Abstract
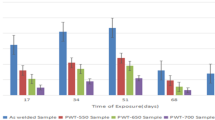
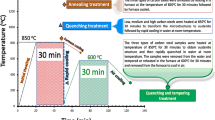
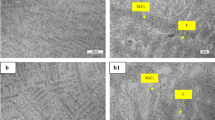
The work investigated the effects of pre-surface treatment on wear and corrosion rates of copper electrodeposition on medium-carbon steel. Medium-carbon steel samples were subjected to heat treatment (annealing, normalizing and quenching) in a muffle furnace. The samples were prepared and subjected to nickel striking and copper electroplating using standard bath composition. Electroplated samples were taken for weight test, optical microscopy, wear test and corrosion test. Result showed that the heat-treated copper-electrodeposited substrate had homogenous distribution of fine copper deposition, while the tempered heat-treated substrate was observed with the lowest wear rate of 3.5 × 10−3 g/mm3/min and lowest corrosion rate of 0.01833 mmpy in seawater environment of 0.5 M NaCl, thus having a low resistance to wear and corrosion in saline environment. This research has established that un-electroplated medium-carbon steel is highly corrosive in saline environment due to its higher corrosion penetration rates, while the tempered heat-treated copper-electrodeposited steels are lowly corrosion resistant and therefore suitable for use as a good material of choice in saline environments.
























Similar content being viewed by others
Data Availability
No data were used to support this study.
References
I. Momoh, O. Olateju, D. Oloruntoba, Investigation on the corrosion performance of nickel electrodeposited tempered steel substrate. J. Sci. Technol. 1, 400–404 (2012)
O.O. Ajibola, B.O. Adewuyi, D.T. Oloruntoba, Wear behaviour of sand cast eutectic Al-Si alloy in hydraulic brake fluid. Int. J. Innov. Appl. Stud. 6, 420–430 (2014)
O.O. Ajibola, D.T. Oloruntoba, Effects of polishing grades and Saccharin-550 additive on copper electroplating on NST60Mn and NST50-2 steels. Int. J. Innov. Sci. Res. 8(2), 334–344 (2014)
O.O. Ajibola, D.T. Oloruntoba, B.O. Adewuyi, Effects of hard surface grinding and activation on electroless-nickel plating on cast aluminium alloy substrates. Hindawi Publ. Corp. J. Coat. 2014, 1–10 (2014). https://doi.org/10.1155/2014/841619
P.-Y. Chen, Y.-T. Chang, Voltammetric study and electrodeposition of copper in 1-butyl-3-methylimidazolium salicylate ionic liquid. Electrochim. Acta 75, 339–346 (2012)
O. Fayomi, ZnO as corrosion inhibitor for dissolution of zinc electrodeposited mild steel in varying HCl concentration. Int. J. Phys. Sci. 6, 2447–2454 (2011)
M. Pasquale, L. Gassa, A. Arvia, Copper electrodeposition from an acidic plating bath containing accelerating and inhibiting organic additives. Electrochim. Acta 53, 5891–5904 (2008)
C.V. Pecequilo, Z. Panossian, Study of copper electrodeposition mechanism from a strike alkaline bath prepared with 1-hydroxyethane-1, 1-diphosphonic acid through cyclic voltammetry technique. Electrochim. Acta 55, 3870–3875 (2010)
R. Schrebler Arratia, H. Aros Meneses, R. Schrebler Guzman, C. Carlesi Jara, Use of polyethylene glycol as organic additive in copper electrodeposition over stainless steel cathodes. Latin Am. Appl. Res. 42, 371–376 (2012)
T.I. Török, V. Orosz, Z. Fekete, G. Szirmai, Direct cathodic deposition of copper on steel wires from pyrophosphate baths. Mater. Sci. Eng. 37, 99–110 (2012)
O. Oluwole, O. Olawale, Corrosion behaviour of nickel plated low carbon steel in tomato fluid. Leonardo Electron. J. Pract. Technol. 9, 33–42 (2010)
O.O. Ajibola, D.T. Oloruntoba, B.O. Adewuyi, Investigation of corrosion of cast aluminium alloy piston in brake fluid. Afr. Corros. J. 2 (2015)
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest regarding the publication of this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Oloruntoba, D.T., Adesina, O.S., Falana, O. et al. Effect of Preheat Treatment on Wear and Corrosion Rates of Copper Electrodeposition on Medium-Carbon Steel. J Fail. Anal. and Preven. 20, 1754–1764 (2020). https://doi.org/10.1007/s11668-020-00982-y
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11668-020-00982-y