares that she has no conflict of interest. Author Schagen declares that she has no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Menning, S., de Ruiter, M.B., Veltman, D.J. et al. Changes in brain white matter integrity after systemic treatment for breast cancer: a prospective longitudinal study. Brain Imaging and Behavior 12, 324–334 (2018). https://doi.org/10.1007/s11682-017-9695-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11682-017-9695-x