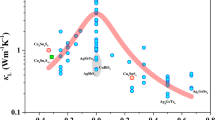
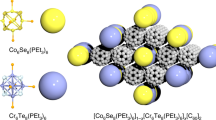
Coinage-metal(I) polychalcogenide halides represent a new class of materials featuring high ion dynamics of different substructures capable of an effective phonon scattering process in the solid state. The interplay of mobile coinage-metal ions such as Ag+ or Cu+ on the one hand and the temperature-driven redox reaction of a linear, partially covalent-bonded Te chain on the other hand is responsible for tremendous variations in the electronic structure of such materials. A huge drop of the Seebeck coefficient within a very small temperature range and an extremely low thermal conductivity are the key properties of Ag10Te4Br3, the first representative of this substance class. Two different sets of compounds with the general formula (CM)10Q4X3 and (CM)23Q12X (CM = coinage metal Cu or Ag, Q = chalcogen, X = halogen) have been found, and recent experiments point toward the existence of formerly unknown copper(I) (poly)chalcogenide halides and two more silver(I) (poly)chalcogenide halides in this field. A comparable linear Te chain is present in a number of different compounds such as some alkaline-earth polychalcogenides M5Te3, the mineral stuetzite Ag4.5Te3, and the closely related compounds Ag11AsTe7 and Ag12Te6S. In most cases the thermoelectric potential of these materials has not been verified. A topological approach for the description of the structural features has been developed to understand the electronic properties of these complex materials in detail.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
B.C. Sales, Int. J. Ceram. Technol. 4, 291 (2007). doi:10.1111/j.1744-7402.2007.02143.x.
C.B. Vining, Nat. Mater. 8, 83 (2009). doi:10.1038/nmat2361.
K.F. Hsu, S. Loo, F. Guo, W. Chen, J.S. Dyck, C. Uher, T. Hogan, E.K. Polychroniadis, and M.G. Kanatzidis, Science 303, 818 (2004). doi:10.1126/science.1092963.
T.C. Harman, P.J. Taylor, D.L. Spears, and M.P. Walsh, J. Electron. Mater. 29, L1 (2000). doi:10.1007/s11664-000-0117-1.
T.C. Harman, M.P. Walsh, B.E. Laforge, and G.W. Turner, J. Electron. Mater. 34, L19 (2005). doi:10.1007/s11664-005-0083-8.
R. Venkatasubramanian, E. Siivola, T. Colpitts, and B. O’Quinn, Nature 413, 597 (2001). doi:10.1038/35098012.
X-RED 32, version 1.10; Stoe & Cie GmbH: Darmstadt, Germany (2004) and X-SHAPE, version 2.05; Stoe & Cie GmbH: Darmstadt, Germany (2004).
V. Petříček, M. Dušek, and L. Palatinus, The Crystallographic Computing System JANA2006 (Praha, Czech Republic: Institute of Physics, 2006).
W.F. Kuhs, Acta Crystallogr. Sect. A 48, 80 (1992). doi:10.1107/S0108767391009510.
R. Blachnik and G. Kudermann, Z. Naturforsch. 28b, 1 (1973).
R. Blachnik and H.A. Dreisbach, J. Solid State Chem. 60, 115 (1985). doi:10.1016/0022-4596(85)90171-9.
T. Doert, E. Rönsch, F. Schnieders, P. Böttcher, and J. Sieler, Z. Anorg. Allg. Chem. 626, 89 (2000).
T. Nilges, S. Nilges, A. Pfitzner, T. Doert, and P. Böttcher, Chem. Mater. 16, 806 (2004). doi:10.1021/cm031131c.
M. Bawohl and T. Nilges, Z. Naturforsch. 63b, 1083 (2008).
J. Messel and T. Nilges, Z. Naturforsch. 63b, 1077 (2008).
T. Nilges, J. Messel, M. Bawohl, and S. Lange, Chem. Mater. 20, 4080 (2008). doi:10.1021/cm800425u.
T. Nilges and J. Messel, Z. Anorg. Allg. Chem. 634, 2185 (2008). doi:10.1002/zaac.200800128.
S. Lange and T. Nilges, Chem. Mater. 18, 2538 (2006). doi:10.1021/cm060226m.
S. Lange, M. Bawohl, D. Wilmer, H.-W. Meyer, H.-D. Wiemhöfer, and T. Nilges, Chem. Mater. 19, 1401 (2007). doi:10.1021/cm062731p.
T. Nilges, M. Bawohl, and S. Lange, Z. Naturforsch. 62b, 955 (2007).
T. Nilges and M. Bawohl, Z. Naturforsch. 63b, 629 (2008).
S. Lange, M. Bawohl, and T. Nilges, Inorg. Chem. 47, 2625 (2008). doi:10.1021/ic701988u.
T. Nilges, S. Lange, M. Bawohl, J.-M. Deckwart, H.-D. Wiemhöfer, R. Decourt, B. Chevalier, J. Vannahme, H. Eckert, and R. Weihrich, Nat. Mater. 8, 101 (2009). doi:10.1038/NMAT2358.
