Abstract
Amnestic mild cognitive impairment MCI (aMCI) has a high progression to Alzheimer’s disease (AD). Recently, resting-state functional MRI (RS-fMRI) has been increasingly utilized in studying the pathogenesis of aMCI, especially in resting-state networks (RSNs). In the current study, we aimed to explore abnormal RSNs related to memory deficits in aMCI patients compared to the aged-matched healthy control group using RS-fMRI techniques. Firstly, we used ALFF (amplitude of low-frequency fluctuation) method to define the regions of interest (ROIs) which exhibited significant changes in aMCI compared with the control group. Then, we divided these ROIs into different networks in line with prior studies. The aim of this study is to explore the functional connectivity between these ROIs within networks and also to investigate the connectivity between networks. Comparing aMCI to the control group, our results showed that 1) the hippocampus (HIPP) had decreased FC with the medial prefrontal cortex (mPFC) and inferior parietal lobe (IPL), and the mPFC showed increased connectivity to IPL in the default mode network; 2) the thalamus showed decreased FC with the putamen and HIPP, and the HIPP showed increased connectivity to the putamen in the limbic system; 3) the supplementary motor area had decreased FC with the middle temporal gyrus and increased FC with the superior parietal lobe in the sensorimotor network; 4) increased connectivity between the lingual gyrus and middle occipital gyrus in the visual network; and 5) the DMN has reduced inter-network connectivities with the SMN and VN. These findings indicated that functional brain networks involved in cognition such as episodic memory, sensorimotor and visual cognition in aMCI were altered, and provided a new sight in understanding the important subtype of aMCI.
Similar content being viewed by others
Introduction
Mild cognitive impairment (MCI) referring to patients who experience cognitive impairments, is a transitional phase between Alzheimer’s disease (AD) and healthy aged people (Feldman and Jacova, 2005; Petersen et al. 2001). Amnestic MCI (aMCI), which is one of several MCI subtypes, is characterized by primary memory impairment and has a high progression to AD at an annual rate between 6 %-25 % (Petersen et al. 2014). Neuroimaging studies have shown that patients with aMCI exhibit impairment in many cognitive functions such as memory, sensorimotor, and visual ability (Baker et al. 1996; Bokde et al. 2008; de Leon et al. 2001; Jin et al. 2012; Li et al. 2012).
Recently, resting-state fMRI (RS-fMRI) has been increasingly utilized in studying the pathogenesis of MCI patients. Li et al. (2002) first used RS-fMRI to study the abnormalities of intrinsic functional activity in aMCI subjects and observed reduced regional correlations of spontaneous brain activity in the hippocampus compared with a normal Control. song et al. (2013) also utilized RS-fMRI to investigate functional and structural changes of eight resting-state networks (RSNs) in aMCI/AD and found that brain networks supporting complex cognitive processes were progressively impaired due to brain regions forming tight interconnected neural circuits rather than isolated entities. Thus, it is important to explore interactions between brain regions, especially within the resting-state networks. More and more researchers regarded the network as an integral part to study neurophysiological diseases, and the results suggested that alterations of networks have been directly related to disease pathology. For example, the default mode network (DMN) has been cited in many introspective behaviors including episodic memory processing, stream of consciousness, and monitoring the internal and external environment (Andrews-Hanna et al. 2010; Buckner et al. 2008; Buckner and Carroll, 2007; Fair et al. 2008; Mazoyer et al. 2001; Raichle et al. 2001b; Raichle and Snyder, 2007; Shulman et al. 1997b), and the visual network (VN) is crucial for processing visual information relating to visual cognition (Petroni et al. 2001). AD/aMCI patients showed memory impairment and visual cognition deficits tightly related to the functional loss of the DMN and VN. Additionally, using a technique that combined anatomical MRI and positron emission tomography (PET), Peters et al. (2009) found severe reductions in metabolism throughout the network of limbic structures (e.g., the hippocampal complex and thalamus) which influences or produces emotion and also participates in the activities of learning and memory in patients with mild AD. In general, the research on RSNs can provide a broadened perspective on functional brain disorders in the field of neuroimaging.
One main approach being used to characterize the RSNs is the regions of interest (ROI) analysis, requiring a priori selection of an ROI. Previously, independent component analysis has been widely used in RS-fMRI to study the pathogenesis of patients with aMCI (Beckmann et al. 2005; Damoiseaux et al. 2006; Jafri et al. 2008; Jin et al. 2012; Zhao et al. 2012). However, this method explored the RS-fMRI signal from the aspect of temporal correlation, not from the aspect of regional activity (Zang et al. 2007). Subsequently, Zang et al. (2007) proposed amplitude low-frequency fluctuations (ALFF) to assess spontaneous brain activity in attention deficit hyperactivity disorder and found that it was able to represent different physiological states of the brain during resting-state. Using this method, many researchers successfully applied this index in many diseases, including AD and aMCI (Han et al. 2011b; Song et al. 2013; Wang et al. 2011; Zhao et al. 2014). On the other hand, functional connectivity was first proposed by Biswal et al. (1995), attracting the attention of many researchers to quantify the complex interaction between brain regions in RSNs.
Based on the analysis above, we used the ALFF method to explore abnormal brain regions and considered them as ROIs. Then, we divided these ROIs into different networks in line with prior studies. The aim of this study is to explore the functional connectivity between these ROIs within networks and also to investigate the functional connectivity between networks. We hypothesised that the functional connectivity within and between networks presents abnormal in aMCI patients compared to the control group.
Materials and methods
ADNI
Data used in the preparation of this article were obtained from the Alzheimer’s disease Neuroimaging Initiative (ADNI) database (adni.loni.usc.edu). ADNI was launched in 2003 by the National Institute on Aging (NIA), the National Institute of Biomedical Imaging and Bioengineering (NIBIB), the Food and Drug Administration (FDA), private pharmaceutical companies and non-profit organizations, such as a $60 million, 5-year public-private partnership. The primary goal of ADNI has been to test whether serial MRI, PET, other biological markers, and clinical and neuropsychological assessment can be combined to measure the progression of mild cognitive impairment (MCI) and early Alzheimer’s disease (AD). Determination of sensitive and specific markers of very early AD progression is intended to aid researchers and clinicians to develop new treatments and monitor their effectiveness, as well as lessen the time and cost of clinical trials. For up-to-date information, see www.adni-info.org.
Compliance with ethical standards
This study was carried out in accordance with the recommendations of ADNI database. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the study (www.adni-info.org). We also complied with ADNI’s requirement to “submit all manuscripts to the ADNI Database and Publications Committee (DPC) prior to submitting to a journal”.
