Abstract
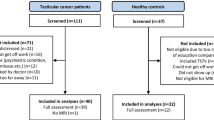
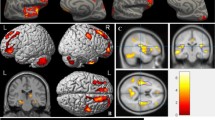
Evidence suggests that testicular cancer (TC) and its treatment are associated with cognitive impairment. However, the underlying neural substrate and biological mechanisms are poorly understood. This study aimed to investigate changes in cognition and brain grey matter (GM) morphology in TC patients undergoing treatment, and to explore associations with immune markers, endocrine markers, and genotype. Sixty-five patients with stage I-III TC underwent assessment after surgery but prior to further treatment and again 6 months after. Twenty-two patients received chemotherapy (+CT), while 43 did not (−CT). Assessments included neuropsychological testing, whole-brain magnetic resonance imaging, and blood samples. Twenty-five healthy controls (HCs) underwent neuropsychological testing with a matching time interval. A regression-based approach was used to determine cognitive changes and longitudinal voxel-based morphometry (VBM) was performed to investigate changes in GM density in the TC groups. Compared with the HCs, both TC groups showed higher rates of cognitive decline (p < 0.05). A trend towards greater decline was observed in + CT (63.6 %) compared with -CT patients (39.5 %) (p = 0.07). VBM revealed widespread GM reductions in both TC groups, but a group-by-time interaction analysis revealed prefrontal reductions specific to the + CT group (p = 0.02), which were associated with poorer cognitive performance. Poorer cognitive performance was also associated with an increase in tumor necrosis factor alpha in + CT patients. Furthermore, an interaction effect was found between the APOE ε4 genotype and chemotherapy on cognitive performance with ε4 carriers performing significantly worse. These findings provide novel evidence of changes in cognition and brain morphology in TC patients undergoing treatment.




Similar content being viewed by others
References
Ahles, T. A., & Saykin, A. J. (2007). Candidate mechanisms for chemotherapy-induced cognitive changes. Nature Reviews Cancer, 7(3), 192–201. doi:10.1038/nrc2073.
Ahles, T. A., Saykin, A. J., Noll, W. W., Furstenberg, C. T., Guerin, S., Cole, B., & Mott, L. A. (2003). The relationship of APOE genotype to neuropsychological performance in long-term cancer survivors treated with standard dose chemotherapy. Psycho-Oncology, 12(6), 612–9. doi:10.1002/pon.742.
Ahles, T. A., Li, Y., McDonald, B. C., Schwartz, G. N., Kaufman, P. A., Tsongalis, G. J., et al. (2014). Longitudinal assessment of cognitive changes associated with adjuvant treatment for breast cancer: the impact of APOE and smoking. Psycho-Oncology, 23(12), 1382–90. doi:10.1002/pon.3545.
Amidi, A., Wu, L. M., Agerbaek, M., Larsen, P. L., Pedersen, A. D., Mehlsen, M., et al. (2015a). Cognitive impairment and potential biological and psychological correlates of neuropsychological performance in recently orchiectomized testicular cancer patients. Psycho-Oncology, 24(9), 1174–1180. doi:10.1002/pon.3804.
Amidi, A., Wu, L. M., Pedersen, A. D., Mehlsen, M., Pedersen, C. G., Rossen, P., et al. (2015b). Cognitive impairment in testicular cancer survivors 2 to 7 years after treatment. Supportive Care in Cancer: Official Journal of the Multinational Association of Supportive Care in Cancer, 23(10), 2973–9. doi:10.1007/s00520-015-2663-3.
Andreotti, C., Root, J. C., Ahles, T. A., McEwen, B. S., & Compas, B. E. (2014). Cancer, coping, and cognition: a model for the role of stress reactivity in cancer-related cognitive decline. Psycho-Oncology. doi:10.1002/pon.3683.
