Abstract
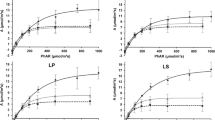
With economic incentives and interests in fast-growing poplar trees for short-rotation production of fiber and veneer, many new poplar hybrids have been bred and planted in China, but how to match the new poplar clones to suitable sites and maintain their higher growth rates is still not very clear. In this study, the photosynthetic response of poplar leaves at various developmental stages during two seasons (summer and autumn) was explored and mechanistic models for the photosynthesis of poplar leaves at different developmental phases in response photosynthetic active radiation (PAR), temperature, and relatively humidity were established using the optimization software package 1st Opt. Mature poplar leaves in autumn had significantly higher photosynthetic capacity than leaves at other stages and seasons. Based on the models established for poplar leaves at different phases, the main limiting factors for photosynthesis at the research site were high PAR and temperature in the summer and low PAR in the autumn. Our results highlight the importance of selecting suitable sites, pruning and stand density control during the plantation development to maintain higher photosynthetic rates of poplar trees and to establish optimum cultivation patterns for various utilization of poplar plantations.






Similar content being viewed by others
References
Baldocchi DD, Harley PC (1995) Scaling carbon dioxide and water vapour exchange from leaf to canopy in a deciduous forest. II. Model testing and application. Plant Cell Environ 18(10):1157–1173. doi:10.1111/j.1365-3040.1995.tb00626.x
Catský J, Šesták Z (1997) Photosynthesis during leaf development. Handbook of photosynthesis. Marcel Dekker, New York, pp 633–660
Constable GA, Rawson HM (1980) Effect of leaf position, expansion and age on photosynthesis, transpiration and water use efficiency of cotton. Funct Plant Biol 7(1):89–100. doi:10.1071/PP9800089
Dale JE (1992) How do leaves grow? Bioscience 42(6):423–432
Deckmyn G, Laureysens I, Garcia J, Muys B, Ceulemans R (2004) Poplar growth and yield in short rotation coppice: model simulations using the process model SECRETS. Biomass Bioenerg 26(3):221–227. doi:10.1016/S0961-9534(03)00121-1
Dillenburg LR, Sullivan JH, Teramura AH (1995) Leaf expansion and development of photosynthetic capacity and pigments in Liquidambar styraciflua (Hamamelidaceae) -effects of UV-B radiation. Am J Bot 82:878–885
Dreyer E, Le Roux X, Montpied P, Daudet FA, Masson F (2001) Temperature response of leaf photosynthetic capacity in seedlings from seven temperate tree species. Tree Physiol 21(4):223–232. doi:10.1093/treephys/21.4.223
Fang S, Xu X, Lu S, Tang L (1999) Growth dynamics and biomass production in short-rotation poplar plantations: 6-year results for three clones at four spacings. Biomass Bioenerg 17(5):415–425. doi:10.1016/S0961-9534(99)00060-4
Fang S, Xie B, Liu D, Liu J (2011) Effects of mulching materials on nitrogen mineralization, nitrogen availability and poplar growth on degraded agricultural soil. New For 41(2):147–162. doi:10.1007/s11056-010-9217-9
Fang S, Zhai X, Wan J, Tang L (2013) Clonal variation in growth, chemistry and calorific value of new poplar hybrids at nursery stage. Biomass Bioenerg 54:303–311. doi:10.1016/j.biombioe.2012.10.005
Flexas J, Gulías J, Jonasson S, Medrano H, Mus M (2001) Seasonal patterns and control of gas exchange in local populations of the Mediterranean evergreen shrub Pistacia lentiscus L. Acta Oecol 22(1):33–43. doi:10.1016/S1146-609X(00)01099-7
Fung LE, Wang SS, Altman A, Hütterman A (1998) Effect of NaCl on growth, photosynthesis, ion and water relations of four poplar genotypes. For Ecol Manag 107(1):135–146. doi:10.1016/S0378-1127(97)00328-9
Greer DH, Halligan EA (2001) Photosynthetic and fluorescence light responses for kiwifruit (Actinidia deliciosa) leaves at different stages of development on vines grown at two different photon flux densities. Funct Plant Biol 28(5):373–382. doi:10.1071/PP00146
Guo XY, Zhang XS (2010) Performance of 14 hybrid poplar clones grown in Beijing, China. Biomass Bioenerg 34(6):906–911. doi:10.1016/j.biombioe.2010.01.036
