Abstract
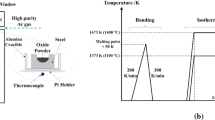
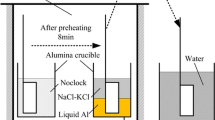
The kinetics of the high-temperature reaction between high-Al- and -Mn-containing steels and synthesized molten calcium silicate-based fluxes from 1623 K to 1643 K (1350 °C to 1370 °C) was studied. Cylindrical steel rods were rotated in the molten fluxes for 300 to 1200 seconds at various temperatures below the melting point of the steels. The rods were connected to a rheometer, and the initial reaction rates were estimated from the torque variations. The dissolution of the steel into the molten slag was correlated to the variation in torque. The kinetics of the reaction between the rods and the slag estimated from the torque and subsequently from the viscosity were confirmed from the mass balance and from the variation in the chemical compositions of the rods and the molten slags, respectively. The liquid-phase mass transfer coefficient of Al2O3 was calculated to be 1.14 × 10−2 cm/s at 1623 K (1350 °C) and 1.52 × 10−2 cm/s at 1633 K (1360 °C). The kinetics calculated assuming liquid-phase mass transfer control was observed to be similar to the aforementioned kinetics determined from the dynamic viscosity variations. On the basis of dimensionless analysis of the Sherwood number (Sh = 0.05·Re0.65Sc0.31), liquid-phase mass transfer from the metal/flux interface was observed to be the rate-controlling step.













Similar content being viewed by others
References
M.S. Kim, S.W. Lee, J.W. Cho, M.S. Park, H.G. Lee, and Y.B. Kang: Metall. Mater. Trans. B, 2013, vol. 44 (2), pp. 299-308.
Y.B. Kang, M.S. Kim, S.W. Lee, J.W. Cho, M.S. Park, and H.G. Lee: Metall. Mater. Trans. B, 2013, vol. 44 (2), pp. 309-316.
D.J. Kim and J.H. Park: Metall. Mater. Trans. B, 2012, vol. 43 (4), pp. 875-886.
Q. Wang, S. Qiu, and P. Zhao: Metall. Mater. Trans. B, 2012, vol. 43 (2), pp. 424-430.
M. Wang, Y. Kobayashi, R. Endo, and M. Susa: ISIJ Int., 2013, vol. 53 (1), pp. 56-61.
H. Kim and I. Sohn: ISIJ Int., 2011, vol. 51 (1), pp. 1-8.
H.S. Park, H. Kim, and I. Sohn: Metall. Mater. Trans. B, 2011, vol. 42 (2), pp. 324-330.
G.H. Kim and I. Sohn: J. Non-Cryst. Solids, 2012, vol. 358 (12-13), pp. 1530-1537.
Y.U. Kim and R.D. Pehlke: Metall. Trans., 1974, vol. 5, pp. 2527-2532.
X. Yu, R.J. Pomfret, and K.S. Coley: Metall. Mater. Trans. B, 1997, vol. 288 (2), pp. 275-279.
J.Y. Choi, H.G. Lee, and J.S. Kim: ISIJ Int., 2002, vol. 42 (8), pp. 852-860.
E.T. Turkdogan, Physical Chemistry of High Temperature Technology, Academic Press, New York, 1980.
A.H. Bui, H.M. Ha, I.S. Chung, and H.G. Lee: ISIJ Int., 2005, vol. 45 (12), pp. 1856-1863.
J.W. Robison and R.D. Pehlke: Metall. Trans., 1974, vol. 5 (5), pp. 1041-1051.
H.G. Katayama, T. Momono, M. Doe, and H. Saitoh: ISIJ int., 1994, vol. 34 (2), pp. 171-176.
J. Park, S. Sridhar, and R. Fruehan: Metall. Mater. Trans. B, 2015, vol. 46 (1), pp. 109-118.
B. Pesic and T. Seal: Metall. Trans. B, 1990, vol. 21 (3), pp. 419-427.
M. Lee, S. Sun, S. Wright, and S. Jahanshahi: Metall. Mater. Trans. B, 2001, vol. 32 (1), pp. 25-29.
Z. Asaki, M. Shiki, H. Kusuno, H. Kato, H. Doi, and Y. Kondo: Metall. Trans. B, 1986, vol. 17 (4), pp. 639-645.
R. Ohno: Metall. Trans., 1973, vol. 4 (4), pp. 909-15.
Acknowledgments
This study was supported by the Brain Korea 21 Plus (BK21 PLUS) Project at the Division of the Eco-Humantronics Information Materials and also supported (in part) by the Yonsei University Future-leading Research Initiative of 2015-22-0161 and the Ministry of Trade, Industry, and Energy 10044705.
Author information
Authors and Affiliations
Corresponding author
Additional information
Manuscript submitted June 12, 2015.
Rights and permissions
About this article
Cite this article
Kim, G.H., Sohn, I. Interfacial Kinetics of High-Al-Containing Ultra-Lightweight Steels with Calcium Silicate-Based Molten Oxides at High Temperature. Metall Mater Trans B 47, 1773–1784 (2016). https://doi.org/10.1007/s11663-016-0650-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11663-016-0650-9