Subjects
We downloaded 82 subjects’ (42 aMCI and 40 normal control subjects) 3 T functional MRI data and clinical data from the ADNI publicly available database (http://adni.loni.usc.edu/). Cognitive function was evaluated with the degree of dementia determined by the clinical dementia rating scale (CDR) and mini-mental state examination (MMSE) (Petersen et al. 2014). Among them, 39 aMCI patients and 38 normal control subjects were used in this study after 5 subjects (3 aMCI, 2 NC) were excluded due to excessive head motion (see data preprocessing). Table 1 shows more details of clinical and demographic data of the remaining subjects in the two groups. There are significant differences in MMSE and CDR scores, and No significant differences in gender or age were noted.
fMRI data and T1 data acquisition
MR images were acquired on a 3.0-Tesla Philips MRI scanner. Resting-state functional images were obtained by using an echo-planar imaging (EPI) sequence with the following scan parameters: repetition time (TR) = 3 s, echo time (TE) = 30 ms, flip angle (FA) = 80°, number of slices =48, slice thickness = 3.3 mm, voxel size = 3 × 3 × 3 mm3, and acquisition matrix =64 × 64. 3D T1-weighted magnetization-prepared rapid gradient echo (MP-RAGE) sagittal images were acquired by using the following parameters: TR = 6700 ms, TE = 3.1 ms, FA = 9°, slice thickness = 1.2 mm, matrix = 256 × 256 × 170. All original image files are available to the general scientific community.
Data preprocessing
The data preprocessing steps are similar with our previous studies (Cai et al. 2015; Cai et al. 2014). Data preprocessing was carried out using the Data Processing Assistant for Resting-State fMRI (DPARSF, http://rfmri.org/DPARSF) (Chao-Gan and Yu-Feng, 2010), which is based on Statistical Parametric Mapping (SPM8) (http://www.fil.ion.ucl.ac.uk/spm) and Resting-State fMRI Data Analysis Toolkit (REST, http://www.restfmri.net) (Song et al. 2011). Initially, the first 10 volumes of the functional images were discarded due to signal equilibration of the magnetic field. The remaining functional images had slice-timing correction done for different signal acquisition between each slice and motion effects (six-parameter rigid body). Participants with head motion exceeding 1.5 mm in any dimension of x, y and z or 1.5° in any angular motion through the resting-state were excluded for further analysis. Next, the remaining images (39 aMCI patients and 38 normal subjects) were spatially normalized to the Montreal neurological institute (MNI) space by a T1 anatomical image, which had been co-registered to the mean functional image after motion correction using a linear transformation and re-sampled to a voxel size of 3 mm × 3 mm × 3 mm. Finally, the functional images were smoothed with a Gaussian kernel of 6 mm full width at half maximum (FWHM) to decrease spatial noise. After the linear trends had been removed to wipe out any residual effects of motion and other non-neuronal factors, a band-pass filter (0.01 Hz < f < 0.08 Hz) was applied to the time series of each voxel to reduce the effect of low-frequency drifts and high-frequency noise such as respiratory and cardiac rhythms. Finally, the nuisance covariates, including 6 head motion parameters, global mean signals, white matter signals and cerebrospinal fluid, were regressed for the next analysis. Since the T1 images were used as an anatomical reference, T1-weighted MRI images were obtained using the FSL tool ‘BET’ (http://fsl.fmrib.ox.ac.uk/fsl/).
Head motion may have impact on resting-state fMRI metrics, such as the method of functional connectivity (Power et al. 2012; Power et al. 2015; Satterthwaite et al. 2012; Van Dijk et al. 2012). In order to examine if there are any differences in head motion between the two groups, we extracted the head motion information of each subject in the two groups. Then, we tested the group differences in head motion followed those in Power et al. (2012)’s study and found that there are no significant differences in head motion between the two groups (Table 1).
ALFF analysis
The ALFF analysis was calculated using REST software to select the ROIs. Briefly, the time series was converted to the frequency domain using a Fast Fourier Transform. The square root of the power spectrum was computed and then averaged across 0.01–0.08 Hz at each voxel. This averaged square root was taken as the ALFF. To reduce the global effects of variability across subjects, the ALFF of each voxel was divided by the global mean ALFF (mALFF) value for each participant, generating a relative ALFF. Finally, we extracted the grey volume of these ROIs using FMRIB’s Software Library (FSL) (http://fsl.fmrib.ox.ac.uk/fsl/) and then contrasted that between aMCI and the control group to make assistant analyses.
Functional connectivity analysis within and between networks
When we obtained the ROIs that showed significant ALFF differences between the two groups, we marked off these ROIs into four networks. Then, a functional connectivity analysis was carried out within and between these networks. We extracted the time series of each ROI, and then the temporal correlation between paired-wise of ROIs was calculated on the voxel time series within each network. For each of the network, the network signal was defined as the mean time series of the ROIs in the same network. The between-network connectivity strength of each subject was calculated by Pearson’s correlation between each pair of network signal. Design of the current study is in Fig. 1.
Design of the current study. Firstly, we preprocessed the necessary data. Then, we conducted the ALFF analysis and compared the baseline brain activity level between aMCI and control groups. Thirdly, among these 12 altered brain regions (12 ROIs), we marked off four resting state networks (RSNs) including the default mode network (DMN), limbic system, sensorimotor network (SMN) and visual network (VN). Fourthly, functional connectivity (FC) and functional network connectivity (FNC) analysis were carried out within each RSN and between networks respectively. Then we obtained the altered FC within and between networks in the aMCI group. Finally, we made a correlation analysis between the FC strength and the clinical variable in the aMCI group
Relationship between functional connectivity strength and clinical variable
We performed a correlation analysis between functional connectivity strength and the clinical variable (MMSE) to explore whether the functional connectivity strength had a relationship with the clinical variable. The clinical variable was the Mini-mental State Examination (MMSE). We also performed a correlation analysis between functional network connectivity strength and the MMSE. We used a statistical significance level of p < 0.05 and corrected the p values using with multiple comparison correction (False Discovery Rate (FDR) correction).
Statistical analysis
After obtaining the mALFF of the two groups, a two-sample t-test was applied to compare the differences in mALFF between patients with aMCI and normal control subjects with ages and grey volumes as the covariance (threshold: k = 30 voxels, P < 0.05, FDR corrected). We averaged the FC strength across the subjects of the two groups, and then obtained the functional connectivity value between each pair of ROIs each pair of networks. We used a Fisher’s r-to-z to convert the correlation coefficients to z values to improve the normality (Lowe et al. 1998). We applied a two-sample t-test with the grey volumes of paired regions and ages as the covariance to detect the FC difference of each pair of ROIs and each pair of networks between the aMCI and control group. Then, we corrected each p value with multiple comparison correction (P < 0.05, FDR correction).
Results
Altered ALFF in aMCI and selection of RSNs
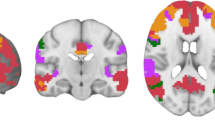
Compared with normal controls, the patients with aMCI exhibited a significant difference in ALFF including the left middle temporal gyrus (MTG), left middle occipital gyrus (MOG), left and right superior parietal lobe (SPL), right inferior parietal lobe (IPL), right hippocampus (HIPP), left and right putamen (PUT), right thalamus (THAL), left medial prefrontal cortex (mPFC), supplementary motor area (SMA) and left lingual gyrus (threshold: k = 30 voxels, P < 0.05, FDR corrected). Please refer to Fig. 2 to acquire more information. Among these altered brain regions, we marked off four networks including the DMN, limbic system, sensorimotor network (SMN) and VN: 1) the left mPFC, right IPL and right HIPP belong to the DMN; 2) right HIPP, right THAL, and left and right putamen belong to the limbic system; 3) right SMA, left MTG, and left and right SPL belong to the SMN; 4) right lingual gyrus and left MOG belong to the VN (Allen et al. 2011; Buckner et al. 2008; Greicius et al. 2003a; Gusnard et al. 2001; Hänggi et al. 2010; Raichle et al. 2001b). In addition, we contrasted the grey volume of these ROIs between aMCI and the control group and found that the right HIPP and right putamen exhibited voluminal atrophy. See Fig. 3 for more information.
