Abstract
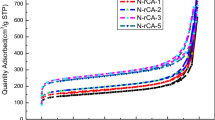
A lithium-based catalyst for carbon aerogel compounds and carbon nanotubes synthesis was used. Lithium hydroxide-catalyzed and CNT-modified carbon aerogel was compared to traditionally synthesized sodium carbonate-catalyzed carbon aerogel, as well as to the same material modified with CNT to evaluate the real effect of lithium hydroxide addition. Enhancement in the specific surface area from 498 m2/g to 786 m2/g and significant change in pore size distribution were observed. Low temperature, supercritical drying in carbon dioxide was used to prepare an organic aerogel with subsequent pyrolysis in an inert gas flow to convert it into carbon aerogel. The as-obtained material was examined with respect to energy storage applications, i.e. symmetric hybrid supercapacitors. It was shown that lithium hydroxide was responsible for shorter gelation time, increased specific surface area, and a greater number of micropores within the structure. For both reference materials prepared using sodium carbonate, quite different data were recorded. It was presented that the proper choice of carbon matrix should combine both high specific surface area and appropriate pore size distribution. High surface area and a relatively large number of micropores were responsible for specific capacity loss.
Article PDF
Similar content being viewed by others
References
S.S. Kistler, Nature 127, 741 (1931).
R.W. Pekala, J. Mater. Sci. 24, 3221 (1989).
M. Ciszewski, E. Szatkowska, A. Koszorek, and M. Majka, J. Mater. Sci. Mater. Electron. 28, 4897 (2017).
M.A. Aegerter, N. Leventis, and M.M. Koebel, Advances in Sol–Gel Derived Materials and Technologies, Aerogels Handbook (New York: Springer, 2011).
N. Job, A. Théry, R. Pirard, J. Marien, L. Kocon, J.N. Rouzaud, F. Béguin, and J.P. Pirard, Carbon 43, 2481 (1995).
W.C. Li, A.H. Lu, and F. Schüth, Chem. Mater. 17, 3620 (2005).
M.F. Yan, L.H. Zhang, R. He, and Z.F. Liu, J. Porous Mater. 22, 699 (2015).
M.F. Grenier-Loustalot, S. Larroque, D. Grande, P. Grenier, and D. Bedel, Polymer 37, 1363 (1996).
C. Moreno-Castilla, F.J. Maldonado-Hódar, J. Rivera-Utrilla, and E. Rodríguez-Castellón, Appl. Catal. A Gen. 183, 345 (1999).
F.J. Maldonado-Hódar, A.F. Pérez-Cadenas, and C. Moreno-Castilla, Carbon 41, 1291 (2003).
C.I. Merzbacher, S.R. Meier, J.R. Pierce, and M.L. Korwin, J. Non. Cryst. Solids 285, 210 (2001).
R. Brandt, R. Petricevic, H. Pröbstle, and J. Fricke, J. Porous Mater. 10, 171 (2003).
K.S. Rejitha, P.A. Abraham, N.P.R. Panicker, K.S. Jacob, and N.C. Pramanik, Adv. Nanoparticles 2, 99 (2013).
S.W. Pattinson, K. Prehn, I.A. Kinloch, D. Eder, K.K.K. Koziol, K. Schulte, and A.H. Windle, RSC Adv. 2, 2909 (2012).
Acknowledgements
The authors would particularly like to thank Prof. Ginter Nawrat from the Department of Inorganic, Analytical Chemistry and Electrochemistry, Silesian University of Technology, Poland for giving the opportunity to perform electrochemical tests. The authors are also grateful for the opportunity to synthesize CNTs in the Electric Carbon Nanomaterials Group headed by Dr. Krzysztof Koziol at the Department of Materials Science and Metallurgy, Cambridge University, UK.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Ciszewski, M., Szatkowska, E. & Koszorek, A. Lithium-Catalyzed Carbon Aerogel and Its Possible Application in Energy Storage Materials. J. Electron. Mater. 46, 4612–4617 (2017). https://doi.org/10.1007/s11664-017-5454-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11664-017-5454-4