Abstract
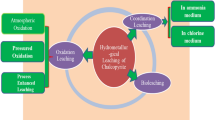
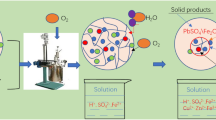
Due to the increasing complexity and dilution of copper resources, a large number of refractory polymetallic complex chalcopyrite are produced. In this study for resolving puzzle in oxygen pressure acid leaching of polymetallic complex chalcopyrite, such as prone to produce dangerous solid wastes like lead jarosite and high iron content in leaching solution, the technology of hydrothermal leaching under oxygen pressure without acid is proposed to extract copper efficiently and selectively and to precipitate iron by hematite process simultaneously. The results show that under the experimental conditions of initial sulfuric acid concentration of 0 g/L, reaction temperature of 200 °C, oxygen partial pressure of 1.2 MPa, liquid–solid ratio of 10 mL/g, sodium lignosulfonate addition of 0.5 pct mass of raw material, leaching time of 120 minutes, and stirring speed of 400 r/min, copper leaching rate can reach 99.86 pct. At this time, the iron content of leaching solution is only 4.3 g/L; Chalcopyrite (CuFeS2), porphyrite (Cu5FeS4), pyrite (FeS2), galena (PbS), and other mineral phases can completely react in the system to form corresponding metal sulfate, and Fe3+ is converted to hematite by directed hydrolysis, thus inhibiting the generation of dangerous solid waste such as lead jarosite.

















Similar content being viewed by others
References
B.W. Schipper, H.C. Lin, M.A. Meloni, K. Wansleebon, R. Heijunga, and E. Voet: Resour. Conserv. Recycl., 2018, vol. 132, pp. 28–36. https://doi.org/10.1016/j.resconrec.2018.01.004.
A. Elshkaki, T.E. Graedel, L. Ciacci, and B. Reck: Glob. Environ. Chang., 2016, vol. 39, pp. 305–15. https://doi.org/10.1016/j.gloenvcha.2016.06.006.
S. Raghavan: Int. J. Miner. Process., 1979, vol. 6(2), pp. 170–71. https://doi.org/10.1016/0301-7516(79)90026-7.
E.M. Córdoba, J.A. Muñoz, M.L. Blázquez, F. González, and A. Ballester: Hydrometallurgy, 2009, vol. 93(3–4), pp. 81–87. https://doi.org/10.1016/j.hydromet.2008.04.015.
H.R. Watling: Hydrometallurgy, 2013, vol. 140, pp. 163–80. https://doi.org/10.1016/j.hydromet.2013.09.013.
J. Zhou, Y.L. Liao, B.J. Li, and F.R. Huang: Chem. Ind. Eng. Prog., 2015, vol. 34(01), pp. 252–57. https://doi.org/10.16085/j.issn.1000-6613.2015.01.045.
Q. Liu and Y.H. Zhang: Miner. Eng., 2000, vol. 13(13), pp. 1405–416. https://doi.org/10.1016/S0892-6875(00)00122-9.
F. Nakhaei and M. Irannajad: Miner. Process. Extr. Metall. Rev., 2018, vol. 39(2), pp. 89–124. https://doi.org/10.1080/08827508.2017.1391245.
P. Huang, L. Wang, and Q. Liu: Int. J. Miner. Process., 2014, vol. 128, pp. 6–15. https://doi.org/10.1016/j.minpro.2014.02.004.
W.Q. Qin, Q. Wei, F. Jiao, N. Li, P.P. Wang, and L.F. Ke: Int. J. Min. Sci. Technol, 2012, vol. 22(3), pp. 345–49. https://doi.org/10.1016/j.ijmst.2012.04.011.
J. Esmaeil and G. Ahmad: Hydrometallurgy, 2017, vol. 171, pp. 333–43. https://doi.org/10.1016/j.hydromet.2017.06.011.
A.G.R. Toledo, S.P. Tayar, F.A. Arena, A.V. Benedetti, and D. Bevilaqua: Miner. Eng., 2022, vol. 180, p. 107467. https://doi.org/10.1016/j.mineng.2022.107467.
B.V. Ali, N. Sabereh, and D. Esmaeel: Miner. Eng., 2022, vol. 175, p. 107281. https://doi.org/10.1016/j.mineng.2021.107281.
