Abstract
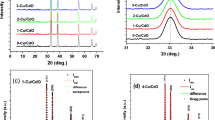
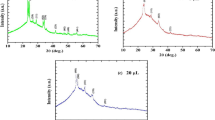
Zinc sulphide (ZnS) thin films with both n- and p-type electrical conductivity were grown on glass/fluorine-doped tin oxide-conducting substrates from acidic and aqueous solution containing ZnSO4 and (NH4)2S2O3 by simply changing the deposition potential in a two-electrode cell configuration. After deposition, the films were characterised using various analytical techniques. X-ray diffraction analysis reveals that the materials are amorphous even after heat treatment. Optical properties (transmittance, absorbance and optical bandgap) of the films were studied. The bandgaps of the films were found to be in the range (3.68–3.86) eV depending on the growth voltage. Photoelectrochemical cell measurements show both n- and p-type electrical conductivity for the films depending on the growth voltage. Scanning electron microscopy shows material clusters on the surface with no significant change after heat treatment at different temperatures. Atomic force microscopy shows that the surface roughness of these materials remain fairly constant reducing only from 18 nm to 17 nm after heat treatment. Thickness estimation of the films was also carried out using theoretical and experimental methods. Direct current conductivity measurements on both as-deposited and annealed films show that resistivity increased after heat treatment.
Similar content being viewed by others
References
T. Yasuda, K. Hara, and H. Kukimoto, J. Cryst. Growth 77, 485 (1986).
S. Tec-Yam, J. Rojas, V. Rejon, and A.I. Oliva, Mater. Chem. Phys. 136, 386 (2012).
O.K. Echendu, F. Fauzi, A.R. Weerasinghe, and I.M. Dharmadasa, Thin Solid Films 556, 529 (2014).
O.K. Echendu and I.M. Dharmadasa, Energies 8, 4416 (2015). doi:10.3390/en8054416.
O.K. Echendu and I.M. Dharmadasa, J. Electron. Mater. 43, 791 (2014).
O.K. Echendu, A.R. Weerasinghe, D.G. Diso, F. Fauzi, and I.M. Dharmadasa, J. Electron. Mater. 42, 692 (2013).
N.K. Abbas, K.T. Al-Rasoul, and Z.J. Shanan, Int. J. Electrochem. Sci. 8, 3049 (2013).
J. Han, G. Fu, V. Krishnakumar, C. Liao, W. Jaegermann, and M.P. Besland, J. Phys. Chem. Solids 74, 1879 (2013).
A. Pudov, J. Sites, and T. Nakada, http://www2.physics.colo state.edu/groups/photovoltaic/PDFs/CSU-AGU%20paper. PDF. Accessed 04/08/2015.
Z. Limei, X. Yuzhi, and L. Jianfeng, J. Environ. Sci. Suppl. 21, S76–S79 (2009).
A. Ates, M.A. Yildirim, M. Kundakci, and A. Astam, Mater. Sci. Semicond. Process. 10, 281 (2007).
C. Falcony, M. Garcia, A. Ortiz, and J.C. Alonso, J. Appl. Phys. 72, 1525 (1992).
A.B. Bhalerao, C.D. Lokhande, and B.G. Wagh, IEEE Trans. Nano Technol. 12, 996 (2013).
D. Peng, Z. Xi-Qing, S. Xue-Bai, Y. Zhi-Gang, and W. Yong-Sheng, Chin. Phys. 15, 1370 (2006).
I.M. Dharmadasa, A.P. Samantilleke, J. Young, and M.H. Boyle, J. Mater. Sci. Mater. Electron. 10, 441 (1999).
I.M. Dharmadasa, P.A. Bingham, O.K. Echendu, H.I. Salim, T. Druffel, R. Dharmadasa, G.U. Sumanasekera, R.R. Dharmadasa, M.B. Dergacheva, K.A. Mit, K.A. Urazov, L. Bowen, M. Walls, and A. Abbas, Coatings 4, 380 (2014).
D. Kurbatov, V. Kosyak, M. Kolesnyk, A. Opanasyuk, and S. Danilchenko, Integr. Ferroelectr. 103, 32 (2008).
J. Diaz-Reyes, R. Castillo-Ojeda, J. Martinez-Juarez, O. Zaca-Moran, J.E. Flores-Mena, and M. Galvan-Arellano, Int. J. Circuits, Syst. Signal Process. 8, 15–21 (2014).
S. Radhu and C. Vijayan, Mater. Chem. Phys. 129, 1132 (2011).
J. Tauc, R. Grigorovini, and A. Vancu, Phys. Status Solidi. 15, 627 (1966).
K.L. Chopra, P.D. Paulson, and V. Dutta, Prog. Photovolt. Res. Appl. 12, 69 (2004).
R.K. Pandey, S.B. Sahu, and S. Chandra, Handbook of Semiconductor Electrodeposition (New York: Allen H. Hermann, Marcel Dekker Inc., 1996).
O.K. Echendu (Doctoral Thesis, 106, Sheffield Hallam University, Sheffield, United Kingdom).
J.D. Joseph and R.C. Neville, J. Appl. Phys. 48, 1941 (1977).
A.U. Ubale and D.K. Kulkarni, Bull. Mater. Sci. 28, 43 (2005).
H.K. Sadekar, N.G. Deshpande, Y.G. Gudage, A. Ghosh, S.D. Chavhan, S.R. Gosavi, and R. Sharma, J. Alloys Compd. 453, 519 (2008).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Madugu, M.L., Olusola, O. O., Echendu, O. . et al. Intrinsic Doping in Electrodeposited ZnS Thin Films for Application in Large-Area Optoelectronic Devices. J. Electron. Mater. 45, 2710–2717 (2016). https://doi.org/10.1007/s11664-015-4310-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11664-015-4310-7