Brain regions of interest showing significant ALFF differences comparing the aMCI group with the NC group. The red color represents increased ALFF and blue color represents decreased ALFF. The statistical threshold was set at p < 0.05 and a minimum cluster size of 30 voxels (FDR corrected). All of the abbreviations are shown in Table 2
Comparison of the grey volume of 12 ROIs between the aMCI and control groups. The grey volume was obtained by cross-subject averaging for aMCI (n = 39, light blue bar) and control (n = 38, light green bar) groups. Error bars represent the standard deviation. The R.HIPP and right putamen showed significant differences in grey volume between the two groups by the two-sample t-tests (p < 0.05). All of the abbreviations are shown in Table 2
Altered functional connectivity within and between networks in the aMCI group
We found altered functional connectivity within four networks in the aMCI group relative to the control group. Our results showed that: 1) the HIPP had decreased FC with the mPFC and IPL, and increased connectivity between the mPFC and IPL in the default mode network; 2) the thalamus showed decreased FC with the putamen and HIPP, and increased connectivity between the HIPP and putamen in the limbic system; 3) the SMA had decreased FC with the MTG and increased FC with the right SPL in the sensorimotor network; and 4) increased connectivity between the lingual gyrus and middle occipital gyrus in the visual network. For more information please see Fig. 4. Besides, we also detected reduced inter-network connectivities between the DMN and SMN, and between the DMN and VN in aMCI group. No significant differences of inter-network connectivity were found in other networks (Fig. 5).
Altered functional connectivity within the four networks in the aMCI group. Compared with the control group, the aMCI group showed that: 1) the R.HIPP had decreased FC with the L.mPFC and R.IPL (blue lines), and the L.mPFC showed increased connectivity to the R.IPL (red lines) in the default mode network; 2) the R.THAL showed decreased FC with the putamen and HIPP (blue lines), and the HIPP showed increased connectivity to the putamen (red lines) in the limbic system; 3) the R.SMA had decreased FC with the L.MTG (blue lines) and increased FC with the right SPL (red lines) in the sensorimotor network; and 4) increased connectivity between the right lingual gyrus and L.MOG (red lines) in the visual network (p < 0.05). The number on the lines stands for the statistic p value. No significant FC alterations were observed in the other connections. All of the abbreviations are shown in Table 2
Bar graph of the DMN-SMN and DMN-VN connectivity strength in the two groups. The functional network connectivity strength of DMN-SMN and DMN-VN differed significantly in the control group compared with the aMCI (p = 0.019 and p = 0.026) groups. The aMCI group is indicated by black rectangles, the control group is indicated by white rectangles. The error bars represent the standard error of each group
Relationship between FC Strength and clinical variables
To determine the relationship between FC strength and MMSE scores of aMCI, we extracted the average FC strength of all voxels between each pair of ROIs. Several connections which had remarkable positive correlations between FC values and MMSE scores were detected in the aMCI group. These FCs included the connection between the left mPFC and right HIPP and the connection between the right SMA and left MTG. No significant correlations were detected in other connections. For more information refer to Fig. 6. Besides, there are no significant correlations between functional network connectivity strength and the MMSE.
The correlation between the functional connectivity strength and MMSE scores in the aMCI group (p < 0.05). L.mPFC—R.HIPP: the functional connectivity strength between the left medial prefrontal cortex and right hippocampus (r = 0.496, p = 0.01); R.SMA—L.MTG: the functional connectivity strength between the right supplementary motor area and left middle temporal gyrus (r = 0.362, p = 0.021)
Discussion
In the current study, we employed ALFF to define the ROIs which exhibited significant changes in spontaneous brain activity when comparing aMCI with the control group. Then, we divided these ROIs into different networks in line with prior studies. The aim of this study is to explore the functional connectivity between these ROIs within networks and also to investigate the functional connectivity between networks. We found that 1) the altered brain regions were distributed within four networks, which included the DMN, SMN, VN and limbic system; 2) the functional connectivity within the four networks was changed in the aMCI group; and 3) the inter-network connectivity between several networks was changed. The evidence indicated that there were differences in the resting state networks related to cognition compared with the age-matched control group.
Altered ALFF in aMCI
In this study, we found that there were a set of regions showed significant differences in spontaneous brain activity when comparing aMCI with the control group (Fig. 2 and Table 2). The decreased brain spontaneous activity in the mPFC, HIPP, THAL and putamen are consistent with previous studies (Han et al. 2011b; Rombouts et al. 2007; Wang et al. 2011). The reduced ALFF in the lingual gyrus is a new finding in the current study. The lingual gyrus is an important region to process visual function and destruction of it can affect visual processing. In addition, we also detected increased ALFF in MTG, SPL and MOG. Wang et al. (2011) found increased activity in the lateral temporal regions and superior parietal regions in aMCI as compared with controls. A previous RS-fMRI study has revealed that aMCI patients had increased connectivity in the middle temporal region relative to the control group (Qi et al. 2010). Using PET, Truchot et al. (2008) observed a significant increase in metabolism in the occipital region in aMCI. Likewise, increased occipital activities in AD were also found during the performance of cognitive tasks (Backman et al. 2000; Prvulovic et al. 2002). These previous results provided further support for our findings. Moreover, our findings of increased ALFF in aMCI suggested that the patients had relative preservation of brain spontaneous activity in these regions.
