Abstract
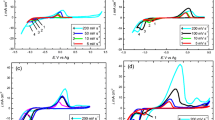
Cobalt chloride and neodymium chloride were dissolved in the urea and choline chloride deep eutectic solvents (DESs), and the electrochemical mechanisms of Co-Nd alloy codeposition were investigated under ambient conditions. Results from cyclic voltammetry indicated that the deposition of metallic Co on the tungsten electrode proceeds by an apparent two-electron transfer irreversible process at − 1.02 V (vs Ag). It is impossible to deposit pure Nd in urea and choline chloride DESs, whereas Co-Nd alloys can be codeposited from electrolytes with both Co(II) and Nd(III) ions. Furthermore, chronoamperometric experiments demonstrated that the electrochemical deposition of metallic Co can be considered as a three-dimensional (3-D) progressive nucleation process, while Co-Nd alloys were formed by instantaneous nucleation with diffusion-controlled growth. The average value of the diffusion coefficient of Co(II) ions in the urea and choline chloride DESs containing 3.8 × 10−5 mol/cm3 CoCl2 at 373 K was calculated to be 1.98 × 10−7 cm2/s. Moreover, analysis by scanning electron microscopy and energy dispersive X-ray spectroscopy suggested that a flat and uniform film consisting of Co (96.26 wt pct) and Nd (3.74 wt pct) was prepared by electrodeposition on a Cu substrate.








Similar content being viewed by others
References
A.I. Bhatt, I. May, V.A. Volkovich, D. Collison, M. Helliwell, I.B. Polovov, and R.G. Lewin: Inorg. Chem, 2005, vol. 44, pp. 4934–40.
C.J. Rao, K.A. Venkatesan, K. Nagarajan, T.G. Srinivasan, and P.R.V. Rao: Electrochim. Acta, 2009, vol. 54, pp. 4718–25.
S. Legeai, S. Diliberto, N. Stein, C. Boulanger, J. Estager, N. Papaiconomou, and M. Draye: Electrochem. Commun., 2008, vol. 10, pp. 1661–64.
D.W. Hatchett, J. Droessler, J.M. Kinyanjui, B. Martinez, and K.R. Czerwinski: Electrochim. Acta, 2013, vol. 89, pp. 144–51.
M. Matsumiya, Y. Kikuchi, T. Yamada, and S. Kawakami: Sep. Purif. Technol., 2014, vol. 130, pp. 91–101.
A. Kurachi, M. Matsumiya, K. Tsunashima, and S. Kodama: J. Appl. Electrochem., 2012, vol. 42, pp. 961–68.
T. Tsuda, T. Nohira, and Y. Ito: Electrochim. Acta, 2001, vol. 46, pp. 1891–97.
P. Kim, H.W. Xie, H.M. Gu, and Y.C. Zhai: Rare Met. Mater. Eng., 2012, vol. 41, pp. 599–602.
Y. Qiu, S.H. Yan, R.Y. Miao, L. Zhou, X.W. Zhang, and D.H. Chen: Chin. J. Rare Met., 2016, vol. 40, pp. 1126–31.
H. Yushanjiang, R.Q. Liu, and H.Y. Mi: Chin. J. Rare Met., 2014, vol. 38, pp. 432–40.
C.Y. Guo, J.C. Wang, B.Q. Chen, and J.G. Wang: Trans. Nonferr. Met. Soc., 2005, vol. 15, pp. 1190–93.
C.Y. Guo, J.C. Wang, J.G. Wang, and B.Q. Chen: J. Rare Earth, 2005, vol. 23, pp. 441–44.
P. Liu, Y.P. Du, Q.Q. Yang, Y.X. Tong, and G.A. Hope: J. Electrochem. Soc., 2006, vol. 153, pp. C57–C62.
P. Liu, Y.P. Du, Q.Q. Yang, G.R. Li, and Y.X. Tong: Electrochim. Acta, 2006, vol. 52, pp. 710–14.
J.G. Wang, J.C. Wang, B.Q. Chen, C.Y. Guo: J. Northwest Normal Univ. (Nat. Sci.), 2007, vol. 43, pp. 60–63.
