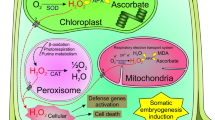
Abstract
We examined how reactive oxygen species, in the form of hydrogen peroxide (H2O2), affect osmotic stress–induced programmed cell death during somatic embryogenesis from cotyledon explants of Manchurian ash (Fraxinus mandshurica Rupr.). We found that substantial osmotic stress was essential for Manchurian ash somatic cells to obtain embryogenic competence. The explant cells displayed hallmarks of programmed cell death, chromatin condensation, and DNA fragmentation to oligonucleotides during somatic embryogenesis. Increasing concentrations of plant growth regulators and sucrose in the medium increased osmotic stress thereby inducing H2O2 accumulation in the explant cells. We found that H2O2 concentration was significantly decreased in explant cells when the induction medium was modified, i.e., when reducing the concentration of sucrose, which reduces the osmotic pressure of the medium, or by withdrawing plant growth regulators at mid-culture. These treatments also decreased the proportion of explant cells undergoing programmed cell death. Accordingly, a decreased rate of somatic embryo induction was observed. These results show that PCD occurred during tissue browning and death of some explant cells during somatic embryogenesis in F. mandshurica. The ROS contributed to PCD in abiotic stress stimulated F. mandshurica cells.




Similar content being viewed by others
References
Apel K, Hirt H (2004) Reactive oxygen species: metabolism, oxidative stress, and signal transduction. Annu Rev Plant Biol 55:373–399
Balestrazzia A, Agonia V, Tavab A, Avatoc P, Biazzib E, Raimondia E, Macoveia A, Carbonera D (2011) Cell death induction and nitric oxide biosynthesis in white poplar (Populus alba) suspension cultures exposed to alfalfa saponins. Physiol Plant 141:227–238
Bozhkov PV, Filonova LH, von Arnold S (2002) A key developmental switch during Norway spruce somatic embryogenesis is induced by withdrawal of growth regulators and is associated with cell death and extracellular acidification. Biotechnol Bioeng 77:658–667
Bozhkov PV, Filonova LH, Suarez MF (2005) Programmed cell death in plant embryogenesis. Curr Top Dev Biol 67:135–179
Businge E, Brackmann K, Moritz T, Egertsdotter U (2012) Metabolite profiling reveals clear metabolic changes during somatic embryo development of Norway spruce (Picea abies). Tree Physiol 32:232–244. https://doi.org/10.1093/treephys/tpr142
Dan YH, Zhang S, Zhong H, Yi H, Sainz MB (2015) Novel compounds that enhance Agrobacterium-mediated plant transformation by mitigating oxidative stress. Plant Cell Rep 34:291–309
de Pinto MC, Tommasi F, Gara LD (2002) Changes in the antioxidant systems as part of the signaling pathway responsible for the programmed cell death activated by nitric oxide and reactive oxygen species in tobacco Bright-Yellow 2 cells. Plant Physiol 130:698–708. https://doi.org/10.1007/s11240-013-0345-8
Fortes AM, Costa J, Santos F, Seguı´-Simarro JM, Palme K, Altabell T, Tiburcio AF, Pais S (2011) Arginine decarboxylase expression, polyamines biosynthesis and reactive oxigen species during organogenic nodule formation in hop. Plant Signal Behav 6:258–269
Gechev TS, Van Breusegem F, Stone JM, Denev I, Laloi C (2006) Reactive oxygen species as signals that modulate plant stress responses and programmed cell death. BioEssays 28:1091–1101
Greenberg JT (1996) Programmed cell death: a way of life for plants. PNAS 93:12094–12097
Groß F, Durner J, Gaupels F (2013) Nitric oxide, antioxidants and prooxidants in plant defence responses. Front Plant Sci 4:419. https://doi.org/10.3389/fpls.2013.00419