Altered functional connectivity within the RSNs in aMCI
The DMN has the highest metabolic activity at rest (Raichle et al. 2001a) and presents “deactivation” during various tasks (O’Callaghan et al. 2015; Shulman et al. 1997a). Connectivity of the DMN plays a pivotal role in the activity of human cognition such as the integration of short-term memory and episodic retrieval (Greicius et al. 2003b). Dunn et al. (2014) have revealed that deficits in episodic memory retrieval is related to impaired DMN connectivity in amnestic MCI. In this study, we found that patients with aMCI showed decreased connectivity between the right HIPP and left mPFC, and between the right HIPP and right IPL suggested that the connectivity of DMN appeared abnormal. The abnormal connectivity in DMN might have relationship with the deficits in episodic memory retrieval. The HIPP is an important brain region in the DMN. Hirano et al. (1962) found that neurofibrillary tangles selectively afflict specific cortical layers in the HIPP. Lacoste et al. (1993) demonstrated that these specific cortical layers serve as the gateway of neural projection between the HIPP and the rest of the brain. Therefore, damage in the specific cortical layers might affect the connectivity between the HIPP and the other cerebral cortex (Hyman et al. 1986; Hyman et al. 1984) and enhance the probability that functional interactions between them might be abnormal in patients with aMCI (Delbeuck et al. 2003). The mPFC and IPL showed decreased connectivity relative to the HIPP which most likely contributed to the impact of specific cortical layers in the HIPP. Moreover, there is some evidence demonstrating that the mPFC is involved in episodic retrieval, especially the autobiographical event from neuroimaging studies (Cabeza et al. 2004; Levine et al. 2004). For example, Cabeza et al. (2004) detected aberrant brain activity in a network including the HIPP, mPFC, bilateral visual cortices and IPL during an autobiographic retrieval task. The disconnected brain areas which we found in the current study overlapped considerably with these aberrant regions. More importantly, we examined the loss of grey volume in the HIPP (Fig. 4). Atrophy is regarded as an important marker of neurodegenerative pathology. In addition to HIPP atrophy, we also found the FC strength between the HIPP and mPFC had a positive correlation with clinical scores (Fig. 6). Combining these facts with our finding, the decreased HIPP connectivity with the mPFC and IPL suggested that decreased activity of the DMN contributed to episodic memory impairment in patients. In addition to the reduced connectivity within the DMN, increased connectivity between the mPFC and right IPL was also detected in aMCI patients. The mPFC is vital for episodic retrieval and the IPL is reported to be involved in short-term memory storage and retrieval (Todd and Marois, 2004). Previous studies showed that the early stage of AD patients presented increased functional connectivity within the prefrontal regions during various memory tasks (Pariente et al. 2005). This increased connectivity has been interpreted to be compensatory recruitment of cognitive resources to maintain task performance (Gould et al. 2006; Grady et al. 2003). The increased interactions between the mPFC and IPL observed in our study potentially indicated that patients possess a relative boost in short-term memory function compared with the control group. The enhanced short-term memory temporarily remedied its defect in long-term memory. With the worsening of the disease, short-term memory would eventually be lost. Our findings provided further support that aMCI patients could utilize additional brain resources in the frontal lobe to compensate for the deficit in cognitive function. Moreover, increased connectivity between the mPFC and IPL was consistent with one of our previous studies (Zhong et al. 2014).
In the limbic system, the right THAL exhibited decreased connectivity to the right HIPP and right putamen. The HIPP is the primary brain region of neuronal degeneration in Alzheimer’s disease (Braak et al. 2006). As we mentioned before, we also examined grey volume atrophy in the HIPP (Fig. 4). Moreover, Zarei et al. (2010) successfully detected regional THAL atrophy in AD combining shape and connectivity methods. The decreased connectivity between the HIPP and THAL might be due to grey matter atrophy suggesting altered degeneration in the thalamo-hippocampal circuit. This result was consistent with a previous study of THAL functional connectivity which also detected decreased connectivity between the THAL and HIPP (Cai et al. 2014). Besides, a new findings is the right putamen exhibited decreased connectivity to its other side and THAL (Fig. 4). For many years, the functional connectivity of putamen very little has been reported in AD patients. Some previous literatures showed that the putamen, as part of the striatum, is observed as being active in working memory tasks (Dahlin et al. 2008) and probabilistic learning tasks (Bellebaum et al. 2008; Graybiel, 2005). Furthermore, the putamen was correlated with the occurrence of dementia in other neurodegenerative disorders, such as Parkinson’s disease (Emre 2003). In addition, we demonstrated that the volume of putamen is reduced in aMCI group. This finding suggested that deep grey matter structure in aMCI suffer atrophy and is consistent with a result of putamen reported by Jong et al. (2008). Thus, our finding of impaired functional connectivity and volumetric atrophy of putamen together with the recognition that AD pathology hinted that the putamen is an important brain region worthy of our attention. In contrast to the putamen, more information is known on the THAL and its cognitive functions involved in suppressing irrelevant sensory input and directing attention (Newman, 1995). Grey matter structures in aMCI suffered atrophy in the putamen and a reduced connection between putamen and the THAL indicated that degenerative processes in these regions, like the HIPP, might lead to cognitive decline in Alzheimer’s disease. In addition to decreased connectivity, we found increased connectivity between the right HIPP and putamen as well. The enhanced connectivity is in order to compensate the reduced connectivity from the right HIPP and right putamen to the THAL which we have mentioned above. In summary, alterations in the limbic system, which is implicated in working memory, learning and some other cognitive functions, could explain a set of the clinical characteristics of AD.
In the SMN, likewise, we also detected several abnormal connections. A recent work conducted by Wang et al. (2015) reported that intra-network connectivity of SMN were impaired in AD and then extended to the key functional systems in the AD individuals. In the current study, we confirmed the abnormal connectivities within SMN. The SMA is mainly involved in movement of production and control (Luppino et al. 1993) and the role of the superior parietal lobe (SPL) is to maintain internal representations (Wolpert et al. 1998). Yetkin et al. (2006) found that aMCI patients had more functional activation in the SMA during a working memory task as compared with controls. The increased functional connectivity between the SMA and right SPL has been proposed as a compensatory reallocation or recruitment of motion and sensation cognitive resources in aMCI patients. Except for increased connectivity, we found the SMA had decreased connectivity with the left middle temporal gyrus (MTG). Felleman et al. (1987) demonstrated that the middle temporal visual area of primates plays a remarkable role in the visual motion function using physiological experiments, and Newsome et al. (1988) observed there was selective damage of motion perception following lesions of the middle temporal visual area. Thus, the decreased connectivity between the SMA and MTG might be due to lesions in the MTG. Interestingly, we also observed that the functional connectivity strength between SMA and MTG had a positive correlation with MMSE scores (Fig. 6). Combining our findings and previous studies, we could obtain that the changes in functional connectivity had been expanded to the primary motor region in aMCI patients.
In addition, we detected increased connectivity between the right lingual gyrus and left middle occipital gyrus (MOG) in the visual network. The lingual gyrus is a significant brain area to process visual function and bilateral destruction of the lingual gyrus can affect complex visual processing. However, a deficit in the lingual gyrus alone is not sufficient to lead to impaired visual processing. A study reported that the MOG is located in the primary visual cortex and appears to be involved in processing visual recognition. The abnormal connectivity between them would lead to impaired visual cognition in aMCI patients. This interpretation supported and extended several previous studies indicating that AD could impact many aspects of visual processing, such as perceptual discrimination, spatial frequency, colors, orientation and objects (A., 1995; Cronin-Golomb et al. 1993; Kurylo et al. 1994; Rizzo et al. 2000). From another aspect, Golby et al. (2005) reported that the AD group presented intact implicit memory, as well as intact implicit encoding in the occipital cortex. The increased connectivity between the MOG and lingual gyrus might help guarantee intact implicit memory in patients.