Andres, A. L., Gong, X., Di, K., & Bota, D. A. (2014). Low-doses of cisplatin injure hippocampal synapses: a mechanism for “chemo” brain? Experimental Neurology, 255, 137–44. doi:10.1016/j.expneurol.2014.02.020.
Ashburner, J., & Friston, K. J. (2000). Voxel-based morphometry--the methods. NeuroImage, 11(6 Pt 1), 805–21. doi:10.1006/nimg.2000.0582.
Bandak, M., Aksglaede, L., Juul, A., Rørth, M., & Daugaard, G. (2011). The pituitary-Leydig cell axis before and after orchiectomy in patients with stage I testicular cancer. European Journal of Cancer, 47(17), 2585–2591. doi:10.1016/j.ejca.2011.05.026.
Beauchet, O. (2006). Testosterone and cognitive function: current clinical evidence of a relationship. European Journal of Endocrinology / European Federation of Endocrine Societies, 155(6), 773–81. doi:10.1530/eje.1.02306.
Benton, A. L., & Hamsher, K. D. S. (1989). Multilingual aphasia examination. Iowa City: AJA Associates.
Bu, G. (2009). Apolipoprotein E and its receptors in Alzheimer’s disease: pathways, pathogenesis and therapy. Nature Reviews Neuroscience, 10(5), 333–44. doi:10.1038/nrn2620.
Buller, K. M. (2001). Role of circumventricular organs in pro-inflammatory cytokine-induced activation of the hypothalamic-pituitary-adrenal axis. Clinical and Experimental Pharmacology & Physiology, 28(7), 581–9. http://www.ncbi.nlm.nih.gov/pubmed/11458886 . Accessed 5 June 2015.
Carozzi, V. A., Canta, A., Chiorazzi, A., & Cavaletti, G. (2014). Chemotherapy-induced peripheral neuropathy: What do we know about mechanisms? Neuroscience Letters. doi:10.1016/j.neulet.2014.10.014.
Cherrier, M. M. (2009). Testosterone effects on cognition in health and disease. Frontiers of Hormone Research, 37(0301–3073 (Print)), 150–62. doi:10.1159/000176051.
Cheung, Y. T., Lim, S. R., Ho, H. K., & Chan, A. (2013). Cytokines as mediators of chemotherapy-associated cognitive changes: current evidence, limitations and directions for future research. PLoS ONE, 8(12), e81234. doi:10.1371/journal.pone.0081234.
Conroy, S. K., McDonald, B. C., Smith, D. J., Moser, L. R., West, J. D., Kamendulis, L. M., et al. (2013). Alterations in brain structure and function in breast cancer survivors: effect of post-chemotherapy interval and relation to oxidative DNA damage. Breast Cancer Research and Treatment, 137(1573–7217 (Electronic)), 493–502. doi:10.1007/s10549-012-2385-x.
Dahl, O., & Brydøy, M. (2012). Testicular germ cell tumours - still many challenges. Acta Oncologica (Stockholm, Sweden), 51(2), 147–50. doi:10.3109/0284186X.2011.653441.
Dietrich, J., Han, R., Yang, Y., Mayer-Pröschel, M., & Noble, M. (2006). CNS progenitor cells and oligodendrocytes are targets of chemotherapeutic agents in vitro and in vivo. Journal of Biology, 5(7), 22. doi:10.1186/jbiol50.
Eiseman, J. L., Beumer, J. H., Rigatti, L. H., Strychor, S., Meyers, K., Dienel, S., & Horn, C. C. (2014). Plasma pharmacokinetics and tissue and brain distribution of cisplatin in musk shrews. Cancer Chemotherapy and Pharmacology. doi:10.1007/s00280-014-2623-5.
Ganz, P. A., Bower, J. E., Kwan, L., Castellon, S. A., Silverman, D. H. S., Geist, C., et al. (2013). Does tumor necrosis factor-alpha (TNF-α) play a role in post-chemotherapy cerebral dysfunction? Brain, Behavior, and Immunity, 30, S99–108. doi:10.1016/j.bbi.2012.07.015.
