Abstract
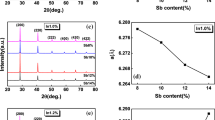
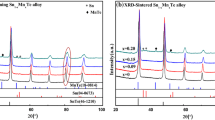
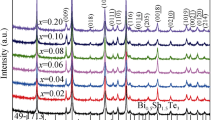
A series of (Sn0.8Pb0.2)1−x Mn x Te alloys with x = 0, 0.03, 0.06, 0.09, 0.12 and 0.15 were prepared by melting, quenching and spark plasma sintering (SPS) techniques to investigate their phases and thermoelectric properties. Mn was used as doped element in Sn0.8Pb0.2Te solid solution to reduce the carrier concentration, enhance the Seebeck coefficient and reduce the thermal conductivity of the material. Experimental results show that the SnTe-based solid solution single phase was formed in the alloys with x = 0 and 0.03. The minor irregular-shaped MnTe2 phase presents in the alloys with x ≥ 0.06, while the minor needle-like MnTe phase appears in the alloys with x ≥ 0.12, together with the SnTe-based solid solution matrix. The lattice parameter a of SnTe-based solid solution decreases nearly linearly as Mn content x increases up to 0.12, but keeps constant as x further increases. All the samples show p-type conduction. Mn doping in Sn0.8Pb0.2Te decreases its carrier concentration and thus increases its Seebeck coefficient. The solute Mn and Pb atoms in the SnTe-based solid solution, and the minor phases MnTe2 and MnTe, enhance the phonon scattering and thus reduce the thermal conductivity. As a result, the figure-of-merit ZT of the (Sn0.8Pb0.2)1−x Mn x Te composites can be enhanced with proper Mn substitution. The maximum ZT of 0.65 was obtained in the sample (Sn0.8Pb0.2)0.88Mn0.12Te at 723 K, which is higher than the 0.29 of its parent alloy Sn0.8Pb0.2Te.
Similar content being viewed by others
References
K. Biswas, J.Q. He, Q.C. Zhang, G.Y. Wang, C. Uher, V.P. Dravid, and M.G. Kanatzidis, Nat. Chem. 3, 160 (2011).
J.Q. Li, X.Y. Liu, Y. Li, S.H. Song, F.S. Liu, and W.Q. Ao, J. Alloy. Compd. 600, 8 (2014).
M. Fujikane, K. Kurosaki, H. Muta, and S. Yamanaka, J. Alloy. Compd. 396, 280 (2005).
E.M. Levin, B.A. Cook, J.L. Harringa, S.L. Bud’ko, R. Venkatasubramanian, and K. Schmidt-Rohr, Adv. Funct. Mater. 21, 441 (2011).
K. Ahn, K. Biswas, J.Q. He, I. Chung, V. Dravid, and M.G. Kanatzidis, Energy Environ. Sci. 6, 1529 (2013).
J.Q. Li, S.P. Li, Q.B. Wang, L. Wang, F.S. Liu, and W.Q. Ao, J. Alloy. Compd. 509, 4516 (2011).
S.P. Li, J.Q. Li, Q.B. Wang, L. Wang, F.S. Liu, and W.Q. Ao, Solid State Sci. 13, 399 (2011).
F. Herman, R.L. Kortum, I.B. Ortenburger, and J.P. Van Dyke, J. Phys. Colloques 29, 62 (1968).
R.F. Brebrick, J. Phys. Chem. Solids 24, 27 (1963).
J.A. Kafalas, R.F. Brebrick, and A.J. Strauss, Appl. Phys. Lett. 4, 93 (1964).
Z.W. Lu, J.Q. Li, C.Y. Wang, Y. Li, F.S. Liu, and W.Q. Ao, J. Alloy. Compd. 621, 345 (2015).
G. Tan, L.D. Zhao, F.Y. Shi, J.W. Doak, S.H. Lo, H. Sun, C. Wolverton, V.P. Dravid, C. Uher, and M.G. Kanatzidis, J. Am. Chem. Soc. 136, 7006 (2014).
M.K. Han, J. Androulakis, S.J. Kim, and M.G. Kanatzidis, Adv. Energy. Mater. 2, 157 (2012).
Y. Chen, M.D. Nielsen, Y.B. Gao, T.J. Zhu, X.B. Zhao, and J.P. Heremans, Adv. Energy. Mater. 2, 58 (2012).
M.K. Han, X.Y. Zhou, C. Uher, S.J. Kim, and M.G. Kanatzidis, Adv. Energy. Mater. 2, 1218 (2012).
K. Guergouri and R. Triboulet, J. Cryst. Growth 132, 20 (1993).
M. Sondermann, J. Magn. Magn. Mater. 2, 216 (1976).
B. Kim, I. Kim, B.K. Min, M. Oh, S. Park, and H. Lee, Electron. Mater. Lett. 9, 477 (2013).
H. Zhang, J. Luo, H.T. Zhu, Q.L. Liu, J.K. Liang, J.B. Li, and G.Y. Liu, Chinese Phys. B 21, 106101 (2012).
J. He, X.J. Tan, J.T. Xu, G.Q. Liu, H.Z. Shao, Y.J. Fu, X. Wang, Z. Liu, J.Q. Xu, H.C. Jiang, and J. Jiang, J. Mater. Chem. A 3, 19974 (2015).
H.J. Wu, C. Chang, D. Feng, Y. Xiao, X. Zhang, Y.L. Pei, L. Zheng, D. Wu, S.K. Gong, Y. Chen, J.Q. He, M.G. Kanatzidis, and L.D. Zhao, Energy Environ. Sci. 8, 3298 (2015).
G.J. Tan, F.Y. Shi, S.Q. Hao, H. Chi, T.P. Bailey, L.D. Zhao, C. Uher, C. Wolverton, V.P. Dravid, and M.G. Kanatzidis, J. Am. Chem. Soc. 137, 11507 (2015).
L.D. Didkin, V.S. Gaidukova, and L.M. Ostrovskaya, Inor. Mater. (USSR) 7, 1503 (1971).
G.J. Snyder and E.S. Toberer, Nat. Mater. 7, 105 (2008).
A. Bali, R. Chetty, and R.C. Mallik, Mater. Sci. Semicon. Proc. 34, 326 (2015).
Acknowledgements
The work was supported by the National Natural Science Foundation of China (Nos. 51571144 and 11504239) and Shenzhen Science and Technology Research Grant (Nos. JCYJ20140418182819176, JCYJ20140418181958489 and JCYJ201503241417 11684).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, J.Q., Lu, Z.W., Wang, C.Y. et al. Enhanced Thermoelectric Properties of Sn0.8Pb0.2Te Alloy by Mn Substitution. J. Electron. Mater. 45, 2879–2885 (2016). https://doi.org/10.1007/s11664-016-4352-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11664-016-4352-5