Abstract
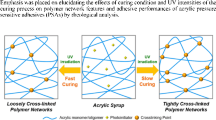
Ultraviolet (UV)-curable urethane acrylic oligomers were synthesized and then modified by the incorporation of silicone diols with different molecular weights to improve thermal stability for temporary bonding and debonding adhesives in a three-dimensional multi-chip package process. The UV-curing behaviors were investigated using photo-differential scanning calorimetry, Fourier transform infrared spectroscopy-attenuated total reflectance, and gel fraction, while the UV-curing kinetics was also studied. In addition, the thermal stability of the samples was checked using thermogravimetric analysis. UV-curing and thermal stability were more affected by the molecular weight of the silicone diols than by UV dose due to both the flexibility and the steric hindrance of the synthesized oligomer structures.
Similar content being viewed by others
References
W.-H. Chen, C.-F. Yu, H.-C. Cheng, Y.-M. Tsai, and S.-T. Lu, Microelectron. Reliab. 53, 30 (2013).
C.-T. Peng, C.-M. Liu, J.-C. Lin, H.-C. Cheng, and K.-N. Chiang, IEEE Trans. Compon. Packag. Technol. 27, 684 (2004).
H.-C. Cheng, Y.-H. Tsai, K.-N. Chen, and J. Fang, Appl. Therm. Eng. 30, 2632 (2010).
H.-C. Cheng, K.-Y. Hsieh, and K.-M. Chen, Microelectron. Reliab. 51, 826 (2011).
K. Tu, Microelectron. Reliab. 51, 517 (2011).
E. Cheng and Y.-L. Shen, Microelectron. Reliab. 52, 534 (2012).
C.-T. Ko and K.-N. Chen, Microelectron. Reliab. 52, 302 (2012).
K. Takahashi, M. Umemoto, N. Tanaka, K. Tanida, Y. Nemoto, Y. Tomita, M. Tago, and M. Bonkohara, Microelectron. Reliab. 43, 1267 (2003).
L.-C. Shen, C.-W. Chien, H.-C. Cheng, and C.-T. Lin, Microelectron. Reliab. 50, 489 (2010).
C.-J. Zhan, C.-C. Chuang, J.-Y. Juang, S.-T. Lu, and T.-C. Chang, in IEEE Electronic Components and Technology Conference (ECTC), 1043 (2010).
F. Niklaus, G. Stemme, J.Q. Lu, and R.J. Gutmann, J. Appl. Phys. 99, 031101 (2006).
B.G. Yacobi, S. Martin, K. Davis, A. Hudson, and M. Hubert, J. Appl. Phys. 91, 6227 (2002).
B. Bilenberg, T. Niesen, B. Clausen, and A. Kristensen, J. Micromech. Microeng. 14, 814 (2004).
R. Puligadda, S. Pillalamarri, W. Hong, C. Brubaker, M. Wimplinger, and S. Pargfrieder, Dig. Mater. Res. Soc. Symp. Proc. 970 (2006). doi:10.1557/PROC-0970-Y04-09.
J. Moore, A. Smith, D. Nguyen, and S. Kulkarni, MANTECH 810 (2004).
C. Brubaker, M. Wimplinger, A. Malzer, and P. Lindner, MANTECH 261 (2005).
S. Combe, J. Cullen, and M. O’Keefe, MANTECH 193 (2006).
W. Noll, Chemistry and Technology of Silicones (New York: Academic Press, 1968), p. 388.
M.G. Voronokov, V.P. Milenshkevich, and Y.A. Yuzhelevskii, The Siloxane Bond (New York: Consultants Bureau, 1978).
İ. Yilgör and J.E. McGrath, Polysiloxane Copolymers/Anionic Polymerization (Germany: Springer, 1988), p. 1.
T.L. Smith, Rubber Chem. Technol. 51, 225 (1978).
K. Matsukawa, K. Hasegawa, H. Inoue, A. Fukuda, and Y. Arita, J. Polym. Sci. Part A 30, 2045 (1992).
W.C. Shih and C.C.M. Ma, J. Appl. Polym. Sci. 69, 51 (1998).
A.T. Holohan, M.H. George, J.A. Barrie, and D.G. Parker, Polymer 35, 977 (1994).