Hansen EA (1991) Poplar woody biomass yields: a look to the future. Biomass Bioenerg 1(1):1–7. doi:10.1016/0961-9534(91)90046-F
Harley PC, Tenhunen JD (1991) Modeling the photosynthetic response of C3 leaves to environmental factors. In: Boote KJ, Loomis RS (eds) Modeling crop photosynthesis—from biochemistry to canopy. CSSA, Madison, pp 17–39
Jiang CD, Li PM, Gao HY, Zou Q, Jiang GM, Li LH (2005) Enhanced photoprotection at the early stages of leaf expansion in field-grown soybean plants. Plant Sci 168(4):911–919. doi:10.1016/j.plantsci.2004.11.004
Lauenroth WK, Dodd JL, Sims PL (1978) The effects of water-and nitrogen-induced stresses on plant community structure in a semiarid grassland. Oecologia 36(2):211–222. doi:10.1007/BF00349810
Lautner S, Grams T, Matyssek R, Fromm J (2005) Characteristics of electrical signals in poplar and responses in photosynthesis. Plant Physiol 138(4):2200–2209. doi:10.1104/pp.105.064196
Le Roux X, Gran S, Dreyer E, Daudet FA (1999) Parameterization and testing of a biochemically based photosynthesis model for walnut (Juglans regia) trees and seedlings. Tree Physiol 19:481–492
Medlyn BE, Loustau D, Delzon S (2002) Temperature response of parameters of a biochemically based model of photosynthesis. I. Seasonal changes in mature maritime pine (Pinus pinaster Ait.). Plant Cell Environ 25(9):1155–1165. doi:10.1046/j.1365-3040.2002.00890.x
Meir P, Kruijt B, Broadmeadow M, Barbosa E, Kull O, Carswell F, Nobre A, Jarvis PG (2002) Acclimation of photosynthetic capacity to irradiance in tree canopies in relation to leaf nitrogen concentration and leaf mass per unit area. Plant Cell Environ 25:343–357
Niinemets Ü, Tenhunen JD (1997) A model separating leaf structural and physiological effects on carbon gain along light gradients for the shade-tolerant species Acer saccharum. Plant, Cell Environ 20(7):845–866. doi:10.1046/j.1365-3040.1997.d01-133.x
Ogaya R, Peñuel ASJ (2003) Comparative seasonal gas exchange and chlorophyll fluorescence of two dominant woody species in a Holm Oak Forest. Flora 198(2):132–141. doi:10.1078/0367-2530-00085
Pokovai K, Kovacs GJ (2003) Development of crop models: a critical review. Novenytermeles 52:573–582
Powles SB (1984) Photoinhibition of photosynthesis induced by visible light. Annu Rev Plant Physiol 35(1):15–44. doi:10.1146/annurev.pp.35.060184.000311
Pury DD, Farquhar GD (1997) Simple scaling of photosynthesis from leaves to canopies without the errors of big-leaf models. Plant Cell Environ 20(5):537–557. doi:10.1111/j.1365-3040.1997.00094.x
Reddy AR, Chaitanya KV, Vivekanandan M (2004) Drought-induced responses of photosynthesis and antioxidant metabolism in higher plants. J Plant Physiol 161(11):1189–1202. doi:10.1016/j.jplph.2004.01.013
Roden J, Van Volkenburgh E, Hinckley TM (1990) Cellular basis for limitation of poplar leaf growth by water deficit. Tree Physiol 6(2):211–219. doi:10.1093/treephys/6.2.211
Roper TR, Kennedy RA (1986) Photosynthetic characteristics during leaf development in bing sweet cherry. J Am Soc Hortic Sci 111(6):938–941
Wang X, He W, Qu F, Feng X, Li C (2013) Effect of different cultivation patterns of blueberry on soil quality. Acta Agri Jiangxi 25(8):17–21 (in chinese)
Yan Y, Fang S, Tian Y, Deng S, Tang L, Chuong DN (2015) Influence of tree spacing on soil nitrogen mineralization and availability in hybrid poplar plantations. Forests 6(3):636–649. doi:10.3390/f6030636
Author information
Authors and Affiliations
Corresponding author
Additional information
Project funding
This work was financially supported by the National Key Technology R & D Program (2015BAD09B0203) and the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD).
The online version is available at http://www.springerlink.com.
Corresponding editor: Hu Yanbo
Qingqing Wu and Yang Liu contributed equally to this work
Rights and permissions
About this article
Cite this article
Wu, Q., Liu, Y., Fang, S. et al. Photosynthetic response of poplar leaves at different developmental phases to environmental factors. J. For. Res. 28, 909–915 (2017). https://doi.org/10.1007/s11676-016-0358-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11676-016-0358-3