P. Sandeep, A. Ata, P. Nilotpala, and D. Haci: Bioresour. Technol., 2015, vol. 196, pp. 694–706. https://doi.org/10.1016/j.biortech.2015.08.064.
C.L. Brierley: Hydrometallurgy, 2010, vol. 104(3–4), pp. 324–28. https://doi.org/10.1016/j.hydromet.2010.03.021.
E.M. Cordoba, J.A. Munoz, M.L. Blazquez, F. Gonzalez, and F. Ballester: Miner. Eng., 2009, vol. 22, pp. 229–35. https://doi.org/10.1016/j.mineng.2008.07.004.
R.P. Hackl, D.B. Dreisinger, E. Peters, and J.A. King: Hydrometallurgy, 1995, vol. 39, pp. 25–48. https://doi.org/10.1016/0304-386X(95)00023-A.
Y.J. Xian, S.M. Wen, J.S. Deng, Q. Liu, and J. Nie: Can. Metall. Q., 2013, vol. 51, pp. 133–40. https://doi.org/10.1179/1879139512Y.0000000001.
T.S. Qiu, G.H. Nie, J.F. Wang, and L.F. Cui: Trans. Nonferrous Met. Soc. China, 2007, vol. 17, pp. 418–22. https://doi.org/10.1016/S1003-6326(07)60108-3.
J.J. Wu, J. Ahn, and J. Lee: Miner. Process. Extr. Metall. Rev., 2020, vol. 2, pp. 1–8. https://doi.org/10.1080/08827508.2020.1795850.
W.U. Sf, C.R. Yang, W.Q. Qin, F. Jiao, J. Wang, and Y.S. Zhang: Trans. Nonferrous Met. Soc. China, 2015, vol. 25, pp. 4110–18. https://doi.org/10.1016/S1003-6326(15)64062-6.
M.B. Syott, H.R. Watling, P.D. Franzmann, and D. Sutton: Miner. Eng., 2000, vol. 13, pp. 1117–27. https://doi.org/10.1016/S0892-6875(00)00095-9.
J. Chaidez, J. Parga, J. Valenzuela, R. Carrillo, and I. Almaguer: Metal, 2019, vol. 9(2), p. 189. https://doi.org/10.3390/met9020189.
A.A. Baba, M.K. Ghosh, S.R. Pradhan, D.S. Rao, A. Baral, and F.A. Adekola: Trans. Nonferrous Met. Soc. China, 2014, vol. 24, pp. 1587–95. https://doi.org/10.1016/S1003-6326(14)63229-5.
R.G. Mcdonald and D.M. Muir: Hydrometallurgy, 2007, vol. 86, pp. 191–205. https://doi.org/10.1016/j.hydromet.2006.11.015.
L.L. Godirilwe, R.S. Magwaneng, S. Riku, H. Kazutoshi, B. Altansukh, A. Shogo, K. Takashi, M. Hidekazu, M. Kohei, K. Masanobu, and S. Atsushi: Miner. Eng., 2021, vol. 173, p. 107181. https://doi.org/10.1016/j.mineng.2021.107181.
Y.Y. Wang, H.F. Yang, G. Zhang, J.X. Kang, and C.L. Wang: Chem. Eng. J. Adv., 2020, vol. 3, pp. 100023–24. https://doi.org/10.1016/j.ceja.2020.100023.
J.G. Ryu: J. Hazard. Mater., 2022, vol. 427, pp. 128283–85. https://doi.org/10.1016/j.jhazmat.2022.128283.
Y.L. Bai, W. Wang, F. Xie, D.K. Lu, and K.X. Jiang: Trans. Nonferrous Met. Soc. China, 2022, vol. 32, pp. 1650–63. https://doi.org/10.1016/S1003-6326(22)65900-4.
G.X. Ji, Y.L. Liao, Y. Wu, J.J. Xi, and Q.F. Liu: J. Sustain. Metall., 2022, vol. 8, pp. 964–77. https://doi.org/10.1007/s40831-022-00561-5.
G. Owusu, D.B. Dreisinger, and E. Peters: Hydrometallurgy, 1995, vol. 38(3), pp. 315–24.
B. Han, B. Altansukh, K. Haga, and Y. Takasaki: J. Sustain. Metall., 2017, vol. 3, pp. 528–42. https://doi.org/10.1007/s40831-017-0135-3.
