Abstract
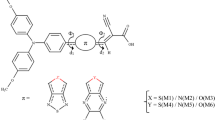
We investigated metal-free dyes based on isoindigo by performing density functional theory and Time-dependent density functional theory calculations to improve the efficiency of dye-sensitized solar cells. The D–π′–A′–π–A organic dyes (TIDP and TIDT) used triphenylamine as donor, thiophene as the π′-linker between the donor and auxiliary acceptor, and a phenyl or thiophene ring as the π-linker between the auxiliary acceptor and acceptor. TIDP and TIDT exhibit good charge-transfer properties. The TIDP-based device provides better device performance with a PCE of 4.11%. Calculated results reveal that the phenyl ring directly linking the auxiliary acceptor and acceptor causes a small tilt angle in the TiO2–adsorped dye, resulting in enhanced electron-injection rates, more efficient packing of adsorbed dye molecules, and slow charge recombination at the TiO2 surface. The performance of the TIDT-based device (η = 2.46%), arises from decreased electron-injection rates and fast charge recombination caused by the large dihedral angle of the adsorbed dye. This research identifies a potential π′-linker group and reveals the influence of the π-linker on photovoltaic performance in organic dyes.
Graphic Abstract
The phenyl ring directly linking the auxiliary acceptor and acceptor causes a smaller tilt angle in the TiO2-absorbed dye compared to thiophene ring resulting in enhanced electron-injection rates with increasing short-circuit current density (Jsc) as well as slow charge recombination at the TiO2 surface with increasing open-circuit voltage (Voc).

Similar content being viewed by others
References
A. Mishra, M.K.R. Fischer, and P. Bäuerle, Angew. Chem. Int. Ed. 48, 2474 (2009).
J. Gong, J. Liang, and K. Sumathy, Renew. Sustain. Energy Rev. 16, 5848 (2012).
F. Gao, Y. Wang, D. Shi, J. Zhang, M. Wang, X. Jing, R. Humphry-Baker, P. Wang, S.M. Zakeeruddin, and M. Grätzel, J. Am. Chem. Soc. 130, 10720 (2008).
M.K. Nazeeruddin, F. De Angelis, S. Fantacci, A. Selloni, G. Viscardi, P. Liska, S. Ito, B. Takeru, and M. Grätzel, J. Am. Chem. Soc. 127, 16835 (2005).
C.-Y. Chen, M. Wang, J.-Y. Li, N. Pootrakulchote, L. Alibabaei, C.-H. Ngoc-le, J.-D. Decoppet, J.-H. Tsai, C. Grätzel, C.-G. Wu, S.M. Zakeeruddin, and M. Grätzel, ACS Nano 3, 3103 (2009).
J.R. Jennings, Y. Liu, and Q. Wang, J. Phys. Chem. C 115, 15109 (2011).
A. Yella, H.-W. Lee, H.N. Tsao, C. Yi, A.K. Chandiran, M.K. Nazeeruddin, E.W.-G. Diau, C.-Y. Yeh, S.M. Zakeeruddin, and M. Grätzel, Science 334, 629 (2011).
C.-P. Lee, C.-T. Li, and K.-C. Ho, Mater. Today 20, 267 (2017).
S.-Q. Fan, C. Kim, B. Fang, K.-X. Liao, G.-J. Yang, C.-J. Li, J.-J. Kim, and J. Ko, J. Phys. Chem. C 115, 7747 (2011).
Z. Yao, M. Zhang, H. Wu, L. Yang, R. Li, and P. Wang, J. Am. Chem. Soc. 137, 3799 (2015).
Z. Yao, H. Wu, Y. Li, J. Wang, J. Zhang, M. Zhang, Y. Guo, and P. Wang, Energy Environ. Sci. 8, 3192 (2015).
D.D. Babu, S.R. Gachumale, S. Anandan, and A.V. Adhikari, Dyes Pigm. 112, 183 (2015).
J. Chen, Q. Liu, H. Li, Z. Zhao, Z. Lu, Y. Huang, and D. Xu, Front. Chem. 6, 200 (2018).
Y. Wu and W. Zhu, Chem. Soc. Rev. 42, 2039 (2013).
P. Naik, R. Su, M.R. Elmorsy, A. El-Shafei, and A.V. Adhikari, Photochem. Photobiol. Sci. 17, 302 (2018).
G.-G. Luo, H. Lu, Y.-H. Wang, J. Dong, Y. Zhao, and R.-B. Wu, Dyes Pigm. 134, 498 (2016).