Altered functional network connectivity between RSNs in aMCI
Besides, we also detected the DMN has reduced inter-network connectivities with the SMN and VN in aMCI group (Fig. 5). Resting-state functional connectivity and network analysis provides an important tool for mapping large-scale function and dysfunction in the brain system. The phenomenon of abnormal inter-network connectivity between the DMN and the SMN and VN prompted us to rethink the role of the interactions between the RSNs in understanding AD pathology. Upon we have mentioned above, The DMN plays a key role in cognitive processes, and the SMN, composed of the primary visual, auditory and somatomotor cortices, plays a role in receiving external signals which could aid in perceiving the world, selecting the relevant information and determining the target. The visual network mainly involved in processing and integrating visual information (Greicius et al. 2003b; Petroni et al. 2001; Wolpert et al. 1995). The multiple integrations of cognitive systems, sensorimotor and visual cognition provide the signals for the organism to perceive and respond to its environment. In other words, deficits in any of these components will result in impaired function at the clinical level (Albers et al. 2015; Cabeza et al. 2008). Usually, lesions in memory, vision and sensory or motor are seen as features of aging; clinically, the performance of these functions is poor in MCI patients and even worse in AD (Galton et al. 2000). Our research prompted us to infer that the impaired connectivity within the SMN, VN and DMN or reduced connectivity between them might be one of the reasons for the cognitive deficits in the early stages of AD. Moreover, convergence evidence has indicated that AD pathology develops over many years, raising the infusive possibility that daily cognitive deficits, even very slight such as lessened sensorimotor and visual ability may be early, noninvasive markers for AD (Albers et al. 2015).
Two concerns need to be taken into account in the current study. First, we removed the global signal as this regressor might be associated with variance in fMRI functional connectivity analyses (Desjardins et al. 2001). However, there is much debate about regressing the global mean signal. We will explore this issue in the future. Second, there are also a set of studies applying whole-brain connectivity to investigate the brain mechanism of neurodegenerative disorders such as (Bai et al. 2011). We recognized the importance of global (whole-brain) connectivity. We will carry out our research by using the global connectivity to study the pathogenesis of AD and find potential biomarkers to diagnose and predict AD.
Conclusions
In summary, the purpose of the current study was to explore the functional alterations on the integrity of resting-state networks in the aMCI relative to the control group. Our results demonstrated that the patient group showed altered functional brain networks involved in cognition such as memory, sensorimotor and visual cognition including the DMN, limbic system, SMN and VN networks. These findings provided new insight in perceiving the crucial subtype of aMCI.
References
A., C.-G. (1995). Vision in Alzheimer’s disease. The Gerontologist, 35, 370–376.
Albers, M. W., Gilmore, G. C., Kaye, J., Murphy, C., Wingfield, A., Bennett, D. A., Boxer, A. L., Buchman, A. S., Cruickshanks, K. J., & Devanand, D. P. (2015). At the interface of sensory and motor dysfunctions and Alzheimer’s disease. Alzheimer's & Dementia, 11, 70–98.
Allen, E. A., Erhardt, E. B., Damaraju, E., Gruner, W., Segall, J. M., Silva, R. F., Havlicek, M., Rachakonda, S., Fries, J., Kalyanam, R., Michael, A. M., Caprihan, A., Turner, J. A., Eichele, T., Adelsheim, S., Bryan, A. D., Bustillo, J., Clark, V. P., Feldstein Ewing, S. W., Filbey, F., Ford, C. C., Hutchison, K., Jung, R. E., Kiehl, K. A., Kodituwakku, P., Komesu, Y. M., Mayer, A. R., Pearlson, G. D., Phillips, J. P., Sadek, J. R., Stevens, M., Teuscher, U., Thoma, R. J., & Calhoun, V. D. (2011). A baseline for the multivariate comparison of resting-state networks. Frontiers in Systems Neuroscience, 5, 2.
Andrews-Hanna, J. R., Reidler, J. S., Huang, C., & Buckner, R. L. (2010). Evidence for the default network’s role in spontaneous cognition. Journal of Neurophysiology, 104, 322–335.
Backman, L., Almkvist, O., Nyberg, L., & Andersson, J. (2000). Functional changes in brain activity during priming in Alzheimer’s disease. Journal of Cognitive Neuroscience, 12, 134–141.
Bai, F., Liao, W., Watson, D. R., Shi, Y., Wang, Y., Yue, C., Teng, Y., Wu, D., Yuan, Y., & Jia, J. (2011). Abnormal whole-brain functional connection in amnestic mild cognitive impairment patients. Behavioural Brain Research, 216, 666–672.
Baker, S. C., Rogers, R. D., Owen, A. M., Frith, C. D., Dolan, R. J., Frackowiak, R. S., & Robbins, T. W. (1996). Neural systems engaged by planning: a PET study of the Tower Of London task. Neuropsychologia, 34, 515–526.
Beckmann, C. F., DeLuca, M., Devlin, J. T., & Smith, S. M. (2005). Investigations into resting-state connectivity using independent component analysis. Philosophical Transactions of the Royal Society Of London B: Biological Sciences, 360, 1001–1013.
Bellebaum, C., Koch, B., Schwarz, M., & Daum, I. (2008). Focal basal ganglia lesions are associated with impairments in reward-based reversal learning. Brain, 131, 829–841.
Biswal, B., Yetkin, F.Z., Haughton, V.M., Hyde, J.S., 1995. Functional connectivity in the motor cortex of resting human brain using echo-planar MRI. Magnetic Resonance in Medicine: Official Journal of the Society Of Magnetic Resonance in Medicine/Society Of Magnetic Resonance in Medicine 34, 537–541.
Bokde, A. L., Lopez-Bayo, P., Born, C., Dong, W., Meindl, T., Leinsinger, G., Teipel, S. J., Faltraco, F., Reiser, M., Moller, H. J., & Hampel, H. (2008). Functional abnormalities of the visual processing system in subjects with mild cognitive impairment: an fMRI study. Psychiatry Research, 163, 248–259.
Braak, H., Alafuzoff, I., Arzberger, T., Kretzschmar, H., & Del Tredici, K. (2006). Staging of Alzheimer disease-associated neurofibrillary pathology using paraffin sections and immunocytochemistry. Acta Neuropathologica, 112, 389–404.
Buckner, R. L., Andrews-Hanna, J. R., & Schacter, D. L. (2008). The brain’s default network: anatomy, function, and relevance to disease. Annals of the New York Academy of Sciences, 1124, 1–38.
Buckner, R. L., & Carroll, D. C. (2007). Self-projection and the brain. Trends in Cognitive Sciences, 11, 49–57.
Cabeza, R., Ciaramelli, E., Olson, I. R., & Moscovitch, M. (2008). The parietal cortex and episodic memory: an attentional account. Nature Reviews Neuroscience, 9, 613–625.
Cabeza, R., Prince, S. E., Daselaar, S. M., Greenberg, D. L., Budde, M., Dolcos, F., LaBar, K. S., & Rubin, D. B. (2004). Brain activity during episodic retrieval of autobiographical and laboratory events: an fMRI study using a novel photo paradigm. Journal of Cognitive Neuroscience, 16, 1583–1594.