J. Peters, O. Conrad, B. Bremer, and B. Krebs, Z. Anorg. Allg. Chem. 622, 1823 (1996). doi:10.1002/zaac.19966221105.
H.-J. Deiseroth and H. Mikus, Z. Anorg. Allg. Chem. 631, 1233 (2005). doi:10.1002/zaac.200500018.
J.S. Wu, K. Leinenweber, J.C.H. Spence, and M. O’Keeffe, Nat. Mater. 5, 647 (2006). doi:10.1038/nmat1687.
L. Palatinus, Acta Crystallogr. Sect. A 60, 604 (2004). doi:10.1107/S0108767304022433.
G. Oszlányi and A. Sütő, Acta Crystallogr. Sect. A 60, 134 (2004). doi:10.1107/S0108767303027569.
G. Oszlányi and A. Sütő, Acta Crystallogr. Sect. A 61, 147 (2005). doi:10.1107/S0108767304027746.
T. Nilges, C. Dreher, and A. Hezinger, Solid State Sci. 7, 79 (2005). doi:10.1016/j.solidstatesciences.2004.10.012.
T. Nilges and S. Lange, Z. Anorg. Allg. Chem. 631, 3002 (2005). doi:10.1002/zaac.200500261.
R. Ollitrault-Fichet, J. Rivet, J. Flahaut, and C.E. Kfouri, J. Solid State Chem. 110, 80 (1994). doi:10.1006/jssc.1994.1139.
M. Gobec and W. Sitte, J. Alloys Compd. 220, 152 (1995). doi:10.1016/0925-8388(94)06000-2.
I. Schewe and P. Böttcher, Z. Naturforsch. 45b, 417 (1990).
I. Schewe-Miller and P. Böttcher, Z. Kristallorgr. 196, 137 (1991).
K. Seifert-Lorenz and J. Haffner, Phys. Rev. B. Condens. Matter 66, 094105 (2002). doi:10.1103/PhysRevB.66.094105.
P. Böttcher, G. Getzschmann, and R. Keller, Z. Anorg. Allg. Chem. 619, 476 (1993). doi:10.1002/zaac.19936190309.
B. Eisenmann and H. Schäfer, Angew. Chem. 90, 731 (1978); Angew. Chem. Int. Ed. 17, 684 (1978). doi:10.1002/anie.197806841.
T. Ono, I. Yoshida, and M. Sakurai, Proceedings of the 19th International Conference on Thermoelectrics (2000), p. 370.
O. Osters and T. Nilges, unpublished results.
E. Ising, Z. Phys. 31, 253 (1925).
G.A. Papoian and R. Hoffmann, Angew. Chem. Int. Ed. 39, 2408 (2000). doi:10.1002/1521-3757(20000717)112:14<2500::AID-ANGE2500>3.0.CO;2-T.
M. Fujikane, K. Kurosaki, H. Muta, and S. Yamanaka, J. Alloys Compd. 393, 299 (2005). doi:10.1016/j.jallcom.2004.10.002.
G. Kalaprasad, P. Pradeep, G. Mathew, C. Pavithran, and S. Thomas, Compos. Sci. Technol. 60, 2967 (2000). doi:10.1016/S0266-3538(00)00162-7.
M. Hattori, Kolloidzeitschrift und Zeitschrift für Polymere (Colloid Polym. Sci.) 1, 11 (1964).
J. Janek, Nat. Mater. 8, 88 (2009). doi:10.1038/nmat2364.
J.-S. Rhyee, K.H. Lee, S.M. Lee, E. Cho, S.I. Kim, E. Lee, Y.S. Kwon, J.H. Shim, and G. Kotliar, Nature 459, 965 (2009). doi:10.1038/nature08088.
H. Yoshida, S. Ahlert, M. Jansen, Y. Okamoto, J.-I. Yamaura, and Z. Hiroi, J. Phys. Soc. Jpn. 77, 074719/1 (2008).
Acknowledgements
This project is financed by the DFG within the Collaborative Research Center SFB 458 Ionic motion in materials with disordered structures. We thank Netzsch, Germany for the measurement of the thermal diffusivity of Ag20Te10BrI.
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated to Prof. Dr. Dr. h.c.M. Jansen on the occasion of his 65th birthday.
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License ( https://creativecommons.org/licenses/by-nc/2.0 ), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Nilges, T., Bawohl, M., Lange, S. et al. Highly Dynamic Chalcogen Chains in Silver(I) (Poly)Chalcogenide Halides: a New Concept for Thermoelectrics?. J. Electron. Mater. 39, 2096–2104 (2010). https://doi.org/10.1007/s11664-009-0987-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11664-009-0987-9