S. Zeng, L. Yin, C. Ji, X. Jiang, K. Li, Y. Li, and Y. Wang, Chem. Commun. 48, 10627 (2012).
K. Narayanaswamy, A. Venkateswararao, V. Gupta, S. Chand, and S.P. Singh, Chem. Commun. 52, 210 (2016).
Y. Wu, W.-H. Zhu, S.M. Zakeeruddin, and M. Grätzel, ACS Appl. Mater. Interfaces 7, 9307 (2015).
A. Hagfeldt, G. Boschloo, L. Sun, L. Kloo, and H. Pettersson, Chem. Rev. 110, 6595 (2010).
W. Zhu, Y. Wu, S. Wang, W. Li, X. Li, J. Chen, Z.-S. Wang, and H. Tian, Adv. Funct. Mater. 21, 756 (2011).
Y. Wang, C. Yang, J. Chen, H. Qi, J. Hua, Y. Liu, E. Baranoff, H. Tan, J. Fan, and W. Zhu, Dyes Pigm. 127, 204 (2016).
J.L. Westrup, L.W. Oenning, M.M. da Silva Paula, R. da Costa Duarte, F.S. Rodembusch, T.E.A. Frizon, L. da Silva, and A.G. Dal-Bó, Dyes Pigm. 126, 209 (2016).
Z. Wang, L. Zhu, Z. Shuai, and Z. Wei, Macromol. Rapid Commun. 38, 1700470 (2017).
H. Zhu, W. Li, Y. Wu, B. Liu, S. Zhu, X. Li, H. Ågren, and W. Zhu, ACS Sustain. Chem. Eng. 2, 1026 (2014).
H.C. Ting, Y.H. Chen, L.Y. Lin, S.H. Chou, Y.H. Liu, H.W. Lin, and K.T. Wong, ChemSusChem 7, 457 (2014).
Y. Jiang, Y. Gao, H. Tian, J. Ding, D. Yan, Y. Geng, and F. Wang, Macromolecules 49, 2135 (2016).
H.T. Zaky, Y.G. Saleh, and N.A. Ahmed, J. Enzyme Inhib. Med. Chem. 28, 853 (2013).
B. Liu, Y. Zou, B. Peng, B. Zhao, K. Huang, Y. He, and C. Pan, Polym. Chem. 2, 1156 (2011).
E. Wang, Z. Ma, Z. Zhang, P. Henriksson, O. Inganas, F. Zhang, and M.R. Andersson, Chem. Commun. 47, 4908 (2011).
E. Wang, Z. Ma, Z. Zhang, K. Vandewal, P. Henriksson, O. Inganäs, F. Zhang, and M.R. Andersson, J. Am. Chem. Soc. 133, 14244 (2011).
J. Mei, K.R. Graham, R. Stalder, and J.R. Reynolds, Org. Lett. 12, 660 (2010).
G. Zhang, Y. Fu, Z. Xie, and Q. Zhang, Macromolecules 44, 1414 (2011).
W. Ying, F. Guo, J. Li, Q. Zhang, W. Wu, H. Tian, and J. Hua, ACS Appl. Mater. Interfaces 4, 4215 (2012).
D. Wang, W. Ying, X. Zhang, Y. Hu, W. Wu, and J. Hua, Dyes Pigm. 112, 327 (2015).
M. Pastore and F. De Angelis, ACS Nano 4, 556 (2010).
B. Peng, S. Yang, L. Li, F. Cheng, and J. Chen, J. Chem. Phys. 132, 034305 (2010).
M.P. Sumsalee, Synthesis and Characterization of Isoindigo Derivatives for Optoelectronic Applications. Suranaree University of Science and Technology (2015).
A.D. Becke, J. Chem. Phys. 98, 5648 (1993).
Y. Zhao and D.G. Truhlar, Acc. Chem. Res. 41, 157 (2008).
A.D. Becke, J. Chem. Phys. 98, 1372 (1993).
T. Yanai, D.P. Tew, and N.C. Handy, Chem. Phys. Lett. 393, 51 (2004).
N.M. O’boyle, A.L. Tenderholt, and K.M. Langner, J. Comput. Chem. 29, 839 (2008).