Gietema, J., Meinardi, M., Messerschmidt, J., Gelevert, T., Alt, F., Uges, D., & Th Sleijfer, D. (2000). Circulating plasma platinum more than 10 years after cisplatin treatment for testicular cancer. The Lancet, 355(9209), 1075–1076. doi:10.1016/S0140-6736(00)02044-4.
Gong, X., Schwartz, P. H., Linskey, M. E., & Bota, D. A. (2011). Neural stem/progenitors and glioma stem-like cells have differential sensitivity to chemotherapy. Neurology, 76(13), 1126–34. doi:10.1212/WNL.0b013e318212a89f.
Gregg, R. W., Molepo, J. M., Monpetit, V. J., Mikael, N. Z., Redmond, D., Gadia, M., & Stewart, D. J. (1992). Cisplatin neurotoxicity: the relationship between dosage, time, and platinum concentration in neurologic tissues, and morphologic evidence of toxicity. Journal of Clinical Oncology: Official Journal of the American Society of Clinical Oncology, 10(5), 795–803. http://www.ncbi.nlm.nih.gov/pubmed/1569451 . Accessed 25 February 2015.
Groll, R. J., Warde, P., & Jewett, M. A. S. (2007). A comprehensive systematic review of testicular germ cell tumor surveillance. Critical Reviews in Oncology/Hematology, 64(3), 182–97. doi:10.1016/j.critrevonc.2007.04.014.
Gultekin, S. H., Rosenfeld, M. R., Voltz, R., Eichen, J., Posner, J. B., & Dalmau, J. (2000). Paraneoplastic limbic encephalitis: neurological symptoms, immunological findings and tumour association in 50 patients. Brain: a Journal of Neurology, 123(Pt 7), 1481–94. http://www.ncbi.nlm.nih.gov/pubmed/10869059 . Accessed 21 February 2016.
Heaton, R. K., Chelune, G. J., Talley, J. L., Kay, G. G., & Curtiss, G. (1993). Wisconsin card sorting test manual revised and expanded. Odessa: Psychological Assessment Resources.
Hermelink, K., Voigt, V., Kaste, J., Neufeld, F., Wuerstlein, R., Bühner, M., et al. (2015). Elucidating pretreatment cognitive impairment in breast cancer patients: the impact of cancer-related post-traumatic stress. Journal of the National Cancer Institute, 107(7). doi:10.1093/jnci/djv099
Holland, J., Bandelow, S., & Hogervorst, E. (2011). Testosterone levels and cognition in elderly men: a review. Maturitas, 69(1873–4111 (Electronic)), 322–337.
Holtzman, D. M., Herz, J., & Bu, G. (2012). Apolipoprotein E and apolipoprotein E receptors: normal biology and roles in Alzheimer disease. Cold Spring Harbor Perspectives in Medicine, 2(3), a006312. doi:10.1101/cshperspect.a006312.
Inagaki, M., Yoshikawa, E., Matsuoka, Y., Sugawara, Y., Nakano, T., Akechi, T., et al. (2007). Smaller regional volumes of brain gray and white matter demonstrated in breast cancer survivors exposed to adjuvant chemotherapy. Cancer, 109(0008-543X (Print)), 146–156.
Janelsins, M. C., Kesler, S. R., Ahles, T. A., & Morrow, G. R. (2014). Prevalence, mechanisms, and management of cancer-related cognitive impairment. International Review of Psychiatry (Abingdon, England), 26(1), 102–13. doi:10.3109/09540261.2013.864260.
Keime-Guibert, F., Graus, F., Broët, P., Reñé, R., Molinuevo, J. L., Ascaso, C., & Delattre, J. Y. (1999). Clinical outcome of patients with anti-Hu-associated encephalomyelitis after treatment of the tumor. Neurology, 53(8), 1719–23. http://www.ncbi.nlm.nih.gov/pubmed/10563618 . Accessed 21 February 2016.