M. Chu, C.X. Li, P. Zhang, W.B. Ji, C. Wei, Z.G. Deng, X.B. Li, G. Fan, and M.T. Li: Chin. J. Nonferrous Met., 2020, vol. 30(5), pp. 1119–30. https://doi.org/10.11817/j.ysxb.1004.0609.2020-35783.
C.X. Li, C. Wei, S.W. Yi, G. Fan, Z.G. Deng, X.B. Li, and M.T. Li: Hydrometallurgy, 2019, vol. 189, p. 105112. https://doi.org/10.1016/j.hydromet.2019.105112.
K.Q. Xie, X.W. Yang, J.K. Wang, J.F. Yan, and Q.F. Sheng: Trans Nonferrous Met Soc China, 2007, vol. 17(1), pp. 187–94. https://doi.org/10.1016/S1003-6326(07)60070-336.
M.C. Ruiz, J. Zapata, and R. Padilla: Hydrometallurgy, 2008, vol. 89(1–2), pp. 32–39. https://doi.org/10.1016/j.hydromet.2007.05.00337.
Z.G. Deng, F. Yang, C. Wei, B.P. Zhu, P. Zeng, X.B. Li, C.X. Li, and M.T. Li: Trans Nonferrous Met Soc China, 2020, vol. 30, pp. 492–500. https://doi.org/10.1016/S1003-6326(20)65229-3.
C.X. Li, C. Wei, Z.G. Deng, X.B. Li, G. Fan, Y.Z. Wang, S.W. Yi, and M.T. Li: Chin. J. Nonferrous Met., 2018, vol. 28(3), pp. 628–36. https://doi.org/10.19476/j.ysxb.1004.0609.2018.03.23.
F. Kastury, W. Tang, C. Herde, M.R. Noerpel, K.G. Scheckel, and A.L. Juhasz: J. Hazard. Mater., 2021, vol. 418, p. 126312. https://doi.org/10.1016/j.jhazmat.2021.126312.
Z. Niu, G. Li, D. He, X. Fu, W. Sun, and T. Yue: J. Hazard. Mater., 2021, vol. 416, p. 125972. https://doi.org/10.1016/j.jhazmat.2021.125972.
R.L. Frost, R.A. Wills, M.L. Weier, A.W. Musumeci, and W. Martens: Thermochim. Acta, 2005, vol. 432, pp. 30–35. https://doi.org/10.1016/j.tca.2005.04.001.
C.X. Li, Z.G. Deng, C. Wei, G. Fan, X.B. Li, M.T. Li, and Y.Z. Wang: Hydrometallurgy, 2018, vol. 178, pp. 294–300. https://doi.org/10.1016/j.hydromet.2018.05.012.
F.X. Yang, Y.B. Xing, Z.G. Deng, C. Wei, X.B. Li, and M.T. Li: Int. J. Chem. Reactor Eng., 2021, vol. 19(10), pp. 1103–113. https://doi.org/10.1515/ijcre-2021-0010.
Z.G. Deng, B.P. Zhu, P. Zeng, C. Wei, X.B. Li, C.X. Li, and G. Fan: Can. Metall. Q., 2018, vol. 58(2), pp. 223–31. https://doi.org/10.1080/00084433.2018.1535929.
K. Kaplun, J. Li, N. Kawashima, and A.R. Gerson: Geochim. Cosmochim. Acta, 2011, vol. 75(20), pp. 5865–78. https://doi.org/10.1016/j.gca.2011.07.003.
S.M. Javad Koleini, V. Aghazadeh, and A. Sandström: Miner. Eng., 2011, vol. 24(5), pp. 381–86. https://doi.org/10.1016/j.mineng.2010.11.008.
H.S. Xu, C. Wei, C.X. Li, G. Fan, Z.G. Deng, X.J. Zhou, and S. Qiu: Sep. Purif. Technol., 2012, vol. 85, pp. 206–12. https://doi.org/10.1016/j.seppur.2011.10.012.
Acknowledgments
The authors express the sincere appreciation to the financial support of the National Natural Science Foundation of China (Project No. 21978122 and 21566017).
Conflict of interest
There is no ethical/legal conflict involved in the article.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Xi, J., Ji, G., Liao, Y. et al. Study on Selective Leaching of Copper and Simultaneous Precipitation of Iron in Polymetallic Complex Chalcopyrite by Hydrothermal Leaching Under Oxygen Pressure. Metall Mater Trans B 54, 2575–2590 (2023). https://doi.org/10.1007/s11663-023-02858-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11663-023-02858-6