Cai, S., Huang, L., Zou, J., Jing, L., Zhai, B., Ji, G., von Deneen, K. M., Ren, J., Ren, A., & Initiative, A.s. D. N. (2014). Changes in thalamic connectivity in the early and late stages of amnestic mild cognitive impairment: A resting-state functional magnetic resonance study from ADNI. PloS One, 10, e0115573–e0115573.
Cai, S., Chong, T., Zhang, Y., Li, J., von Deneen, K. M., Ren, J., Dong, M., Huang, L., & Initiative, A. s. D. N. (2015). Altered functional connectivity of fusiform gyrus in subjects with amnestic mild cognitive impairment: a resting-state fMRI study. Frontiers in Human Neuroscience, 9, 471.
Chao-Gan, Y., & Yu-Feng, Z. (2010). DPARSF: A MATLAB toolbox for "pipeline" data analysis of resting-state fMRI. Frontiers in Systems Neuroscience, 4, 13.
Cronin-Golomb, A., Sugiura, R., Corkin, S., & Growdon, J. H. (1993). Incomplete achromatopsia in Alzheimer’s disease. Neurobiology of Aging, 14, 471–477.
Dahlin, E., Neely, A. S., Larsson, A., Bäckman, L., & Nyberg, L. (2008). Transfer of learning after updating training mediated by the striatum. Science, 320, 1510–1512.
Damoiseaux, J., Rombouts, S., Barkhof, F., Scheltens, P., Stam, C., Smith, S. M., & Beckmann, C. (2006). Consistent resting-state networks across healthy subjects. Proceedings of the National Academy of Sciences, 103, 13848–13853.
De Jong, L., Van der Hiele, K., Veer, I., Houwing, J., Westendorp, R., Bollen, E., De Bruin, P., Middelkoop, H., Van Buchem, M., & Van Der Grond, J. (2008). Strongly reduced volumes of putamen and thalamus in Alzheimer’s disease: an MRI study. Brain, 131, 3277–3285.
de LaCoste, M.-C., & White, C. L. (1993). The role of cortical connectivity in Alzheimer’s disease pathogenesis: a review and model system. Neurobiology of Aging, 14, 1–16.
de Leon, M. J., Convit, A., Wolf, O. T., Tarshish, C. Y., DeSanti, S., Rusinek, H., Tsui, W., Kandil, E., Scherer, A. J., Roche, A., Imossi, A., Thorn, E., Bobinski, M., Caraos, C., Lesbre, P., Schlyer, D., Poirier, J., Reisberg, B., & Fowler, J. (2001). Prediction of cognitive decline in normal elderly subjects with 2-[(18)F]fluoro-2-deoxy-D-glucose/poitron-emission tomography (FDG/PET). Proceedings of the National Academy of Sciences of the United States of America, 98, 10966–10971.
Delbeuck, X., Van der Linden, M., & Collette, F. (2003). Alzheimer’disease as a disconnection syndrome? Neuropsychology Review, 13, 79–92.
Desjardins, A. E., Kiehl, K. A., & Liddle, P. F. (2001). Removal of confounding effects of global signal in functional MRI analyses. NeuroImage, 13, 751–758.
Dunn, C. J., Duffy, S. L., Hickie, I. B., Lagopoulos, J., Lewis, S. J., Naismith, S. L., & Shine, J. M. (2014). Deficits in episodic memory retrieval reveal impaired default mode network connectivity in amnestic mild cognitive impairment. NeuroImage: Clinical, 4, 473–480.
Emre, M. (2003). What causes mental dysfunction in Parkinson’s disease? Movement disorders, 18, 63–71.
Fair, D. A., Cohen, A. L., Dosenbach, N. U., Church, J. A., Miezin, F. M., Barch, D. M., Raichle, M. E., Petersen, S. E., & Schlaggar, B. L. (2008). The maturing architecture of the brain’s default network. Proceedings of the National Academy of Sciences of the United States of America, 105, 4028–4032.
Feldman, H. H., & Jacova, C. (2005). Mild cognitive impairment. The American Journal of Geriatric Psychiatry: Official Journal of the American Association For Geriatric Psychiatry, 13, 645–655.
Felleman, D. J., & Van Essen, D. C. (1987). Receptive field properties of neurons in area V3 of macaque monkey extrastriate cortex. Journal of Neurophysiology, 57, 889–920.
Galton, C. J., Patterson, K., Xuereb, J. H., & Hodges, J. R. (2000). Atypical and typical presentations of Alzheimer’s disease: a clinical, neuropsychological, neuroimaging and pathological study of 13 cases. Brain, 123, 484–498.
Golby, A., Silverberg, G., Race, E., Gabrieli, S., O’Shea, J., Knierim, K., Stebbins, G., & Gabrieli, J. (2005). Memory encoding in Alzheimer’s disease: an fMRI study of explicit and implicit memory. Brain, 128, 773–787.
Gould, R., Arroyo, B., Brown, R., Owen, A., Bullmore, E., & Howard, R. (2006). Brain mechanisms of successful compensation during learning in Alzheimer disease. Neurology, 67, 1011–1017.
Grady, C. L., McIntosh, A. R., Beig, S., Keightley, M. L., Burian, H., & Black, S. E. (2003). Evidence from functional neuroimaging of a compensatory prefrontal network in Alzheimer’s disease. The Journal of Neuroscience, 23, 986–993.
Graybiel, A. M. (2005). The basal ganglia: learning new tricks and loving it. Current Opinion in Neurobiology, 15, 638–644.
Greicius, M. D., Krasnow, B., Reiss, A. L., & Menon, V. (2003a). Functional connectivity in the resting brain: a network analysis of the default mode hypothesis. Proceedings of the National Academy of Sciences of the United States of America, 100, 253–258.
Greicius, M. D., Krasnow, B., Reiss, A. L., & Menon, V. (2003b). Functional connectivity in the resting brain: a network analysis of the default mode hypothesis. Proceedings of the National Academy of Sciences, 100, 253–258.
Gusnard, D. A., Akbudak, E., Shulman, G. L., & Raichle, M. E. (2001). Medial prefrontal cortex and self-referential mental activity: relation to a default mode of brain function. Proceedings of the National Academy of Sciences of the United States of America, 98, 4259–4264.
Hänggi, J., Koeneke, S., Bezzola, L., & Jäncke, L. (2010). Structural neuroplasticity in the sensorimotor network of professional female ballet dancers. Human Brain Mapping, 31, 1196–1206.
Han, Y., Wang, J., Zhao, Z., Min, B., Lu, J., Li, K., He, Y., & Jia, J. (2011b). Frequency-dependent changes in the amplitude of low-frequency fluctuations in amnestic mild cognitive impairment: a resting-state fMRI study. NeuroImage, 55, 287–295.
Hirano, A., & Zimmerman, H. (1962). Alzheimer’s neurofibrillary changes: a topographic study. Archives of Neurology, 7, 227–242.
Hyman, B., Van Hoesen, G., Kromer, L., & Damasio, A. (1986). Perforant pathway changes and the memory impairment of Alzheimer’s disease. Annals of Neurology, 20, 472–481.