Kesler, S., Janelsins, M., Koovakkattu, D., Palesh, O., Mustian, K., Morrow, G., & Dhabhar, F. S. (2013). Reduced hippocampal volume and verbal memory performance associated with interleukin-6 and tumor necrosis factor-alpha levels in chemotherapy-treated breast cancer survivors. Brain, Behavior, and Immunity, 30(Suppl(1090–2139 (Electronic)), S109–16. doi:10.1016/j.bbi.2012.05.017.
Lambert, J. C., Ibrahim-Verbaas, C. A., Harold, D., Naj, A. C., Sims, R., Bellenguez, C., et al. (2013). Meta-analysis of 74,046 individuals identifies 11 new susceptibility loci for Alzheimer’s disease. Nature Genetics, 45(12), 1452–8. doi:10.1038/ng.2802.
Lange, M., Giffard, B., Noal, S., Rigal, O., Kurtz, J.-E., Heutte, N., et al. (2014). Baseline cognitive functions among elderly patients with localised breast cancer. European Journal of Cancer (Oxford, England: 1990), 50(13), 2181–9. doi:10.1016/j.ejca.2014.05.026.
Lee, B. K., Glass, T. A., McAtee, M. J., Wand, G. S., Bandeen-Roche, K., Bolla, K. I., & Schwartz, B. S. (2007). Associations of salivary cortisol with cognitive function in the Baltimore memory study. Archives of General Psychiatry, 64(7), 810. doi:10.1001/archpsyc.64.7.810.
Lentini, E., Kasahara, M., Arver, S., & Savic, I. (2012). Sex differences in the human brain and the impact of sex chromosomes and sex hormones. Cerebral Cortex, 23(10), 2322–2336. doi:10.1093/cercor/bhs222.
Lepage, C., Smith, A. M., Moreau, J., Barlow-Krelina, E., Wallis, N., Collins, B., et al. (2014). A prospective study of grey matter and cognitive function alterations in chemotherapy-treated breast cancer patients. Springerplus, 3, 444. doi:10.1186/2193-1801-3-444.
Li, D.-W., Sun, J.-Y., Wang, K., Zhang, S., Hou, Y.-J., Yang, M.-F., et al. (2015). Attenuation of cisplatin-induced neurotoxicity by cyanidin, a natural inhibitor of ROS-mediated apoptosis in PC12 cells. Cellular and Molecular Neurobiology. doi:10.1007/s10571-015-0194-6.
Lv, X.-F., Zheng, X.-L., Zhang, W.-D., Liu, L.-Z., Zhang, Y.-M., Chen, M.-Y., & Li, L. (2014). Radiation-induced changes in normal-appearing gray matter in patients with nasopharyngeal carcinoma: a magnetic resonance imaging voxel-based morphometry study. Neuroradiology, 56(5), 423–30. doi:10.1007/s00234-014-1338-y.
Maier, S. F., & Watkins, L. R. (2003). Immune-to-central nervous system communication and its role in modulating pain and cognition: Implications for cancer and cancer treatment. Brain, Behavior, and Immunity, 17(Suppl 1), S125–31.
McDonald, B. C., Conroy, S. K., Ahles, T. A., West, J. D., & Saykin, A. J. (2010). Gray matter reduction associated with systemic chemotherapy for breast cancer: a prospective MRI study. Breast Cancer Research and Treatment, 123(3), 819–28. doi:10.1007/s10549-010-1088-4.
McDonald, B. C., Conroy, S. K., Smith, D. J., West, J. D., & Saykin, A. J. (2013). Frontal gray matter reduction after breast cancer chemotherapy and association with executive symptoms: a replication and extension study. Brain, Behavior, and Immunity, 30(Suppl(1090–2139 (Electronic)), S117–25. doi:10.1016/j.bbi.2012.05.007.
