Abstract
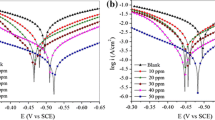
Corrosion inhibition performance of synthesized indoline compounds, namely, 3′-(4-(1-acetyl-5-(4-methoxyphenyl)-4,5-dihydro-1H-pyrazol-3-yl)phenyl)spiro[indoline-3,2′-thiazolidine]-2,4′-dione (MPIT) and 1-(4-(1-acetyl-5-(4-methoxyphenyl)-4,5-dihydro-1H-pyrazol-3-yl)phenyl)spiro[imidazolidine-2,3′-indoline]-2′,5-dione (MPII) on N80 steel in 15% HCl solution was studied using weight loss measurement, potentiodynamic polarization, and electrochemical impedance spectroscopy (EIS) techniques. Polarization studies showed that both studied inhibitors were of mixed type in nature. The adsorption of inhibitors on the N80 steel surface obeys Langmuir adsorption isotherm. The potential of zero charge (E PZC) for the N80 steel was determined by EIS method. Scanning electron microscopy, energy-dispersive x-ray spectroscopy, and atomic force microscopy were used to characterize the surface morphology of uninhibited and inhibited N80 steel specimens. The density functional theory was employed for theoretical calculations.












Similar content being viewed by others
References
M. Yadav, D. Behera, and U. Sharma, Corrosion Protection of N80 Steel in Hydrochloric Acid by Substituted Amino Acids, Corros. Eng. Sci. Technol., 2013, 48, p 19–27
M. Yadav, D. Behera, and U. Sharma, Nontoxic Corrosion Inhibitors for N80 Steel in Hydrochloric Acid, Arab. J. Chem., 2012, doi:10.1016/j.arabjc.2012.03.011.
M. Yadav and U. Sharma, Eco-Friendly Corrosion Inhibitors for N80 Steel in Hydrochloric Acid, J. Mater. Environ. Sci., 2011, 2, p 407–414
M. Yadav, S. Kumar, and D. Sharma, Experimental and Quantum Chemical Studies on Corrosion Inhibition Effect of Synthesized Organic Compounds on N80 Steel in Hydrochloric Acid, Ind. Eng. Chem. Res., 2013, 52, p 14019–14029
S. Vishwanatham and P.K. Sinha, Corrosion Protection of N80 Steel in HCl by Condensation Products of Aniline and Phenol, Anti-Corros. Methods Mater., 2009, 56, p 139–144
T. Kumar, S. Vishawanatam, and G. Udaybhanu, Synergistic Effect of Formaldehyde and Alcoholic Extract of Plants Leaves for Protection of N80 Steel in 15% HCl, Corros. Eng. Sci. Technol., 2004, 39, p 327–332
R. Solmaz, G. Kardaş, M. Çulha, and M. Erbil, Investigation of Adsorption and Inhibitive Effect of 2-Mercaptothiazoline on Corrosion of Mild Steel in Hydrochloric Acid Media, Electrochim. Acta, 2008, 53, p 5941–5952
D. Bhambi, C. Sharma, V.K. Salvi, and G.L. Talesara, Synthesis and Pharmacological Studies of Some Phthalimidoxy Substituted Spiro- Thiazolidinone Derivatives of Isatin, Indian J. Chem. B, 2009, 48, p 1006–1012
I.B. Obot and N.O. Obi-Egbedi, Indeno-1-one [2,3-b]quinoxaline as an effective inhibitor for the corrosion of mild steel in 0.5 M H2SO4 solution, Mater. Chem. Phys., 2010, 122, p 325–328
C. Lee, W. Yang, and R.G. Parr, Development of the Colle-Salvetti Correlation-Energy Formula into a Functional of the Electron Density, Phys. Rev. B, 1988, 37, p 785–789
A.D. Becke, A New Mixing of Hartree-Fock and Local Density-Functional Theories, J. Chem. Phys., 1993, 98, p 1372–1377
X. Wang, H. Yang, and F. Wang, An Investigation of Benzimidazole Derivative as Corrosion Inhibitor for Mild Steel in Concentrated HCl Solutions, Corros. Sci., 2011, 53, p 113–121
D. Jayaperumal, Effect of Alcohol Based Inhibitors on Corrosion of Mild Steel in Hydrochloric Medium, Mater. Chem. Phys., 2010, 119, p 478–484
E.S. Ferreira, C. Giancomlli, F.C. Giacomlli, and A. Spinelli, Evaluation of the Inhibitor Effect of l-Ascorbic Acid on the Corrosion of Mild Steel, Mater. Chem. Phys., 2004, 83, p 129–134
M. Lebrini, F. Robert, A. Lecante, and C. Roos, Corrosion Inhibition of C38 Steel in 1 M Hydrochloric Acid Medium by Alkaloids Extract from Oxandra asbeckii plant, Corros. Sci., 2007, 49, p 2254–2269
H. Gerengi and H.I. Sahin, Schinopsis lorentzii Extract as a Green Corrosion Inhibitor for Low Carbon Steel in 1 M HCl Solution, Ind. Eng. Chem. Res., 2012, 51, p 780–787
B. El Mehdi, B. Mernari, M. Traisnel, F. Bentiss, and M. Lagrenee, Synthesis and Comparative Study of the Inhibitive Effect of Some New Triazole Derivatives Towards Corrosion of Mild Steel in Hydrochloric Acid Solution, Mater. Chem. Phys., 2002, 77, p 489–496
M. Behpour, S.M. Ghoreishi, N. Soltani, M. Salavati-Niasari, M. Hamadanian, and A. Gandomi, Electrochemical and Theoretical Investigation on the Corrosion Inhibition of Mild Steel by Thiosalicylaldehyde Derivatives in Hydrochloric Acid Solution, Corros. Sci., 2008, 50, p 2172–2181
R.G. Pearson, Absolute Electronegativity and Hardness: Application to Inorganic Chemistry, Inorg. Chem., 1988, 27, p 734–740
A.Y. Musa, A.H. Kadhum, A.B. Mohamad, and M.S. Takriff, Experimental and Theoretical Study on the Inhibition Performance of Triazole Compounds for Mild Steel Corrosion, Corros. Sci., 2010, 52, p 3331–3340
P. Mourya, S. Banerjee, R.B. Rastogi, and M.M. Singh, Inhibition of Mild Steel Corrosion in Hydrochloric and Sulfuric Acid Media Using a Thiosemicarbazone Derivative, Ind. Eng. Chem. Res., 2013, 52, p 12733–12747
Acknowledgment
Authors acknowledge Indian School of Mines, Dhanbad for financial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yadav, M., Sarkar, T.K. & Purkait, T. Studies on Adsorption and Corrosion Inhibitive Properties of Indoline Compounds on N80 Steel in Hydrochloric Acid. J. of Materi Eng and Perform 24, 4975–4984 (2015). https://doi.org/10.1007/s11665-015-1765-x
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11665-015-1765-x