M. Ma, R.M. Hill, J.L. Lowery, S.V. Fridrikh, and G.C. Rutledge, Langmuir 21, 5549 (2005).
S.D. Smith, J.M. DeSimone, H. Huang, G. York, D.W. Dwight, G.L. Wilkes, and J.E. McGrath, Macromolecules 25, 2575 (1992).
H. Zhuang and J.A. Gardella, Macromolecules 30, 3632 (1997).
P. Majumdar and D.C. Webster, Macromolecules 38, 5857 (2005).
Q. Dou, C. Wang, C. Cheng, W. Han, P.C. Thüne, and W. Ming, Macromol. Chem. Phys. 207, 2170 (2006).
J.P. Fouassier and J.F. Rabek, Radiation Curing in Polymer Science and Technology (London: Elsevier Science Publishers, 1993).
P. Xiao, Y. Wang, M. Dai, G. Wu, S. Shi, and J. Nie, Polym. Adv. Technol. 19, 409 (2008).
G. Bayramoğlu, M.V. Kahraman, N. Kayaman-Apohan, and A. Güngör, Prog. Org. Coat. 57, 50 (2006).
N. Kayaman-Apohan, R. Demirci, M. Cakir, and A. Gungor, Radiat. Phys. Chem. 73, 254 (2005).
D.K. Chattopadhyay, S.S. Panda, and K.V.S.N. Raju, Prog. Org. Coat. 54, 10 (2005).
S. Tasic, B. Bozic, and B. Dunjic, Prog. Org. Coat. 51, 320 (2004).
H.-D. Hwang and H.-J. Kim, React. Funct. Polym. 71, 655 (2011).
A. Palanisamy and B. Rao, Prog. Org. Coat. 60, 161 (2007).
Q. Yu, S. Nauman, J. Santerre, and S. Zhu, J. Appl. Polym. Sci. 82, 1107 (2001).
K.S. Anseth, C.N. Bowman, and N.A. Peppas, J. Polym. Sci. Part A 32, 139 (1994).
X. Jiang, H. Xu, and J. Yin, Polymer 45, 133 (2004).
C.Y. Bai, X.Y. Zhang, J.B. Dai, and W.H. Li, Prog. Org. Coat. 55, 291 (2006).
M. Sangermano, N. Lak, G. Malucelli, A. Samakande, and R. Sanderson, Prog. Org. Coat. 61, 89 (2008).
K.D. Belfield and J.V. Crivello, Photoinitiated Polymerization (Washington DC: American Chemical Society Publication, 2003).
C.G. Roffey, Photogeneration of Reactive Species for UV Curing (Weinheim: Wiley Chichester, 1997).
A. Asif and W. Shi, Eur. Polym. J. 39, 933 (2003).
S.-W. Lee, J.-W. Park, C.-H. Park, D.-H. Lim, H.-J. Kim, J.Y. Song, and J.H. Lee, Int. J. Adhes. Adhes. 44, 138 (2013).
A. Elzubair, J.C.M. Suarez, C.M.C. Bonelli, and E.B. Mano, Polym. Test. 22, 647 (2003).
I. Banik and A.K. Bhowmick, Radiat. Phys. Chem. 54, 135 (1999).
D. Chattopadhyay and D.C. Webster, Prog. Polym. Sci. 34, 1068 (2009).
A. Asif, W. Shi, X. Shen, and K. Nie, Polymer 46, 11066 (2005).
J. Yang, Z. Wang, Z. Zeng, and Y. Chen, J. Appl. Polym. Sci. 84, 1818 (2002).
A. Asif and W. Shi, Polym. Adv. Technol. 15, 669 (2004).
C.-Y. Bai, X.-Y. Zhang, J.-B. Dai, and W.-H. Li, Chin. Chem. Lett. 17, 369 (2006).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, SW., Lee, TH., Park, JW. et al. Synthesis and UV-Curing Behaviors of Urethane Acrylic Oligomers Modified by the Incorporation of Silicone Diols into the Soft Segments for a 3D Multi-Chip Package Process. J. Electron. Mater. 44, 2406–2413 (2015). https://doi.org/10.1007/s11664-015-3718-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11664-015-3718-4