Helmersson A, ArnoldSV BurgK, Bozhkov PV (2004) High stability of nuclear microsatellite loci during the early stages of somatic embryogenesis in Norway spruce. Tree Physiol 24:1181–1186
Hill RD, Huang S, Stasolla C (2013) Hemoglobins, programmed cell death and somatic embryogenesis. Plant Sci 211:35–41
Hu LJ, Uchiyama K, Shen HL, Ide Y (2010) Multiple-scaled spatial genetic structures of Fraxinus mandshurica over a riparian–mountain landscape in Northeast China. Conserv Genet 11:77–87. https://doi.org/10.1007/s10592-009-0004-0
Kaewubon P, Hutadilok-Towatana N, Teixeira da Silva JA, Meesawat U (2015) Ultrastructural and biochemical alterations during browning of pigeon orchid (Dendrobium crumenatum Swartz) callus. Plant Cell, Tissue Organ Cult 121:53–69
Kong DM, Preece JE, Shen HL (2012a) Somatic embryogenesis in immature cotyledons of Manchurian ash (Fraxinus mandshurica Rupr.). Plant Cell, Tissue Organ Cult 108:485–492. https://doi.org/10.1007/s11240-011-0062-0
Kong DM, Shen HL, Li N (2012b) Influence of AgNO3 on somatic embryo induction and development in Manchurian ash (Fraxinus mandshurica Rupr). Afr J Biotechnol 11(1):120–125. https://doi.org/10.5897/AJB11.3061
Kwak JM, Nguyen V, Schroeder JI (2006) The role of reactive oxygen species in hormonal responses. Plant Physiol 141:323–329. https://doi.org/10.1104/pp.106.079004
Laloi C, Apel K, Danon A (2004) Reactive oxygen signalling: the latest news. Curr Opin Plant Biol 7:323–328
Lara-Chave A, Flinn BS, Egertsdotter U (2011) Initiation of somatic embryogenesis from immature zygotic embryos of Oocarpa pine (Pinus oocarpa Schiede ex Schlectendal). Tree Physiol 31:539–554. https://doi.org/10.1093/treephys/tpr040
Lee CY, Whitaker RJ (1995) Enzymatic browning and its prevention. American Chemical Society, Washington, p 338
Leonardo J, Andre LWDS, Caroline AB, Heloisa RB, Eny ISF (2014) Proteomic analysis and polyamines, ethylene and reactive oxygen species levels of Araucaria angustifolia (Brazilian pine) embryogenic cultures with different embryogenic potential. Tree Physiol 34:94–104. https://doi.org/10.1093/treephys/tpt102
Liu CP, Yang L, Shen HL (2015) Proteomic analysis of immature Fraxinus mandshurica cotyledon tissues during somatic embryogenesis: effects of explant browning on somatic embryogenesis. Int J Mol Sci 16:13692–13713. https://doi.org/10.3390/ijms160613692
Maraschin SDF, Gaussand G, Pulido A, Olmedilla A, Lamers GEM, Korthout H, Spaink HP, Wang M (2005) Programmed cell death during the transition from multicellular structures to globular embryos in barley androgenesis. Planta 221:459–470
Mittler R (2002) Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci 7:405–410
Mittler R, Vanderauwera S, Gollery M, Van Breusegem F (2004) Reactive oxygen gene network of plants. Trends Plant Sci 9:490–498
Mittler R, Vanderauwera S, Suzuki N, Miller G, Tognetti VB, Vandepoele K, Gollery M, Shulaev V, Van BF (2011) ROS signaling: the new wave? Trends Plant Sci 16:300–309. https://doi.org/10.1016/j.tplants.2011.03.007
Naik SK, Chand PK (2011) Tissue culture-mediated biotechnological intervention in pomegranate: a review. Plant Cell Rep 30:707–721. https://doi.org/10.1007/s00299-010-0969-7
Nowak K, Gaj MD (2016) Stress-related function of bHLH109 in somatic embryo induction in Arabidopsis. J Plant Physiol 193:119–126
Petrov V, Hille J, Mueller-Roeber B, Gechev TS (2015) ROS-mediated abiotic stress-induced programmed cell death in plants. Front Plant Sci 18:69. https://doi.org/10.3389/fpls.2015.00069
Petrussa E, Bertolini A, Casolo V, Krajnáková J, Macrì F, Vianello A (2009) Mitochondrial bioenergetics linked to the manifestation of programmed cell death during somatic embryogenesis of Abies alba. Planta 231:93–107. https://doi.org/10.1007/s00425-009-1028-x