D.-M. Gu, J.-Z. Zhang, M. Zhang, Y. Geng, and Z.-M. Su, Dyes Pigm. 132, 136 (2016).
G.W.S. Frisch, G.E. Robb, M.A. Cheeseman, J.R. Scalmani, G. Barone, V. Mennucci, B. Petersson, G.A. Nakatsuji, H. Caricato, M. Li, X. Hratchian, H.P. Izmaylov, A.F. Bloino, J. Zheng, G. Sonnenberg, J.L. Hada, M. Ehara, M. Toyota, K. Fukuda, R. Hasegawa, J. Ishida, M. Nakajima, T. Honda, Y. Kitao, O. Nakai, H. Vreven, T. Montgomery, J.A. Peralta Jr, J.E. Ogliaro, F. Bearpark, M. Heyd, J.J. Brothers, E. Kudin, K.N. Staroverov, V.N. Kobayashi, R. Normand, J. Raghavachari, K. Rendell, A. Burant, J.C. Iyengar, S.S. Tomasi, J. Cossi, M. Rega, N. Millam, J.M. Klene, M. Knox, J.E. Cross, J.B. Bakken, V. Adamo, C. Jaramillo, J. Gomperts, R. Stratmann, R.E. Yazyev, O. Austin, A.J. Cammi, R. Pomelli, C. Ochterski, J.W. Martin, R.L. Morokuma, K. Zakrzewski, V.G. Voth, G.A. Salvador, P. Dannenberg, J.J. Dapprich, S. Daniels, A.D. Farkas, O. Foresman, J.B. Ortiz, J.V. Cioslowski, and J. Fox, Gaussian 09 Revision A.02 (Wallingford, CT: Gaussian Inc, 2009).
F. De Angelis, G. Vitillaro, L. Kavan, M.K. Nazeeruddin, and M. Grätzel, J. Phys. Chem. C 116, 18124 (2012).
P. Persson, R. Bergström, and S. Lunell, J. Phys. Chem. B 104, 10348 (2000).
R. Tarsang, V. Promarak, T. Sudyoadsuk, S. Namuangruk, N. Kungwan, and S. Jungsuttiwong, ChemPhysChem 15, 3809 (2014).
S. Jungsuttiwong, R. Tarsang, T. Sudyoadsuk, V. Promarak, P. Khongpracha, and S. Namuangruk, Org. Electron. 14, 711 (2013).
S. Jungsuttiwong, T. Yakhanthip, Y. Surakhot, J. Khunchalee, T. Sudyoadsuk, V. Promarak, N. Kungwan, and S. Namuangruk, J. Comput. Chem. 33, 1517 (2012).
T. Yakhanthip, S. Jungsuttiwong, S. Namuangruk, N. Kungwan, V. Promarak, T. Sudyoadsuk, and P. Kochpradist, J. Comput. Chem. 32, 1568 (2011).
S. Namuangruk, R. Fukuda, M. Ehara, J. Meeprasert, T. Khanasa, S. Morada, T. Kaewin, S. Jungsuttiwong, T. Sudyoadsuk, and V. Promarak, J. Phys. Chem. C 116, 25653 (2012).
R. Tarsang, V. Promarak, T. Sudyoadsuk, S. Namuangruk, and S. Jungsuttiwong, J. Photochem. Photobiol. A 273, 8 (2014).
R. Tarsang, V. Promarak, T. Sudyoadsuk, S. Namuangruk, N. Kungwan, P. Khongpracha, and S. Jungsuttiwong, RSC Adv. 5, 38130 (2015).
E. Ronca, M. Pastore, L. Belpassi, F. Tarantelli, and F. De Angelis, Energy Environ. Sci. 6, 183 (2013).
Y.-X. Weng, Y.-Q. Wang, J.B. Asbury, H.N. Ghosh, and T. Lian, J. Phys. Chem. B 104, 93 (2000).