McSweeny, A. J., Naugle, R. I., Chelune, G. J., & Lüders, H. (1993). T scores for change”: an illustration of a regression approach to depicting change in clinical neuropsychology. Clinical Neuropsychologist, 7(3), 300–312. doi:10.1080/13854049308401901.
Osburg, B., Peiser, C., Dömling, D., Schomburg, L., Ko, Y. T., Voigt, K., & Bickel, U. (2002). Effect of endotoxin on expression of TNF receptors and transport of TNF-alpha at the blood–brain barrier of the rat. American Journal of Physiology Endocrinology and Metabolism, 283(5), E899–908. doi:10.1152/ajpendo.00436.2001.
Ouimet, L. A., Stewart, A., Collins, B., Schindler, D., & Bielajew, C. (2009). Measuring neuropsychological change following breast cancer treatment: an analysis of statistical models. Journal of Clinical and Experimental Neuropsychology, 31(1), 73–89. doi:10.1080/13803390801992725.
Pedersen, A. D., Rossen, P., Mehlsen, M. Y., Pedersen, C. G., Zachariae, R., & von der Maase, H. (2009). Long-term cognitive function following chemotherapy in patients with testicular cancer. Journal of the International Neuropsychological Society: JINS, 15(2), 296–301. doi:10.1017/S1355617709090316.
Reinvang, I., Espeseth, T., & Westlye, L. T. (2013). APOE-related biomarker profiles in non-pathological aging and early phases of Alzheimer’s disease. Neuroscience and Biobehavioral Reviews, 37(8), 1322–35. doi:10.1016/j.neubiorev.2013.05.006.
Reitan, R. M. (1958). Validity of the trail making test as an indicator of organic brain damage. Perceptual and Motor Skills, 8, 271–276.
Roberts, R. O., Geda, Y. E., Knopman, D. S., Boeve, B. F., Christianson, T. J. H., Pankratz, V. S., et al. (2009). Association of C-reactive protein with mild cognitive impairment. Alzheimer’s & Dementia: the Journal of the Alzheimer’s Association, 5(5), 398–405. doi:10.1016/j.jalz.2009.01.025.
Rzeski, W., Pruskil, S., Macke, A., Felderhoff-Mueser, U., Reiher, A. K., Hoerster, F., et al. (2004). Anticancer agents are potent neurotoxins in vitro and in vivo. Annals of Neurology, 56(3), 351–60. doi:10.1002/ana.20185.
Schagen, S. B., Boogerd, W., Muller, M. J., Huinink, W. T. B., Moonen, L., Meinhardt, W., & Van Dam, F. S. A. M. (2008). Cognitive complaints and cognitive impairment following BEP chemotherapy in patients with testicular cancer. Acta Oncologica (Stockholm, Sweden), 47(1), 63–70. doi:10.1080/02841860701518058.
Schmidt, M. (1996). Auditory and verbal learning test. A handbook. Los Angeles: Western Psychological Services.
Sheldon, J., Riches, P., Gooding, R., Soni, N., & Hobbs, J. R. (1993). C-reactive protein and its cytokine mediators in intensive-care patients. Clinical Chemistry, 39(1), 147–150.
Simó, M., Root, J. C., Vaquero, L., Ripollés, P., Jové, J., Ahles, T., et al. (2015). Cognitive and brain structural changes in a lung cancer population. Journal of Thoracic Oncology: Official Publication of the International Association for the Study of Lung Cancer, 10(1), 38–45. doi:10.1097/JTO.0000000000000345.
Skaali, T., Fosså, S. D., Andersson, S., Cvancarova, M., Langberg, C. W., Lehne, G., & Dahl, A. A. (2011). A prospective study of neuropsychological functioning in testicular cancer patients. Annals of Oncology: Official Journal of the European Society for Medical Oncology / ESMO, 22(5), 1062–70. doi:10.1093/annonc/mdq553.
Stouten-Kemperman, M. M., de Ruiter, M. B., Caan, M. W. A., Boogerd, W., Kerst, M. J., Reneman, L., & Schagen, S. B. (2015). Lower cognitive performance and white matter changes in testicular cancer survivors 10 years after chemotherapy. Human Brain Mapping. doi:10.1002/hbm.22942.