Pirttilä A, Podolich O, Koskimäki JJ, Esa H, Hohtola A (2008) Role of origin and endophyte infection in browning of bud-derived tissue cultures of Scots pine (Pinus sylvestris L.). Plant Cell, Tissue Organ Cult 95:47–55. https://doi.org/10.1007/s11240-008-9413-x
Rani D, Dantu PK (2014) Sustained shoot multiplication and method for overcoming in vitro browning in medicinally important plant, Piper chaba hunt. Proc Natl Acad Sci India Sect B Biol Sci 86:407–413. https://doi.org/10.1007/s40011-014-0461-1
Sun J, Zhang CL, Zhang X, Deng SR, Zhao R, Shen X, Chen SL (2012) Extracellular ATP signaling and homeostasis in plant cells. Plant Signal Behav 7:566–569. https://doi.org/10.4161/psb.19857
Tang QY, Feng GM (2002) Practical statistical analysis and DPS data handling system. Science Publishing, Beijing
van Doorn WG, Beers EP, Dangl JL, Franklin-Tong VE, Gallois P, Hara-Nishimura I, Jones AM, Kawai-Yamada M, Lam E, Mundy J, Mur LA, Petersen M, Smertenko A, Taliansky M, Van Breusegem F, Wolpert T, Woltering E, Zhivotovsky B, Bozhkov PV (2011) Morphological classification of plant cell deaths. Cell Death Differ 18:1241–1246. https://doi.org/10.1038/cdd.2011.36
von Arnold S, Bozhkov P, Clapham D, Dyachok J, Filonova L, Högberg KA, Ingouff M, Wiweger M (2005) Propagation of Norway spruce via somatic embryogenesis. Plant Cell, Tissue Organ Cult 81:323–329
Wang Y, Loake GJ, Chu CC (2013) Cross-talk of nitric oxide and reactive oxygen species in plant programed cell death. Front Plant Sci 4:314. https://doi.org/10.3389/fpls.2013.00314
Wu JH, Zhang XL, Nie YC (2003) Programmed cell death during somatic proliferation and embryogenesis of cotton (Gossypium hirsutum L.). J Plant Physiol Mol Biol 29:515–520
Yang L, Bian L, Shen HL, Li YH (2013) Somatic embryogenesis and plantlet regeneration from mature zygotic embryos of Manchurian ash (Fraxinus mandshurica Rupr.). Plant Cell, Tissue Organ Cult 115:115–125. https://doi.org/10.1007/s11240-013-0345-8
Zimmerman JL (1993) Somatic embryogenesis: a model for early development in higher plants. Plant Cell 5:1411–1423. https://doi.org/10.1105/tpc.5.10.1411
Acknowledgements
We are very grateful for our laboratory colleagues for constructive discussions and technical support.
Author information
Authors and Affiliations
Contributions
Y. L and S. HL conceived and designed the study. Y. L and H. C collected plant materials and prepared SE samples for analysis osmotic stress-induced PCD and intracellular H2O2 concentration. W. C and L. HN analyzed the results for experiments on osmotic stress-induced PCD and intracellular H2O2 concentration. Y. L and Z. DY contributed to the writing of the manuscript and data analyses. L. YH revised the manuscript. All authors read and approved the final manuscript.
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Project funding: The work was supported by the National Key R&D Program of China (2017YFD0600600), the National Natural Science Foundation of China (Nos. 31400535 and 31570596), the National Basic Science Personnel Training Fund of China (No. J1210053), the Innovation Project of State Key Laboratory of Tree Genetics and Breeding (Northeast Forestry University, No. 2016C01) and the Fundamental Research Funds for the Central Universities (Nos. 2572014CA13 and 2572018BW02).
The online version is available at http://www.springerlink.com
Corresponding editor: Tao Xu.
Rights and permissions
About this article
Cite this article
Yang, L., Wei, C., Huang, C. et al. Role of hydrogen peroxide in stress-induced programmed cell death during somatic embryogenesis in Fraxinus mandshurica. J. For. Res. 30, 767–777 (2019). https://doi.org/10.1007/s11676-019-00908-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11676-019-00908-x