Abstract
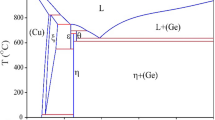
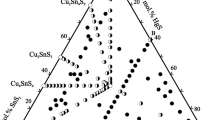
The experimental and thermodynamic descriptions of the Cu-Se-Te ternary system are important in designing alloys for thermoelectric applications. However, information on the Cu-Se-Te ternary system is still lacking in the literature. This study determines the phase equilibria in the isothermal section at 500 °C and liquidus projection of the Cu-Se-Te system by using an experimental and Calphad-approach (Calculation of Phase Diagrams). The isothermal section is determined based on the equilibrated alloys. The liquidus projection is determined based on the results of the primary solidification phase of various Cu-Se-Te alloys in the as-cast condition. Two ternary phases, Cu5Se7Te3 and Cu6Se2Te2, are found. The Calphad-approach is used for thermodynamic modeling of the Cu-Se-Te ternary system by employing thermodynamic descriptions of the three constituent binary systems and the experimental results of the isothermal section and liquidus projection. Very good agreement was obtained between the experiment and calculation.





















Similar content being viewed by others
References
W. Di Liu, L. Yang, Z.G. Chen, and J. Zou, Promising and Eco-Friendly Cu2X-Based Thermoelectric Materials: Progress and Applications, Adv. Mater., 2020, 32, p 87–92. https://doi.org/10.1002/adma.201905703
K. Zhao, H. Duan, N. Raghavendra, P. Qiu, Y. Zeng, W. Zhang, J. Yang, X. Shi, and L. Chen, Solid-State Explosive Reaction for Nanoporous Bulk Thermoelectric Materials, Adv. Mater., 2017, 29, p 1–7. https://doi.org/10.1002/adma.201701148
M. Li, Y. Luo, G. Cai, X. Li, X. Li, Z. Han, X. Lin, D. Sarker, and J. Cui, Realizing High Thermoelectric Performance in Cu2Te Alloyed Cu1.15In2.29Te4, J. Mater. Chem. A, 2019, 7, p 2360–2367. https://doi.org/10.1039/c8ta10741f
R. Schmid-Fetzer, Phase Diagrams: The Beginning of Wisdom, J. Phase Equilib. Diffus., 2014, 35, p 735–760. https://doi.org/10.1007/s11669-014-0343-5
Y.A. Chang, S. Chen, F. Zhang, X. Yan, F. Xie, R. Schmid-Fetzer, and W.A. Oates, Phase Diagram Calculation: Past, Present and Future, Prog. Mater. Sci., 2004, 49, p 313–345. https://doi.org/10.1016/S0079-6425(03)00025-2
Y.A. Chang, Phase Diagram Calculations in Teaching, Research, and Industry, Metall. Mater. Trans. A, 2006, 37, p 273–305.
J.Y. Du, A. Zemanová, Y. Hutabalian, A. Kroupa, and S.W. Chen, Phase Diagram of Ag-Pb-Sn System, Calphad, 2020, 71, p 101997. https://doi.org/10.1016/j.calphad.2020.101997
Y. Hutabalian and S.W. Chen, Interfacial Reactions in Ag/Se, Ag/Se-30at.%Te and Ag2Te/Se Couples and the Phase Equilibria of the Ag-Se-Te Ternary System, J. Alloys Compd., 2021, 889, p 161580. https://doi.org/10.1016/j.jallcom.2021.161580
R. De Medicis and G. Giasson, Le Système Cu-Se-Te, C. R. l’Acad. Des. Sci. Ser. D Sci. Nat., 1971, 272, p 513–515.
V.M. Glazov, A.S. Burkhanov, and N.M. Saleeva, Phase Equilibria in the Quasi-Binary Systems Formed by Cu Chalcogenides, Zhurnal Fiz. Khimii, 1975, 49, p 1658–1661.
I.R. Amiraslanov, K.K. Azizova, and Y.R. Aliyeva, Crystal Structure of Cu4SeTe, Crystallogr. Rep., 2017, 62, p 215–218. https://doi.org/10.1134/S1063774517020043
Z.Y. Aydın, S. Malekghasemi, and S. Abaci, Underpotential Co-Deposition of Ternary Cu-Te-Se Semiconductor Nanofilm on Both Flexible and Rigid Substrates, Appl. Surf. Sci. Surf. Sci., 2019, 470, p 658–667.
P. Villars, H. Okamoto, and A. Prince, Handbook of Ternary Alloy Phase Diagrams. ASM International, 1995.
H.L. Lukas, S.G. Fries, and B. Sundman, Computational Thermodynamics: The Calphad Method. Cambridge University Press, New York, 2007.