Szczepanski, S. M., & Knight, R. T. (2014). Insights into human behavior from lesions to the prefrontal cortex. Neuron, 83(5), 1002–18. doi:10.1016/j.neuron.2014.08.011.
Touitou, Y., Bogdan, A., Levi, F., Benavibes, M., & Auzeby, A. (1996). Disruption of the circadian patterns of serum cortisol in breast and ovarian cancer patients: relationships with tumour marker antigens. British Journal of Cancer, 74(8), 1248–52. Nature Publishing Group. /pmc/articles/PMC2075940/?report = abstract. Accessed 2 September 2014.
Trabert, B., Chen, J., Devesa, S. S., Bray, F., & McGlynn, K. A. (2014). International patterns and trends in testicular cancer incidence, overall and by histologic subtype, 1973–2007. Andrology. doi:10.1111/andr.293.
Wechsler, D. (2008). Wechsler adult intelligence scale-fourth edition. San Antonio: Pearson.
Wefel, J. S., Vardy, J., Ahles, T., & Schagen, S. B. (2011a). International cognition and cancer task force recommendations to harmonise studies of cognitive function in patients with cancer. The Lancet Oncology, 12(7), 703–8. doi:10.1016/S1470-2045(10)70294-1.
Wefel, J. S., Vidrine, D. J., Veramonti, T. L., Meyers, C. A., Marani, S. K., Hoekstra, H. J., et al. (2011b). Cognitive impairment in men with testicular cancer prior to adjuvant therapy. Cancer, 117(1), 190–6. doi:10.1002/cncr.25298.
Wefel, J. S., Vidrine, D. J., Marani, S. K., Swartz, R. J., Veramonti, T. L., Meyers, C. A., et al. (2014). A prospective study of cognitive function in men with non-seminomatous germ cell tumors. Psycho-Oncology, 23(6), 626–33. doi:10.1002/pon.3453.
Wiens, A. N., Fuller, K. H., & Crossen, J. R. (1997). Paced auditory serial addition test: adult norms and moderator variables. Journal of Clinical and Experimental Neuropsychology, 19(4), 473–83. doi:10.1080/01688639708403737.
Wilson, C. J., Finch, C. E., & Cohen, H. J. (2002). Cytokines and cognition-the case for a head-to-toe inflammatory paradigm. Journal of the American Geriatrics Society, 50(12), 2041–2056. doi:10.1046/j.1532-5415.2002.50619.x.
Yuan, P., & Raz, N. (2014). Prefrontal cortex and executive functions in healthy adults: a meta-analysis of structural neuroimaging studies. Neuroscience and Biobehavioral Reviews, 42, 180–92. doi:10.1016/j.neubiorev.2014.02.005.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The study was funded by grants from the Danish Cancer Society (R17-A698) and by Saværksejer Jeppe Juhls og Hustru Ovita Juhls Mindelegat. Lisa M. Wu’s was supported by the National Cancer Institute of the National Institutes of Health under Award Number #7K07CA184145-02. Anders D. Børglum and Ditte Demontis were financially supported by The Lundbeck Foundation.
Conflict of interest
The authors declare that they have no conflict of interest.
Informed consent
Informed consent was obtained from all individual participants included in the study and performed procedures were in accordance with the ethical standards of The National Committee on Health Research Ethics and with the 1964 Helsinki declaration and its later amendments.
Rights and permissions
About this article
Cite this article
Amidi, A., Agerbæk, M., Wu, L.M. et al. Changes in cognitive functions and cerebral grey matter and their associations with inflammatory markers, endocrine markers, and APOE genotypes in testicular cancer patients undergoing treatment. Brain Imaging and Behavior 11, 769–783 (2017). https://doi.org/10.1007/s11682-016-9552-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11682-016-9552-3