Abstract
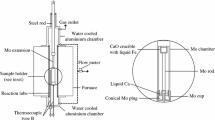
The dissolution equilibrium of calcium vapor in liquid iron was carried out at 1873 K in a two-temperature zone furnace using a vapor-liquid equilibration method. A sealed Mo reaction chamber and a self-made CaO crucible were used in this study. The thermodynamic parameters obtained are as follows.
For reaction Ca (g)=[Ca],
The relation between dissolved calcium in liquid iron and calcium vapor can be expressed as
The interaction parameters of third elements on calcium determined at 1873 K are as follows:
Similar content being viewed by others
References
Xiaodong Zhang, Qiyong Han, and Dong Chen: The 5th China-Japan Symp. on Science and Technology of Iron and Steel, International Academic Publishers, Shanghai, China, Dec. 1989, pp. 301–08.
Qiyong Han, Xiaodong Zhang, Dong Chen, and Penfei Wang: Metall. Trans. B, 1988, vol. 19B, pp. 617–622.
E. Schurmann and R. Schmid: Arch. Eisenhuttenwes., 1975, pp. 773–75.
E.T. Turkdogan: Physical Chemistry of High Temperature Technology, Academy Press, New York, NY, 1980, p. 7.
D.L. Sponseller and R.A. Flinn: Trans. TMS-AIME, 1964, vol. 230 (6), pp. 876–88.
T. Ototani, Y. Kataura, and T. Degawa: Tetsu-to-Hagané, 1975, vol. 61 (9), pp. 2167–81.
M. Kohler, H.J. Engell, and D. Janke: Steel Res., 1985, vol. 56 (8), pp. 419–23.
Yu.A. Ageev and S.A. Archugov: J. Phys. Chem., 1985, vol. 59 (4), pp. 838–841 (in Russian).
T. Fuwa and J. Chipman: Trans. AIME, 1959, vol. 215, p. 708.
M. Nadif and C. Gatellier: “Influence of Calcium and Magnesium Content on the Solubility of Various Non-metallic Elements in Liquid Steel,” IRSID paper Aclmcs 86 RE 1257, Apr 1986.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Song, B., Han, Q. Equilibrium of calcium vapor with liquid iron and the interaction of third elements. Metall Mater Trans B 29, 415–420 (1998). https://doi.org/10.1007/s11663-998-0119-6
Received:
Issue Date:
DOI: https://doi.org/10.1007/s11663-998-0119-6