Hyman, B. T., Van Hoesen, G. W., Damasio, A. R., & Barnes, C. L. (1984). Alzheimer’s disease: cell-specific pathology isolates the hippocampal formation. Science, 225, 1168–1170.
Jafri, M. J., Pearlson, G. D., Stevens, M., & Calhoun, V. D. (2008). A method for functional network connectivity among spatially independent resting-state components in schizophrenia. NeuroImage, 39, 1666–1681.
Jin, M., Pelak, V. S., & Cordes, D. (2012). Aberrant default mode network in subjects with amnestic mild cognitive impairment using resting-state functional MRI. Magnetic Resonance Imaging, 30, 48–61.
Kurylo, D. D., Corkin, S., Dolan, R. P., Rizzo, J. F., Parker, S. W., & Growdon, J. H. (1994). Broad-band visual capacities are not selectively impaired in Alzheimer’s disease. Neurobiology of Aging, 15, 305–311.
Levine, B., Turner, G. R., Tisserand, D., Hevenor, S. J., Graham, S., & McIntosh, A. R. (2004). The functional neuroanatomy of episodic and semantic autobiographical remembering: a prospective functional MRI study. Cognitive Neuroscience, Journal of, 16, 1633–1646.
Li, R., Wu, X., Fleisher, A. S., Reiman, E. M., Chen, K., & Yao, L. (2012). Attention-related networks in Alzheimer’s disease: a resting functional MRI study. Human Brain Mapping, 33, 1076–1088.
Li, S. J., Li, Z., Wu, G., Zhang, M. J., Franczak, M., & Antuono, P. G. (2002). Alzheimer disease: evaluation of a functional MR imaging index as a marker. Radiology, 225, 253–259.
Lowe, M. J., Mock, B. J., & Sorenson, J. A. (1998). Functional connectivity in single and multislice echoplanar imaging using resting-state fluctuations. NeuroImage, 7, 119–132.
Luppino, G., Matelli, M., Camarda, R., & Rizzolatti, G. (1993). Corticocortical connections of area F3 (SMA-proper) and area F6 (pre-SMA) in the macaque monkey. Journal of Comparative Neurology, 338, 114–140.
Mazoyer, B., Zago, L., Mellet, E., Bricogne, S., Etard, O., Houde, O., Crivello, F., Joliot, M., Petit, L., & Tzourio-Mazoyer, N. (2001). Cortical networks for working memory and executive functions sustain the conscious resting state in man. Brain Research Bulletin, 54, 287–298.
Newman, J. (1995). Thalmic contributions to attention and consciousness. Consciousness and Cognition, 4, 172–193.
Newsome, W. T., & Pare, E. B. (1988). A selective impairment of motion perception following lesions of the middle temporal visual area (MT). The Journal of Neuroscience, 8, 2201–2211.
O’Callaghan, C., Shine, J. M., Lewis, S. J., Andrews-Hanna, J. R., & Irish, M. (2015). Shaped by our thoughts–A new task to assess spontaneous cognition and its associated neural correlates in the default network. Brain and Cognition, 93, 1–10.
Pariente, J., Cole, S., Henson, R., Clare, L., Kennedy, A., Rossor, M., Cipoloti, L., Puel, M., Demonet, J. F., & Chollet, F. (2005). Alzheimer’s patients engage an alternative network during a memory task. Annals of Neurology, 58, 870–879.
Peters, F., Collette, F., Degueldre, C., Sterpenich, V., Majerus, S., & Salmon, E. (2009). The neural correlates of verbal short-term memory in Alzheimer’s disease: an fMRI study. Brain, 132, 1833–1846.
Petersen, R., Caracciolo, B., Brayne, C., Gauthier, S., Jelic, V., & Fratiglioni, L. (2014). Mild cognitive impairment: a concept in evolution. Journal of Internal Medicine, 275, 214–228.
Petersen, R. C., Doody, R., Kurz, A., Mohs, R. C., Morris, J. C., Rabins, P. V., Ritchie, K., Rossor, M., Thal, L., & Winblad, B. (2001). Current concepts in mild cognitive impairment. Archives of Neurology, 58, 1985–1992.
Petroni, F., Panzeri, S., Hilgetag, C.-C., KoÈtter, R., & Young, M. P. (2001). Simultaneity of responses in a hierarchical visual network. Neuroreport, 12, 2753–2759.
Power, J. D., Barnes, K. A., Snyder, A. Z., Schlaggar, B. L., & Petersen, S. E. (2012). Spurious but systematic correlations in functional connectivity MRI networks arise from subject motion. NeuroImage, 59, 2142–2154.
Power, J. D., Schlaggar, B. L., & Petersen, S. E. (2015). Recent progress and outstanding issues in motion correction in resting state fMRI. NeuroImage, 105, 536–551.
Prvulovic, D., Hubl, D., Sack, A., Melillo, L., Maurer, K., Frölich, L., Lanfermann, H., Zanella, F., Goebel, R., & Linden, D. E. J. (2002). Functional imaging of visuospatial processing in Alzheimer’s disease. NeuroImage, 17, 1403–1414.
Qi, Z., Wu, X., Wang, Z., Zhang, N., Dong, H., Yao, L., & Li, K. (2010). Impairment and compensation coexist in amnestic MCI default mode network. NeuroImage, 50, 48–55.
Raichle, M. E., MacLeod, A. M., Snyder, A. Z., Powers, W. J., Gusnard, D. A., & Shulman, G. L. (2001a). A default mode of brain function. Proceedings of the National Academy of Sciences, 98, 676–682.
Raichle, M. E., MacLeod, A. M., Snyder, A. Z., Powers, W. J., Gusnard, D. A., & Shulman, G. L. (2001b). A default mode of brain function. Proceedings of the National Academy of Sciences of the United States of America, 98, 676–682.
Raichle, M. E., & Snyder, A. Z. (2007). A default mode of brain function: a brief history of an evolving idea. NeuroImage, 37, 1083–1090 discussion 1097-1089.
Rizzo, M., Anderson, S. W., Dawson, J., & Nawrot, M. (2000). Vision and cognition in Alzheimer’s disease. Neuropsychologia, 38, 1157–1169.
Rombouts, S. A., Scheltens, P., Kuijer, J., & Barkhof, F. (2007). Whole brain analysis of T2* weighted baseline FMRI signal in dementia. Human Brain Mapping, 28, 1313–1317.
Satterthwaite, T. D., Wolf, D. H., Loughead, J., Ruparel, K., Elliott, M. A., Hakonarson, H., Gur, R. C., & Gur, R. E. (2012). Impact of in-scanner head motion on multiple measures of functional connectivity: relevance for studies of neurodevelopment in youth. NeuroImage, 60, 623–632.
Shulman, G. L., Corbetta, M., Buckner, R. L., Fiez, J. A., Miezin, F. M., Raichle, M. E., & Petersen, S. E. (1997a). Common blood flow changes across visual tasks: I. Increases in Subcortical Structures and Cerebellum but not in Nonvisual Cortex. Journal of Cognitive Neuroscience, 9, 624–647.
