Abstract
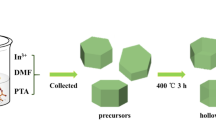
Uniform-size In2O3 hollow spheres were synthesized using carbon microspheres as sacrificial template and In(NO3)3 as the indium resource. The effects of different solvents on the structure and morphology of as-prepared samples were discussed, and it was found that the shell thickness of In2O3 hollow microspheres could be controlled by changing solvents. The formation mechanism of the In2O3 hollow microspheres was also investigated. The gas-sensing results indicated that all of the as-prepared samples showed high gas-sensing response and excellent selectivity to formaldehyde and In2O3 hollow microspheres had an advantage over the bulk In2O3. Notably, In2O3 hollow microspheres with the shell thickness of 20 nm were fabricated by methanol solution and showed a higher response value of 41 toward 100 ppm HCHO than those of In2O3 hollow microspheres obtained by using other solvents. In2O3 hollow microspheres had good selectivity, high response and fast response-recovery, which made this product a promising gas-sensing material for detecting formaldehyde.
Similar content being viewed by others
References
W. Qin, L. Xu, J. Song, R. Xing, and H. Song, Sens. Actuators B Chem. 185, 231 (2013).
L. Wang, Z. Lou, T. Fei, and T. Zhang, J. Mater. Chem. 22, 4767 (2012).
X. Xu, H. Zhang, X. Hu, P. Sun, Y. Zhu, C. He, S. Hou, Y. Sun, and G. Lu, Sens. Actuators B Chem. 227, 547 (2015).
P. Wang, Y. Fu, B. Yu, Y. Zhao, L. Xing, and X. Xue, J. Mater. Chem. A 3, 3529 (2015).
R.A. Kadir, Z. Li, A.Z. Sadek, R.A. Rani, A.S. Zoolfakar, M.R. Field, J.Z. Ou, A.F. Chrimes, and K. Kalantar-zadeh, J. Phy. Chem. C 118, 3129 (2014).
K. Nam, H.G. Kim, H. Choi, H. Park, J.S. Kang, Y.E. Sung, H.C. Lee, and H. Choe, J. Electron. Mater. 46, 3748 (2017).
C.T. Quy, C.M. Hung, N.V. Duy, N.D. Hoa, M. Jiao, and H. Nguyen, J. Electron. Mater. 46, 3406 (2017).
M. Sinha, R. Mahapatra, B. Mondal, and R. Ghosh, J. Electron. Mater. 46, 2476 (2017).
Z. Wang, Z. Li, T. Jiang, X. Xu, and C. Wang, ACS Appl. Mater Interfaces 5, 2013 (2013).
L. Liu, S. Li, J. Zhuang, L. Wang, J. Zhang, H. Li, Z. Liu, Y. Han, X. Jiang, and P. Zhang, Sens. Actuators B Chem. 155, 782 (2011).
M. Xu, Q. Li, Y. Ma, and H. Fan, Sens. Actuators B Chem. 199, 403 (2014).
G. Xi, Y. Yan, Q. Ma, J. Li, H. Yang, X. Lu, and C. Wang, Chem. Eur. J. 18, 13949 (2012).
X.F. Wu, H.Y. Song, J.M. Yoon, Y.T. Yu, and Y.F. Chen, Langmuir 25, 6438 (2009).
J. Zai, J. Zhu, R. Qi, and X. Qian, J. Mater. Chem. A 1, 735 (2013).
X. Li, S. Yao, J. Liu, P. Sun, Y. Sun, Y. Gao, and G. Lu, Sens. Actuator B Chem. 220, 68 (2015).
D. Han, P. Song, H. Zhang, H. Yan, Q. Xu, Z. Yang, and Q. Wang, RSC Adv. 4, 50241 (2014).
X. Liang, T.H. Kim, J.W. Yoon, C.H. Kwak, and J.H. Lee, Sens. Actuators B Chem. 209, 934 (2015).
S. Kim, S. Park, G.J. Sun, S.K. Hyun, K.K. Kim, and C. Lee, Curr. Appl. Phys. 15, 947 (2015).
K.L. Sang, S.H. Hwang, D. Chang, and S. Kim, Sens. Actuators B Chem. 149, 28 (2010).
L. Gao, Z. Cheng, Q. Xiang, Y. Zhang, and J. Xu, Sens. Actuators B Chem. 208, 436 (2015).
Q. Tang, W. Zhou, W. Zhang, S. Ou, K. Jiang, W. Yu, and Y. Qian, Cryst. Growth Des. 5, 147 (2005).
J. Cao, H. Dou, H. Zhang, H. Mei, S. Liu, T. Fei, R. Wang, L. Wang, and T. Zhang, Sens. Actuators B Chem. 198, 180 (2014).
X. Lai, D. Wang, N. Han, J. Du, J. Li, C. Xing, Y. Chen, and X. Li, Chem. Mater. 22, 3033 (2010).
B. Li, Y. Xie, M. Jing, G. Rong, Y. Tang, and G. Zhang, Langmuir 22, 9380 (2006).
W.J. Tseng, T.T. Tseng, H.M. Wu, Y.C. Her, and T.J. Yang, J. Am. Ceram. Soc. 96, 719 (2013).
L.Y. Chen and Z.D. Zhang, J. Phys. Chem. C 112, 18798 (2008).
X. Lai, J. Li, B.A. Korgel, Z. Dong, Z. Li, F. Su, J. Du, and D. Wang, Angew. Chem. Int. Ed. 50, 2738 (2011).
J. Wang, N. Yang, H. Tang, Z. Dong, Q. Jin, M. Yang, D. Kisailus, H. Zhao, Z. Tang, and D. Wang, Angew. Chem. 52, 6417 (2013).
J.S. Lee and S.C. Choi, J. Eur. Ceram. Soc. 25, 3307 (2005).
X. Li, X. Zhou, H. Guo, C. Wang, J. Liu, P. Sun, F. Liu, G. Lu, and A.C.S. Appl, Mater. Interfaces 6, 18661 (2014).
J. Zhang, X. Liu, S. Wu, M. Xu, X. Guo, and S. Wang, J. Mater. Chem. 20, 6453 (2010).
L. Wang, J. Cao, X. Qian, and H. Zhang, Mater. Lett. 171, 30 (2016).
X. Shen, L. Guo, G. Zhu, C. Xi, Z. Ji, and H. Zhou, RSC Adv. 5, 64228 (2015).
F. Huang, W. Yang, F. He, and S. Liu, Sens. Actuators B Chem. 235, 86 (2016).
L. Xiao, S. Shu, and S. Liu, Sens. Actuators B Chem. 221, 120 (2015).
W. Zheng, X. Lu, W. Wang, Z. Li, H. Zhang, Y. Wang, Z. Wang, and C. Wang, Sens. Actuators B Chem. 142, 61 (2009).
Acknowledgements
This work is supported by the National Natural Science Foundation of China (51272165, 51672138 and 5157277), the Research Foundation of Key Laboratory of Material Corrosion and Protection of Sichuan Province (2014CL13), and the Research Foundation of Sichuan University of Science and Engineering (2014PY11, 2015PY01 and 2012RC03), the Special Funds for the Development of Strategic Emerging Industries in Shenzhen City (JCYJ20150525092941022).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zou, Y., Wang, H., Lai, X. et al. Synthesis and Enhanced Formaldehyde-Sensing Properties of In2O3 Hollow Spheres with Thin Shells. J. Electron. Mater. 47, 2165–2170 (2018). https://doi.org/10.1007/s11664-017-6027-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11664-017-6027-2