F. De Angelis, A. Tilocca, and A. Selloni, J. Am. Chem. Soc. 126, 15024 (2004).
M. Khoudiakov, A.R. Parise, and B.S. Brunschwig, J. Am. Chem. Soc. 125, 4637 (2003).
F. De Angelis, S. Fantacci, E. Mosconi, M.K. Nazeeruddin, and M. Grätzel, J. Phys. Chem. C 115, 8825 (2011).
B. Delley and D.E. Ellis, J. Chem. Phys. 76, 1949 (1982).
T. Lu and F. Chen, J. Comput. Chem. 33, 580 (2012).
C.L. Wang, J. Wang, F.Q. Bai, J. Chen, and H.X. Zhang, Int. J. Quantum Chem. 114, 560 (2014).
R. Zhang, W. Liu, R. Hu, Y. Ma, Y. Sun, J. Zhang, Y. Pu, J. Yang, L. Chu, and X.A. Li, Sol. Energy Mater. Sol. Cells 191, 183 (2019).
X. Mu, J. Wang, and M. Sun, J. Phys. Chem. C 123, 14132 (2019).
H. Zong, J. Wang, X. Mu, X. Xu, J. Li, X. Wang, F. Long, J. Wang, and M. Sun, Phys. Chem. Chem. Phys. 20, 13558 (2018).
R. Li, X. Wang, Y. Zhou, H. Zong, M. Chen, and M. Sun, J. Nanophotonics 12, 1 (2018).
H. Zong, X. Mu, J. Wang, H. Zhao, Y. Shi, and M. Sun, Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 209, 228 (2019).
B. Xu, Y. Li, P. Song, F. Ma, and M. Sun, Sci. Rep. 7, 45688 (2017).
H. Zong, X. Wang, J. Quan, C. Tian, and M. Sun, Phys. Chem. Chem. Phys. 20, 19720 (2018).
X. Shi, Y. Yang, L. Wang, and Y. Li, J. Phys. Chem. C 123, 4007 (2019).
Y. Li, B. Xu, P. Song, F. Ma, and M. Sun, J. Phys. Chem. C 121, 12546 (2017).
X. Wang, J. Xu, M. Li, D. Fang, B. Chen, L. Wang, and W. Xu, RSC Adv. 3, 5227 (2013).
L.L. Estrella, M.P. Balanay, and D.H. Kim, J. Phys. Chem. A 120, 5917 (2016).
L.L. Estrella, S.H. Lee, and D.H. Kim, Dyes Pigm. 165, 1 (2019).
P. Ren, C. Sun, Y. Shi, P. Song, Y. Yang, and Y. Li, J. Mater. Chem. C 7, 1934 (2019).
H.-H.G. Tsai, J.-C. Hu, C.-J. Tan, Y.-C. Sheng, and C.-C. Chiu, J. Phys. Chem. A 120, 8813 (2016).
B.C. O’Regan, K. Walley, M. Juozapavicius, A. Anderson, F. Matar, T. Ghaddar, S.M. Zakeeruddin, C. Klein, and J.R. Durrant, J. Am. Chem. Soc. 131, 3541 (2009).
A.N.M. Green, R.E. Chandler, S.A. Haque, J. Nelson, and J.R. Durrant, J. Phys. Chem. B 109, 142 (2005).
C.E. Richards, A.Y. Anderson, S. Martiniani, C. Law, and B.C. O’Regan, J. Phys. Chem. Lett. 3, 1980 (2012).
M. Tuikka, P. Hirva, K. Rissanen, J. Korppi-Tommola, and M. Haukka, Chem. Commun. 47, 4499 (2011).
J.N. Clifford, E. Palomares, M.K. Nazeeruddin, M. Grätzel, and J.R. Durrant, J. Phys. Chem. C 111, 6561 (2007).
J. Nyhlen, G. Boschloo, A. Hagfeldt, L. Kloo, and T. Privalov, ChemPhysChem 11, 1858 (2010).
M. Xie, J. Wang, F.-Q. Bai, L. Hao, and H.-X. Zhang, Dyes Pigm. 120, 74 (2015).
M. Xie, F.-Q. Bai, J. Wang, C.-P. Kong, J. Chen, and H.-X. Zhang, Comput. Mater. Sci. 111, 239 (2016).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hadsadee, S., Promarak, V., Sudyoadsuk, T. et al. Theoretical Study on Factors Influencing the Efficiency of D–π′–A′–π–A Isoindigo-Based Sensitizer for Dye-Sensitized Solar Cells. J. Electron. Mater. 49, 318–332 (2020). https://doi.org/10.1007/s11664-019-07777-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11664-019-07777-4