Shulman, G. L., Fiez, J. A., Corbetta, M., Buckner, R. L., Miezin, F. M., Raichle, M. E., & Petersen, S. E. (1997b). Common blood flow changes across visual tasks: II. Decreases in Cerebral Cortex. Journal of Cognitive Neuroscience, 9, 648–663.
Song, J., Qin, W., Liu, Y., Duan, Y., Liu, J., He, X., Li, K., Zhang, X., Jiang, T., & Yu, C. (2013). Aberrant functional organization within and between resting-state networks in AD. PloS One, 8, e63727.
Song, X. W., Dong, Z. Y., Long, X. Y., Li, S. F., Zuo, X. N., Zhu, C. Z., He, Y., Yan, C. G., & Zang, Y. F. (2011). REST: a toolkit for resting-state functional magnetic resonance imaging data processing. PloS One, 6, e25031.
Todd, J. J., & Marois, R. (2004). Capacity limit of visual short-term memory in human posterior parietal cortex. Nature, 428, 751–754.
Truchot, L., Costes, N., Zimmer, L., Laurent, B., Le Bars, D., Thomas-Anterion, C., Mercier, B., Hermier, M., Vighetto, A., & Krolak-Salmon, P. (2008). A distinct [18 F] MPPF PET profile in amnestic mild cognitive impairment compared to mild Alzheimer’s disease. NeuroImage, 40, 1251–1256.
Van Dijk, K. R., Sabuncu, M. R., & Buckner, R. L. (2012). The influence of head motion on intrinsic functional connectivity MRI. NeuroImage, 59, 431–438.
Wang, Z., Yan, C., Zhao, C., Qi, Z., Zhou, W., Lu, J., He, Y., & Li, K. (2011). Spatial patterns of intrinsic brain activity in mild cognitive impairment and Alzheimer’s disease: a resting-state functional MRI study. Human Brain Mapping, 32, 1720–1740.
Wang, P., Zhou, B., Yao, H., Zhan, Y., Zhang, Z., Cui, Y., Xu, K., Ma, J., Wang, L., & An, N. (2015). Aberrant intra-and inter-network connectivity architectures in Alzheimer’s disease and mild cognitive impairment. Scientific Reports, 5, 14824.
Wolpert, D. M., Ghahramani, Z., & Jordan, M. I. (1995). An internal model for sensorimotor integration. Science, 269, 1880.
Wolpert, D. M., Goodbody, S. J., & Husain, M. (1998). Maintaining internal representations: the role of the human Superior parietal lobe. Nature Neuroscience, 1, 529–533.
Yetkin, F. Z., Rosenberg, R. N., Weiner, M. F., Purdy, P. D., & Cullum, C. M. (2006). FMRI of working memory in patients with mild cognitive impairment and probable Alzheimer’s disease. European Radiology, 16, 193–206.
Zang, Y. F., He, Y., Zhu, C. Z., Cao, Q. J., Sui, M. Q., Liang, M., Tian, L. X., Jiang, T. Z., & Wang, Y. F. (2007). Altered baseline brain activity in children with ADHD revealed by resting-state functional MRI. Brain & Development, 29, 83–91.
Zarei, M., Patenaude, B., Damoiseaux, J., Morgese, C., Smith, S., Matthews, P. M., Barkhof, F., Rombouts, S., Sanz-Arigita, E., & Jenkinson, M. (2010). Combining shape and connectivity analysis: an MRI study of thalamic degeneration in Alzheimer’s disease. NeuroImage, 49, 1–8.
Zhao, X., Liu, Y., Wang, X., Liu, B., Xi, Q., Guo, Q., Jiang, H., Jiang, T., & Wang, P. (2012). Disrupted small-world brain networks in moderate Alzheimer’s disease: a resting-state FMRI study. PloS One, 7, e33540.
Zhao, Z., Lu, J., Jia, X., Chao, W., Han, Y., Jia, J., & Li, K. (2014). Selective changes of resting-state brain oscillations in aMCI: an fMRI study using ALFF. BioMed Research International, 2014, 920902.
Zhong, Y., Huang, L., Cai, S., Yun, Z., Deneen, K. M. V., Ren, A., & Ren, J. (2014). Altered effective connectivity patterns of the default mode network in Alzheimer’s disease: an fMRI study. Neuroscience Letters, 578, 171–175.
Acknowledgments
This study was funded by the National Natural Science Foundation of China under grant NOs.81071221 and 31271063; the Fundamental Science Research Funds for the Central Universities under grant NO. NSIY131409. Also, all of the authors disclose no conflict of interest for the current study.
Data collection and sharing for this project were funded by the ADNI (National Institutes of Health Grant U01 AG024904) and DOD ADNI (Department of Defense award number W81XWH-12-20012). ADNI is funded by the National Institute on Aging, the National Institute of Biomedical Imaging and Bioengineering, and through generous contributions from the following: Alzheimer’s Association; Alzheimer’s Drug Discovery Foundation; BioClinica, Inc.; Biogen Idec Inc.; Bristol-Myers Squibb Company; Eisai Inc.; Elan Pharmaceuticals, Inc.; Eli Lilly and Company; F.Hoffmann-La Roche Ltd. and its affiliated company Genentech, Inc.; GE Healthcare; Innogenetics, N.V.; IXICO Ltd.; Janssen Alzheimer Immunotherapy Research & Development, LLC.; Johnson & Johnson Pharmaceutical Research & Development LLC.; Medpace, Inc.; Merck & Co., Inc.; Meso Scale Diagnostics, LLC.; NeuroRx Research; Novartis Pharmaceuticals Corporation; Pfizer Inc.; Piramal Imaging; Servier; Synarc Inc.; and Takeda Pharmaceutical Company. Private sector contributions were facilitated by the Foundation for the National Institutes of Health (www.fnih.org). The grantee organization is the Northern California Institute for Research and Education, and the study is coordinated by the Alzheimer’s disease Cooperative Study at the University of California, San Diego. ADNI data are disseminated by the Laboratory for Neuro Imaging at the University of Southern California.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflict of interest
Suping Cai, Tao Chong, Jun Li, Yanlin Peng, Wenyue Shen, Karen M. von Deneen, Liyu Huang and Alzheimer’s Disease Neuroimaging Initiative declare that they have no conflict of interest.
Additional information
Suping Cai and Tao Chong contributed equally to this study.
Data used in preparation of this article were obtained from the Alzheimer’s disease Neuroimaging Initiative (ADNI) database (adni.loni.usc.edu). As such, the investigators within ADNI contributed to the design and implementation of ADNI and/or provided data but did not participate in the analysis or writing of this report. A complete listing of ADNI investigators can be found at:
http://adni.loni.usc.edu/wp-content/uploads/how_to_apply/ADNI_Acknowledgement_List.pdf
Rights and permissions
About this article
Cite this article
Cai, S., Chong, T., Peng, Y. et al. Altered functional brain networks in amnestic mild cognitive impairment: a resting-state fMRI study. Brain Imaging and Behavior 11, 619–631 (2017). https://doi.org/10.1007/s11682-016-9539-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11682-016-9539-0