D.C. Harris and E.W. Nuffield, Bambollaite, A New Copper Telluro-Selenide, Can. Mineral., 1972, 11, p 738–742.
N.A. Alieva, G.G. Guseinov, V.A. Gasymov, Y.I. Alyev, and T.R. Mekhtiev, Structural Phase Transitions of Polycrystalline Cu4SeTe, Inorg. Mater., 2015, 51, p 661–664. https://doi.org/10.1134/S0020168515070018
K. Zhao, M. Guan, P. Qiu, A.B. Blichfeld, E. Eikeland, C. Zhu, D. Ren, F. Xu, B.B. Iversen, X. Shi, and L. Chen, Thermoelectric Properties of Cu2Se1−XTex Solid Solutions, J. Mater. Chem. A, 2018, 6, p 6977–6986. https://doi.org/10.1039/c8ta01313f
F.N. Rhines, Phase Diagrams in Metallurgy, Their Development and Application. McGraw-Hill, New York, 1956.
S.W. Chen, Y. Hutabalian, W. Gierlotka, C.H. Wang, and S.T. Lu, Phase Diagram of Bi-In-Se Ternary System, Calphad, 2020, 68, p 101744. https://doi.org/10.1016/j.calphad.2020.101744
W. Gierlotka and W.-H. Wu, The Reoptimization of the Binary Se-Te System, Int. J. Mater. Res., 2012, 103, p 698–701. https://doi.org/10.3139/146.110677
Z. Du, C. Guo, M. Tao, and C. Li, Thermodynamic Modeling of the Cu-Se System, Int. J. Mater. Res., 2008, 99, p 294–300.
K.C. Hari Kumar and P. Wollants, Some Guidelines for Thermodynamic Optimisation of Phase Diagrams, J. Alloys Compd., 2001, 320, p 189–198. https://doi.org/10.1016/S0925-8388(00)01491-2
W. Cao, S. Chen, F. Zhang, K. Wu, Y. Yang, Y.A. Chang, R. Schmid-fetzer, and W.A. Oates, CALPHAD: Computer Coupling of Phase Diagrams and Thermochemistry PANDAT Software with PanEngine, PanOptimizer and PanPrecipitation for Multi-Component Phase Diagram Calculation and Materials Property Simulation, Calphad, 2009, 33, p 328–342. https://doi.org/10.1016/j.calphad.2008.08.004
D. Huang, R. Han, Y. Wang, and T. Ye, The Cu-Te System: Phase Relations Determination and Thermodynamic Assessment, J. Alloys Compd., 2021, 855, p 157373. https://doi.org/10.1016/j.jallcom.2020.157373
P.R. Subramanian, Cu-Te (copper-tellerium), 2nd edn. ASM International, Ohio, 1990.
A. Dinsdale, SGTE Pure Element Database (UNARY). Version 5.0, (2009). www.sgte.net.
M. Hillert, The Compound Energy Formalism, J. Alloys Compd., 2001, 320, p 161–176.
R. Schmid and Y.A. Chang, A Thermodynamic Study on an Associated Solution Model for Liquid Alloys, Calphad, 1985, 9, p 363–382. https://doi.org/10.1016/0364-5916(85)90004-5
D.J. Chakrabarti and D.E. Laughlin, The Cu-Se (Copper-Selenium) System, Bull. Alloy Phase Diagr., 1981, 2, p 305–315. https://doi.org/10.1007/BF02868284
V.M. Glazov, A.S. Pashinkin, and V.A. Fedorov, Phase Equilibria in the Cu-Se System, Inorg. Mater., 2000, 36, p 641–652. https://doi.org/10.1007/BF02758413
A.S. Pashinkin and V.A. Fedorov, Phase Equilibria in the Cu-Te System, Inorg. Mater., 2003, 39, p 647–663.
A.S. Pashinkin and L.M. Pavlova, Standard Functions of Formation and Thermodynamic Stability of Compounds in the Cu-Te System, Inorg. Mater., 2005, 41, p 1050–1054. https://doi.org/10.1007/s10789-005-0259-x
Y. Hutabalian and S.W. Chen, Phase Relationships in the Cu-Se-Te Ternary System, Preparation for Submission, 2023.
Acknowledgments
The authors acknowledge the financial support of the Ministry of Science and Technology, Taiwan (MOST 107–2923-E-007–005-MY3) and (MOST 110–2634-F-007–024).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hutabalian, Y., Chen, Sw. & Gierlotka, W. Phase Equilibria of the Cu-Se-Te Ternary System. J. Phase Equilib. Diffus. 44, 181–199 (2023). https://doi.org/10.1007/s11669-023-01032-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11669-023-01032-4