Y.Y Yang, C.Y. Xu, Y.X. Hua, M.M. Wang, and Z.L. Su: Ionics, 2017, vol. 23, pp. 1703–10.
M. Li, Z.W. Wang, and R.G. Reddy: Electrochim. Acta, 2014, vol. 123, pp. 325–31.
L. Wang, Y.J. Fan, L. Wei, H.X. Liu, and S.G. Sun: J. Electrochem., 2015, vol. 21, pp. 543–47.
E. Gómez, P. Cojocaru, L. Magagnin, and E. Valles: J. Electroanal. Chem., 2011, vol. 658, pp. 18–24.
D.Y. Yue, Y.Z. Jia, Y. Yao, J.H. Sun, and Y. Jing: Electrochim. Acta, 2012, vol. 65, pp. 30–36.
H.Y. Yang, X.W. Guo, X.B. Chen, S.H. Wang, G.H. Wu, W.J. Ding, and N. Birbilis: Electrochim. Acta, 2012, vol. 63, pp. 131–38.
C.N. Su, M.Z. An, P.X. Yang, H.W. Gu, and X.H. Guo: Appl. Surf. Sci., 2010, vol. 256, pp. 4888–93.
B. Scharifker and G. Hills: Electrochim. Acta, 1983, vol. 28, pp. 879–89.
R.T. Carlin, P.C. Trulove, and H.C. Long: J. Electrochem. Soc., 1996, vol. 143, pp. 2747–58.
A.M.P. Sakita, R.D. Noce, C.S. Fugivara, and A.V. Benedetti: Phys. Chem. Chem. Phys., 2016, vol. 18, pp. 25048–25057.
F.R. Bento and L.H. Mascaro: J. Braz. Chem. Soc., 2002, vol. 13, pp. 502–09.
M.P. Pardavé, I. González, A.B. Soto, and E.M. Arce: J. Electroanal. Chem., 1998, vol. 443, pp. 125–36.
A. Sahari, A. Azizi, G. Schmerber, M. Abes, J.P. Bucher, and A. Dinia: Catal. Today, 2006, vol. 113, pp. 257–62.
X.L. Xie, X.L. Zou, X.G. Lu, K. Zheng, H.W. Cheng, Q. Xu, and Z.F. Zhou: J. Electrochem. Soc., 2016, vol. 163, pp. D537–D543.
F.G. Cottrell: Z. Phys. Chem., 1903, vol. 42, p. 385.
J.C. Wang, C.W. Xu, P. Liu, Y.X. Tong, and G.K. Liu: Trans. Nonferr. Met. Soc., 2002, vol. 12, pp. 1191–94.
R.T. Carlin, H.C. Long, J. Fuller, and P.C. Trulove: J. Electrochem. Soc., 1998, vol. 145, pp. 1598–1607.
A.N. Correia, R.C.B. Oliveira, and P.L. Neto: J. Braz. Chem. Soc., 2006, vol. 17, pp. 90–97.
R. Fukui, Y. Katayama, and T. Miura: Electrochim. Acta, 2011, vol. 56, pp. 1190–96.
Y.P. Lin and J.R. Selman: J. Electrochem. Soc., 1993, vol. 140, pp. 1304–11.
X.L. Xie, X.L. Zou, X.G. Lu, Q. Xu, C.Y. Lu, C.Y. Chen, and Z.F. Zhou: J. Appl. Electrochem., 2017, vol. 47, pp. 679–89.
Acknowledgments
The authors acknowledge the financial support from the Fundamental Research Funds for the Central Universities (Grant Nos. N182503033 and N172502003), China Postdoctoral Science Foundation (Grant No. 2018M640258), Guangxi Innovation Driven Development Project (Grant No. GUIKE AA18118030), National Science Foundation Grant No. DMR-1310072, and ACIPCO. We also thank the University of Alabama for providing the experimental and analytical facilities.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Manuscript submitted November 6, 2019.
Rights and permissions
About this article
Cite this article
Liu, A., Shi, Z. & Reddy, R.G. Electrochemical Synthesis of Co-Nd Films in Urea and Choline Chloride Deep Eutectic Solvents. Metall Mater Trans B 51, 1162–1168 (2020). https://doi.org/10.1007/s11663-020-01826-8
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11663-020-01826-8